Ceratomyxa arcuata Thélohan, 1892
|
publication ID |
https://doi.org/ 10.11646/zootaxa.3887.2.3 |
|
publication LSID |
lsid:zoobank.org:pub:B697D6BA-836B-44E2-A8D9-07661554FE59 |
|
DOI |
https://doi.org/10.5281/zenodo.5626240 |
|
persistent identifier |
https://treatment.plazi.org/id/038887C4-FFC6-FFDF-B3BB-F93C83FA4151 |
|
treatment provided by |
Plazi |
|
scientific name |
Ceratomyxa arcuata Thélohan, 1892 |
| status |
|
Ceratomyxa arcuata Thélohan, 1892
Type host: Gaidropsarus mediterraneus (syn. Motella tricirrata ), shore rockling ( Gadiformes : Gadidae ).
Other hosts: Pagellus bogaraveo (Brünnich, 1756) (syn. P. centrodontus ) blackspot sea bream; Callionymus lyra (Linnaeus, 1758) dragonet; Chromis chromis (Linnaeus, 1758) (syn. Heliases chromis ) damselfish; Crenilabrus melops (Linnaeus, 1758) corkwing wrasse; Dicentrarchus labrax (Linnaeus, 1758) Mediterranean sea bass; Gadus morhua (Linnaeus, 1758) Atlantic cod; Gobius paganellus (Linnaeus, 1758) rock goby; Merlangius merlangus (Linnaeus, 1758) whiting; Parophidium vasalli (Risso) (syn. Ophidium vasalli ); Sarpa salpa (Linnaeus, 1758) goldline sea bream; Scorpaena scrofa (Linnaeus, 1758) red scorpionfish; S. porcus (Linnaeus, 1758) black scorpionfish; S. notata (Rafinesque, 1810) small red scorpionfish; Uranoscopus scaber (Linnaeus, 1758) Atlantic stargazer.
Type locality: Roscoff, Atlantic coast of France.
Other localities: Northeast Atlantic, Black Sea, Adriatic Sea, Mediterranean Sea, Gulf of Tunis, Tunisia (36°45’N, 10°15’E).
Site of infection: Within gall bladder
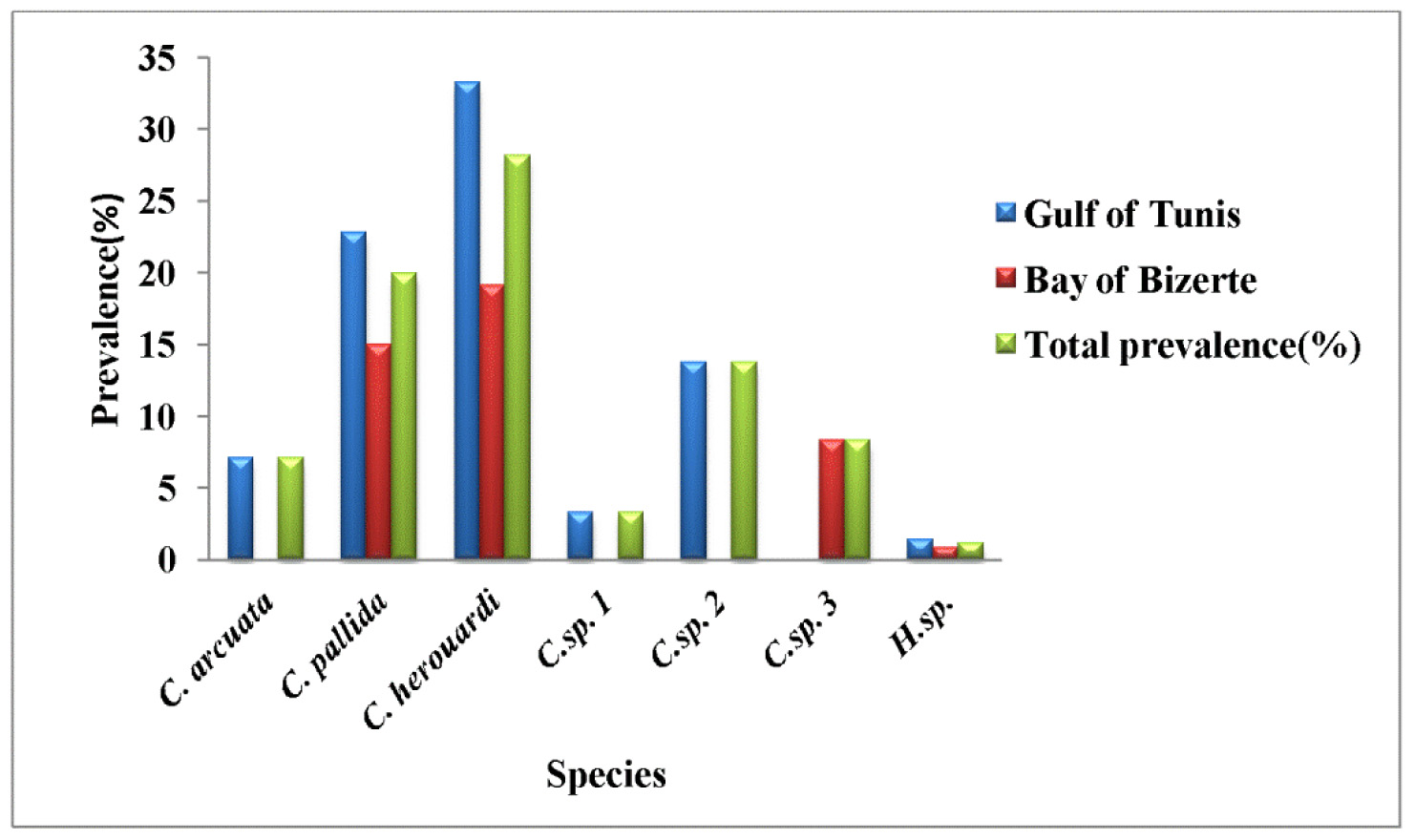
Prevalence: the overall prevalence is 4.5% (15/330) ( Fig. 9 View FIGURE 9 ). None fish 0% (0/120) recolted from Bay of Bizerte, was infected with C. arcuata . The infection was confined only at Gulf of Tunis with prevalence 7.14% (15/ 210) distributed as following, 03/2012: 0% (0/30); 04/2012: 0% (0/30);05/2012: 10% (3/30); 06/2012: 10% (3/30); 07/2012: 16.7% (5/30); 08/2012: 0% (0/30); 05/2013: 15% (3/20); 06/2013: 10% (1/10) (see Table 4).
Mean intensity: 70.2 ± 14.6 spores/infected fish (+++++) ( Fig. 10 View FIGURE 10 ) (see Table 4).
Type-material: Digitized photos of syntype spores were deposited in the parasitological collection of the Museum National d’Histoire Naturelle ( MNHN), Paris, Coll. No. ZS 116.
Description
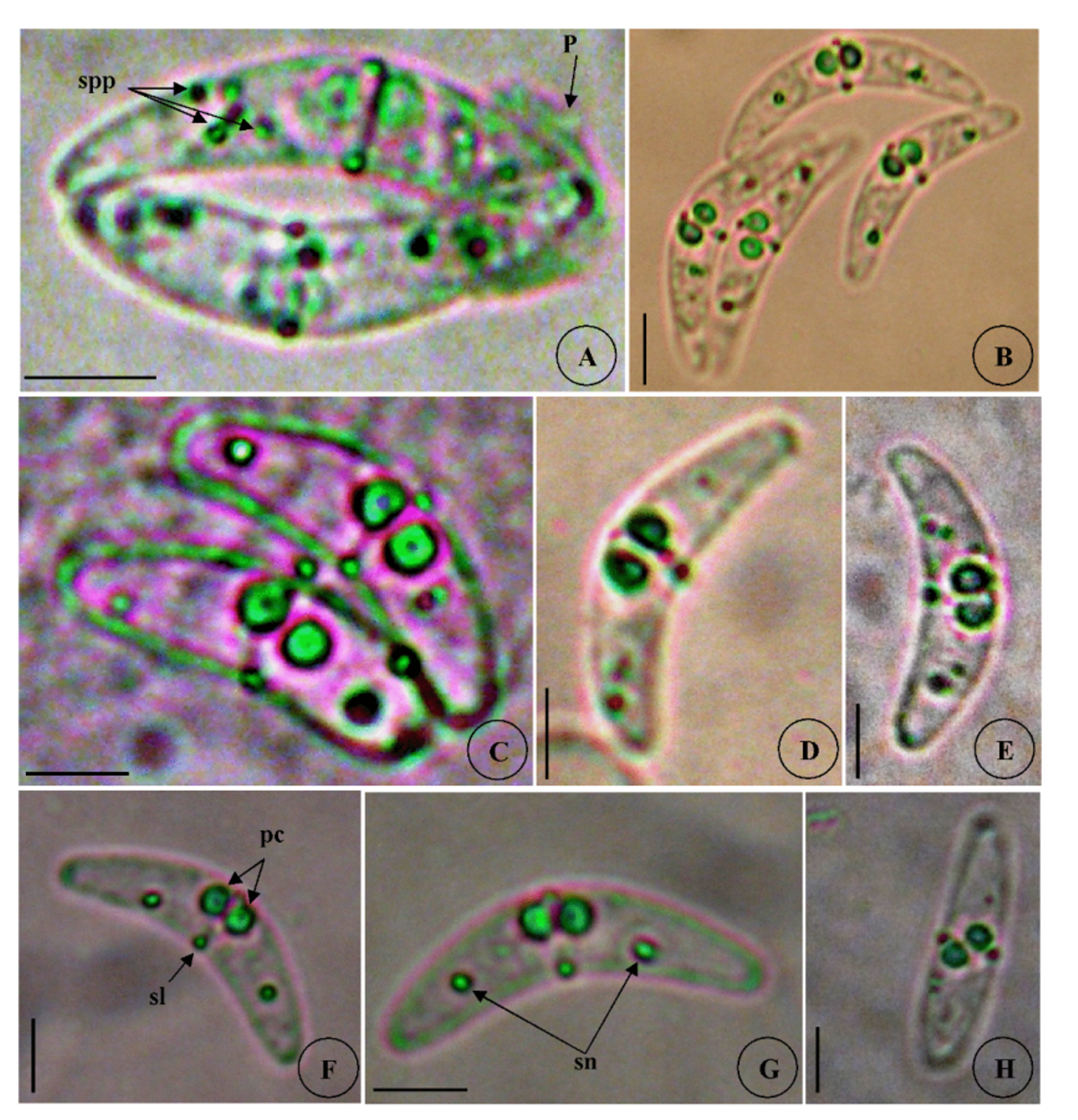
Vegetative stages. Polymorphous but generally club-shaped. Only one post-mature disporic plasmodium was seen in this study when the spores were being separated ( Fig. 1 View FIGURE 1 A).
Spores (n = 30 fresh spores). Mature spores were arcuate in sutural view ( Figs. 1 View FIGURE 1 A–E, 8A) and elongated in apical view ( Fig. 1 View FIGURE 1 H), measuring 7.5 ± 0.4 (7–9) Μm in length and 35.6 ± 3.3 (30–40) Μm in thickness. Posterior angle was concave 150.6 ± 4.2 (142–156°). Valves equal slightly tapering with rounded ends and smoothly ovoid in lateral view ( Fig. 1 View FIGURE 1 G). Straight sutural line visible between the two valves. A binucleate sporoplasm, with several sporoplasmosomes, granular in which remnants of valvogenic nuclei persist at tips of the shell valves ( Figs. 1 View FIGURE 1 F,8A). Polar capsules were subspherical to pyriform, 3.3 ± 0.4 (3–4) Μm in length and 3 ± 0.4 (2.5-3.5) Μm in width (n = 30). Four to five coils of polar filaments could be observed inside the polar capsules.
Remarks
According to the available literature, C. arcuata Thélohan, 1892 was identified as the first typical species described from the myxosporean genus Ceratomyxa . It was originally reported from several hosts in different families of fishes from Atlantic and Mediterranean off France and Mediterranean off Italy ( Kudo 1920). Until now, in addition to the present work, C. arcuata Thélohan, 1892 has been described in 10 different families of fish ( Callionymidae , Gadidae , Gobiidae , Labridae , Moronidae , Ophidiidae , Scorpaenidae , Sparidae , Pomacentridae , Uranoscopidae ) ( Kalavati & Mackenzie 1999). This confirm that C. arcuata is euryxenous, infecting a huge range of unrelated hosts. However, Kalavati & Mackenzie (1999) believed that some host could be infected accidently by C. arcuata Thélohan, 1892 due to weak intensity and prevalence of infection. Nevertheless, Mackenzie et al. (2005) revealed that C. arcuata can be suitable as biological tags for stock discrimination of some fish species. Moreover, this myxosporean has the most worldwide distribution. It has been reported from different areas around the world as the Northeast Atlantic and a record of 15 different fish species has been infected with C. arcuata (Thélohan 1892,1895; Dunkerly 1921; Théodoridès 1955; Noble 1957; Kabata 1967; Shotter 1970; Feist & Ronga 1996; Kalavati & Mackenzie 1999; Mackenzie et al. 2005), Mediterranean Sea ( Thélohan 1892, 1895; Parisi 1912; Jameson 1913; Georgèvitch 1916; Present study), Black Sea ( Zaika 1966) and Adriatic Sea ( Lubat et al. 1989). The absence of reports of C. arcuata from the Northwest Atlantic suggests that this species is restricted to temperate waters of the Northeast Atlantic ( Kalavati & Mackenzie 1999; Mackenzie et al. 2005). Historically, the difference in the published descriptions suggested the possibility to include more than one species of Ceratomyxa . On the basis of the presence or absence of small refringent globules in the trophozoite and the size of the pseudopodia, Labbé (1899) divided C. arcuata into two subspecies: C. arcuata typica , from P. bogaraveo , C. melops and G. mediterraneus and C. arcuata scorpaenarum , from S. scrofa and S. porcus , but this division was rejected by Parisi (1912) and Kudo (1920) as being too arbitrary. Both Parisi (1912) and Meglitsch (1960), noted in their descriptions that the specimens of C. arcuata were smaller with valves of unequal length. Mackenzie et al. (2005) declared that the differences between the description of Meglitsch and both the original description of C. arcuata and the redescription of Kalavati & MacKenzie (1999) suggested that Meglitsch was mistaken in his identification of “ C. arcuata ” from the serranid Anthias punchellas from New Zealand waters. The author is in perfect agreement with this declaration since the comparison between the measurements of the current species and the species identified by Meglitsch (1960) shows no similarity neither in shape nor in size. In this study, the features characteristic of C. arcuata with both sporogonic stages (Disporous) and the mature spores (Equal valves) are in agreement with the original description of Thélohan (1895) and the study of Kalavati & Mackenzie (1999). Likewise, this study records the goldline sea bream S. salpa (L.) as a new host of C. arcuata from the family of sparidae from the Mediterranean Sea after the blackspot sea bream P. bogaraveo (Brünnich, 1756) .
Ecological notes
In this study, based to the classification of parasites by Valtomen et al. (1997), C. arcuata has a parasitic status as scarce species (P <10%). Infection by C. arcuata was confined only in Gulf of Tunis. During the sampling period, the highest prevalence of infection was noted in July 16.7% (see Table 4) and mean intensity was moderate with 70.2 ± 14.6 spores per infected fish ( Fig. 10 View FIGURE 10 ).
| MNHN |
Museum National d'Histoire Naturelle |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.