Zagymnus clerinus ( LeConte, 1873 )
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4457.3.6 |
|
publication LSID |
lsid:zoobank.org:pub:0DED6B72-F735-48AD-B686-7A858AD7BA14 |
|
DOI |
https://doi.org/10.5281/zenodo.6490797 |
|
persistent identifier |
https://treatment.plazi.org/id/038887EA-400A-CE3B-FF24-B8F9FD9BF9DB |
|
treatment provided by |
Plazi |
|
scientific name |
Zagymnus clerinus ( LeConte, 1873 ) |
| status |
|
Zagymnus clerinus ( LeConte, 1873) View in CoL
[ Figs. 5–9, 11 View FIGURES 5–8. 5 View FIGURES 9–11. 9 ]
History. Zagymnus clerinus was described from a specimen from Florida. It has only been collected once in Cuba ( Devesa et al. 2015; Devesa pers. comm., 2018) and there are only two records published from Georgia ( Fattig 1947). C. V. Riley (1880) stated about Zagymnus clerinus : “bores in the dry leaf stems of Chamaerops palmetto , in Florida, the beetles appearing in April and May.”
Biological notes. This species is typically found with color patterns as in Figs. 5–7 View FIGURES 5–8. 5 . The pronotum may be either reddish-orange or black. Elytral markings may occasionally be reduced in size or, rarely, completely absent as in Figs. 8 View FIGURES 5–8. 5 & 9 View FIGURES 9–11. 9 . Individuals have scattered, whitish hairs on the body.
In Florida, adults can be found from April to July, usually on Cabbage Palmetto ( Sabal palmetto (Walter) Lodd. ex Schult. & Schult. f.). Long series of Zagymnus were collected on S. palmetto , particularly those which had been fire-damaged, in Pinellas County, Florida (R. Morris, II, R. M. Brattain, J. A. Green pers. comm.), indicating they have a strong attraction to damaged plants, or their vascular secretions. Sabal palmetto naturally ranges throughout Florida and the coastal plains of Georgia, South Carolina, and southeastern North Carolina but has also been introduced throughout the gulf coast as an ornamental ( BONAP 2014).
A single, freshly crushed specimen of Z. clerinus was discovered by one of the authors (Gemmill) in a campus building corridor at the Medical University of South Carolina on 3-VII-2007 (new state record). RG noted S. palmetto trees growing near Charleston, SC with exit holes in their leaf petioles similar to those of the A. lepturoides he observed during a joint trip to the SPS with the Texas authors. These S. palmetto trees were located within or near the maritime forest of the South Carolina coast. In June of 2016, adult Z. clerinus were discovered on the shoreline of Edisto Island, SC hiding behind leaf petioles on S. palmetto trees.
Two Z. clerinus were captured in Lindgren funnel traps in the SPS in Texas (VII-2009 and VII-2010) by Heffern and Raber (DJHC & BTRC) (new state record). A desiccated adult was found in a pupal chamber in a dead leaf stalk of S. mexicana (new host record) at the SPS, II-2017 by Heffern. That same leaf stalk was also populated by an active colony of ants. The dead Zagymnus was located in the leaf stalk about 0.75 m from the trunk. Zagymnus clerinus was recently recorded from Galveston Island, Texas, (iNaturalist 2018).
In South Carolina, sites in Charleston, Colleton, and Georgetown counties with S. palmetto trees were surveyed for evidence of Z. clerinus by Austin. Surveying was done between VI-2016 and X-2017 by visually inspecting all S. palmetto trees ( Fig. 12 View FIGURE 12–14. 12 ) for Z. clerinus emergence holes and live adults. The age and abundance of the emergence holes, as well as the presence of any transplanted palmettos with emergence holes, was noted. All observations of live Z. clerinus in their natural habitat were made during daylight hours on Pockoy Island, a small barrier sandspit within the Botany Bay Wildlife Management Area, on Edisto Island, SC. Petioles displaying evidence of previous usage by Z. clerinus larvae, chiefly emergence holes, were collected from native Cabbage Palmettos on private land. Larval tunnels were examined by longitudinally splitting Palmetto petioles that exhibited Z. clerinus emergence holes. The dimensions of larval tunnels were measured and pupal chambers were examined. Several additional petioles that lacked emergence holes were placed into a mesh enclosure to capture any adults that might emerge the following summer.
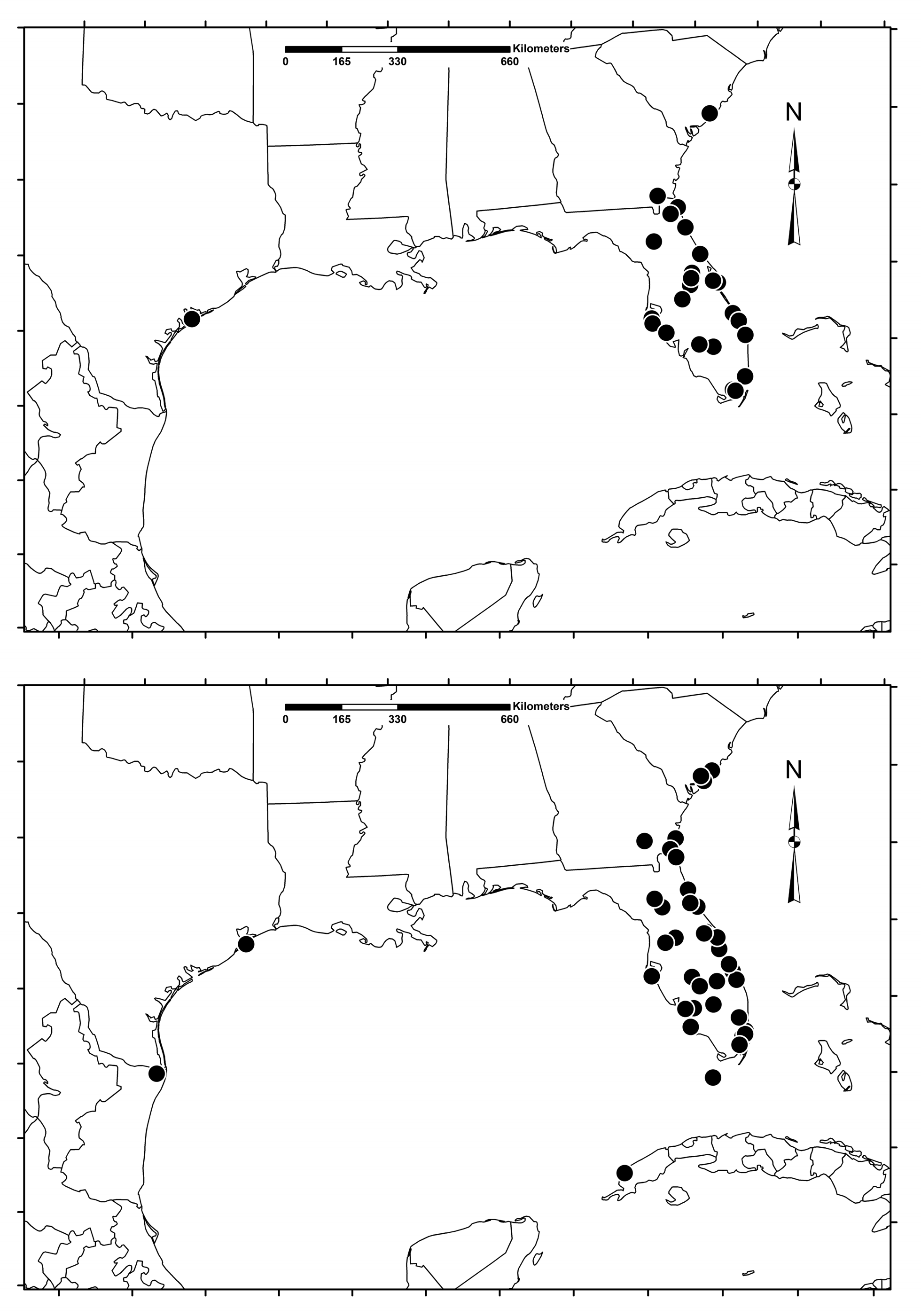
Five adult Z. clerinus were observed and three specimens collected between 2007 and 2017 at three sites in Charleston County, SC. Additional indirect evidence at numerous other sites suggests that Z. clerinus populations in South Carolina are located throughout Charleston County within the saltwater tidal zone, from Edisto Beach to Isle of Palms.
The larvae utilize the petioles and forked bases (boots) of senesced, desiccated petioles that remain attached to the palm trunk. The larvae consume the longitudinal fibers of the petiole and move down the petiole towards the base at the trunk. The larval burrow is densely packed with frass. Each petiole can support two larvae, one on either side of the sagittal plane of the petiole. Larval burrows are found below the epidermis and oblique fibers of the petiole. They are typically 30cm long, 3cm wide, and 1cm deep, although dimensions vary with the shape of the host petiole. There is a clear preference for petioles that senesced over a year prior and show outward signs of degradation. This degree of degradation is visible as a dull, light gray color on the exterior. Occasional emergence holes in old inflorescence stalks indicates this tissue is also suitable for larval development. Zagymnus clerinus is not known to be harmful to the host tree as the larvae consume only dead plant material.
Pupation begins in late spring with adults emerging in early summer. After eclosing, adults exit the host petiole through an elliptical emergence hole averaging 5 to 9mm in length, and parallel to the grain of the wood ( Fig. 12 View FIGURE 12–14. 12 ). Emergence holes are most often located 10 to 20 centimeters below the fork of the petiole in S. palmetto and most commonly on the underside of the petiole. Adults are diurnal and have been observed resting and ovipositing on S. palmetto throughout the day ( Fig. 11 View FIGURES 9–11. 9 ). Adults have been found resting, or possibly hiding, between the petiole base and the trunk near the tops of some palmetto trees. Females oviposit rather indiscriminately on the edges and broken tips of petioles and have been observed to oviposit on both live fronds and desiccated petioles. However, larvae have not been found to utilize living or recently senesced fronds. Adults are short-lived and die within a few weeks of emergence.
The distinctly shaped emergence holes of this species has allowed for the tracking of its distribution within South Carolina. These holes can expand over time due to weathering or use by other insects. Pupal chambers of Z. clerinus are utilized by other arthropods after emergence of the beetle. Parancistrocerus histrio (Lepeletier, 1841) , a species of Mason Wasp ( Hymenoptera : Vesperidae : Eumeninae) native to Coastal SC, GA, and much of FL, has been observed to nest within these abandoned pupal chambers. Several species of small spiders ( Araneae ) have also been observed using the empty pupal chambers as a daytime refuge. The resultant wear and weathering of the exit holes provides an additional means to estimate the relative age of a given population.
Zagymnus clerinus has a marked preference for host palmettos that receive direct sunlight and grow in close proximity to saltwater tidal systems. The age and height of palmettos appears to be an unimportant factor in host selection, so long as the tree has sufficiently deteriorated boots attached to its trunk. Additionally, Z. clerinus appears to have a strong preference for stunted palmettos. These trees are typically found on the edges of salt marsh hammocks, or similar habitats with poor growing conditions. Such stunted palmettos have substantially reduced leaf size, shorter overall height, increased leaf senescence rates, and an overall unhealthy appearance. It is not uncommon for there to be two-dozen or more emergence holes on a stunted palmetto less than two meters in height. The harsh growing conditions these trees reside in and their general poor health results in wood that decomposes relatively rapidly. This accelerated decomposition may create wood more easily digested by Z. clerinus larvae. This species is also present in shaded understory palmettos in South Carolina but at a substantially lower density than on host trees found in more exposed locations. In Texas, Z. clerinus was found at the SPS on palms in shaded understory, many miles from saltwater.
Sabal palmetto trees reduce their growth rate after they are transplanted, resulting in a distinct compression of the layers of boots encircling the trunk. Boot compression is visible as an alteration in the angle that the petioles make with the trunk, providing a convenient means to identify transplanted trees. The condensed boot ring is also useful in assessing the status of any associated population of Z. clerinus , since emergence holes above this band indicate persistence of Z. clerinus after transplantation. Transplantation of palmettos to locations above the saltwater tidal zone typically resulted in the loss of Z. clerinus larvae hosting on the tree. This conclusion is based on the presence of emergence holes in petioles older than the transplantation band but absent above this band. In contrast, palmettos transplanted to barrier islands or waterfront properties within the saltwater tidal zone often showed evidence of continuous use as a host, extending for years after transplantation ( Fig. 13 View FIGURE 12–14. 12 ). This is consistent with a larval cold intolerance that can be mitigated in proximity to saline water bodies.
The density of Z. clerinus emergence holes was greatest in areas where palmettos exist undisturbed in their natural habitat. Protected areas of coastal maritime forest near barrier island communities exhibited the highest densities of emergence holes. Manicured trees used in landscapes exhibited much lower densities, perhaps because their petioles are trimmed to just above the fork, removing a significant portion of suitable host material.
Our findings extend the range of Z. clerinus to include the South Carolina coastal plain and coastal Texas. Evidence from emergence holes on recently transplanted landscape palmettos suggests that the species was introduced to South Carolina by anthropogenic means and that this is a major factor in its current distribution. However, Z. clerinus has demonstrated the ability to naturally disperse and establish itself in new habitats within the state. Self-sustaining populations have been discovered at several protected or isolated sites where no landscape palmettos have been imported. Nevertheless, its natural dispersal ability appears to be limited, as several uninhabited barrier islands near likely populations showed no evidence of its presence. Zagymnus clerinus is likely present in Beaufort and Jasper counties, due to their significant coastal development and milder winters, but these counties were not surveyed. No populations were discovered in Georgetown or Conway counties and the colder winters of these counties may severely reduce larval survival. Zagymnus clerinus may still exist in isolated sites in these areas but these counties were not surveyed comprehensively. Further work will be required to fully understand the distribution of Z. clerinus in coastal South Carolina.
Distribution. A distribution map ( Fig. 16 View FIGURE 15–16. 15 ) for Zagymnus clerinus was prepared based on records from DJHC, FSCA, TAMU, RFMC, RMBC, RMGC, TBAC, Devesa (2015), Fattig (1947), and iNaturalist (2018).
Summary. With widespread planting of numerous palm species for landscaping, all three Agallissini species known from the United States can disperse to areas where they are not native. Increased awareness by collectors, naturalists, and photographers will likely result in additional observations for these species, especially now that their life histories are being documented. There are no observations to indicate that any of these species are a threat to healthy plants. Just the opposite, Agallissini appear to be important natural agents in the decomposition of fibrous, dead leaf stalks, facilitating the shedding of dead leaves by palmettos and returning the mineral nutrients trapped within to the soil below.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |