Scinax cosenzai, Lacerda, João Victor A., Peixoto, Oswaldo Luiz & Feio, Renato N., 2012
|
publication ID |
https://doi.org/ 10.5281/zenodo.280807 |
|
DOI |
https://doi.org/10.5281/zenodo.5626066 |
|
persistent identifier |
https://treatment.plazi.org/id/0397F417-FF8A-FFF7-8FF2-FBA1BA3DFDB5 |
|
treatment provided by |
Plazi |
|
scientific name |
Scinax cosenzai |
| status |
sp. nov. |
Scinax cosenzai View in CoL sp. nov.
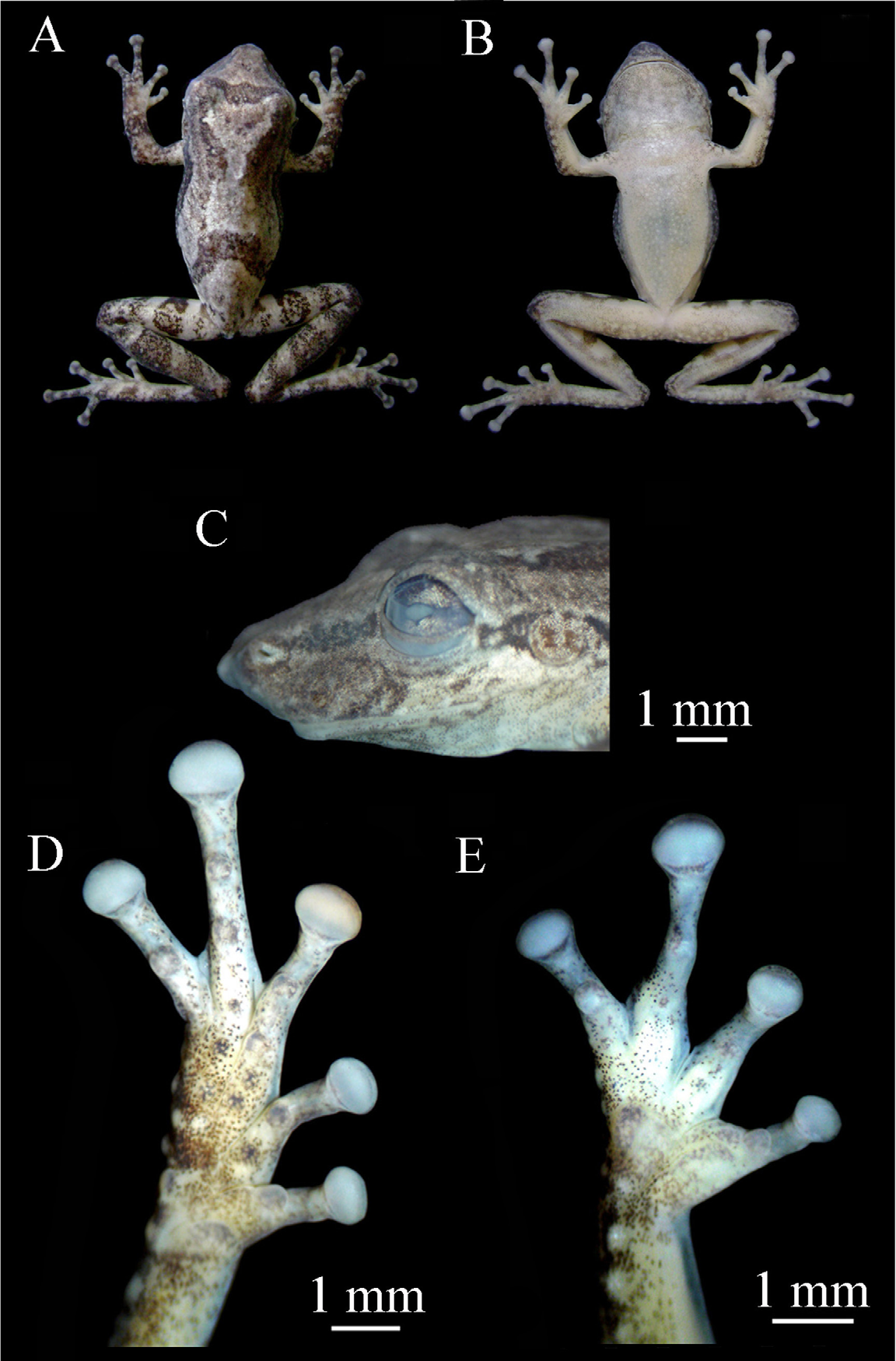
( Figs 1 View FIGURE 1 , 2 View FIGURE 2 , 3 View FIGURE 3 )
Holotype. MNRJ 75462, adult male, collected at Lage das Bromélias, Serra do Brigadeiro (20º43’13.5´´ S and 42º28’48.7´´ W; 1385 m above sea level), Municipality of Araponga, state of Minas Gerais, Brazil. Collected by J.V.A. Lacerda, B. Assis and J.S. Dayrell on October 28, 2007. GoogleMaps
Paratopotypes. MZUFV 3895, adult male collected by Feio, R.N. and Santos-Filho, J.B, on 0 3 September 1999; MZUFV 6653, adult male collected by R.N. Feio, E.F. Oliveira and J.S. Dayrell, on 0 5 December 2005; MZUFV 8275, adult male collected by J.V.A. Lacerda and B. Assis, on 29 January 2008; MNRJ 75463– 64, adult males collected by Feio, R.N. and Santos-Filho, J.B, on 0 3 September 1999; MNRJ 75465, adult male collected by R.N. Feio, E.F. Oliveira, H.C. Costa and L.L. Moraes, on 22 September 2005; MNRJ 75466, adult male collected by C.S. Cassini and R.N. Feio, on 21 February 2006.
Referred specimens. Scinax cosenzai sp. nov.: BRAZIL: State of Minas Gerais: Municipality of Araponga (topotypes): MZUFV 4956, 6654, 7682, 7779, 8029, 8092, 8093, 8567, 8568, 11458–11463, 11470–11473); Municipality of Ervália: Distrito do Careço (20°51’9.24’’ S and 42°31’5.56’’ W; 980 m above sea level): MZUFV 11532–11537, GoogleMaps 11541–11543. GoogleMaps
Diagnosis. Scinax cosenzai sp. nov. belongs to the S. perpusillus group based on the use of bromeliads for reproduction and reduced webbing between toes II and III. The new species is characterized by: (1) moderate-size within the group (males 17.54–20.97 mm SVL, females 22.65–24.02 mm SVL); (2) snout protruding in lateral view and slightly acuminated in dorsal view; (3) snout with a distinct medial process, defined by Silva and Alves- Silva (2008) as a protuberance caused by the cartilaginous medial prenasal process of the nasal tectum under the skin; (4) medium-sized, elliptical choanae; (5) vomerine teeth in two arc shape series between choanae; (6) head longer than wide; (7) protruding eye; (8) canthus rostralis distinct; (9) loreal region concave; (10) tympanum round and pigmented, with diameter less than half the diameter of the eye; (11) thigh plus tibia length shorter than SVL; (12) webbing lacking between toes I and II and vestigial between II and III; (13) few tubercles scattered throughout the dorsal surface, including the head; (14) ventral skin granulated, more intensively near the cloacal region; (15) dorsum different tones of gray with transversal dark bars originating on each inguinal region and converging on the third portion of the dorsum in an inverted V-shaped figure; (16) lateral dark stripe, from the region of the eye to the anterior margin of the inguinal region; (17) head with an interocular dark transversal bar; (18) transversal dark bars along the anterior and posterior members; (19) discreet nuptial glandular pads on the base of thumb; (20) gular region with irregular dark pigmentation; (21) in life, hidden portion of the thigh and tibia region with some irregular bright yellow colored patches ( Fig. 3 View FIGURE 3 ); (22) a distinct vocalization with heterogeneous multipulsed notes (2–14 notes/call, call duration 177.62–2066.75 ms, 1–33 pulses/note and dominant frequency of 3375.91–4571.21 Hz).
Advertisement call. We analyzed a total of 45 calls recorded at the type locality belonging to three call series emitted by three individuals that were not collected. We were unable to observe direct interaction of these males with other males and females during the recordings. The duration of the call series was 34.4, 55.1 and 68.9 s, with 11, 16 and 18 calls/call series, call rate of 0.25, 0.29 and 0.32 calls/sec and interval between calls of 2.78 s (1.9–4.32; SD=0.64; n=42 intervals). Calls are constituted by 2–14 multipulsed notes with duration of 177.62–2066.75 ms and dominant frequency of 3375.91–4571.21 Hz ( Table 1 View TABLE 1 ). Note duration of 36.15 ms (1.81–111.56; SD=23.94; n=236 notes), 10.53 pulses/note (1–33; SD=7.08; n=236 notes) and rate of 315.12 pulses/ second (58.60–571.42; SD=103.08; n=236 notes). Notes are heterogeneous in duration, number of pulses and amplitude. Figure 4 View FIGURE 4 shows the sonogram of the call series with 11 calls emitted by a single male and sonograms and spectrograms of each of these calls.
Comparison with other species. Adult individuals of Scinax cosenzai sp. nov. may be distinguished from other species of the Scinax perpusillus group by a combination of traits. The new species differs from S. alcatraz by its smaller size (SVL 19.7–24.4 mm in males and 27.0– 29.8 mm in females of S. alcatraz ; Brasileiro et al. 2007a) and by lacking inguinal glands (easily visible in S. alcatraz ; Faivovich et al. 2010). Scinax cosenzai sp. nov. can be distinguished from S. arduous by its smaller size [19.3–22.7 mm (x=21.0; SD=0.8; n=20) in males of S. arduous ], slender body, less protruding eyes, more evident canthus rostralis, loreal region more concave, THL + TBL shorter than SVL (longer in S. arduous ) and a distinct advertisement call. It differs from S. atratus by the larger size of the female (maximum SVL of females of S. atratus 20.0 mm; Peixoto 1988b) and by its dorsal color pattern typical of the S. perpusillus group (melanic in S. atratus ; Peixoto 1988b). The new species is promptly distinguished from S. belloni by not possessing dorsum of body and limbs densely covered by prominent granules, having trace of markings on dorsum, inguinal region and hidden surfaces of hind limbs, lacking inguinal glands and by the smaller size (19.8–23.0 mm SVL in males and 26.3–29.2 in females of S. belloni ; Faivovich et al. 2010). It differs from S. faivovichi by its larger SVL (SVL 16.2–18.0 mm in 23 males of S. faivovichi and 18.6–21.7 mm in six females; Brasileiro et al. 2007b), furthermore S. faivovichi has brown undersurface of tarsus and foot (not brown in the other species of the S. perpusillus group; Brasileiro et al. 2007b). Scinax cosenzai sp. nov. is promptly distinguishable from S. insperatus by the presence of a yellow flash color on inguinal region and hidden surfaces of thigh and tibia (absence of yellow markings in the inguinal region and hidden surfaces of limbs in S. insperatus ; Silva & Alves-Silva 2011) and by its smaller SVL (18.5–21.9 mm in males of S. insperatus ; Silva & Alves-Silva 2011). The new species differs from S. littoreus by lacking inguinal glands (easily visible in S. littoreus ; Faivovich et al. 2010), by the less pronounced eyes, dorsal color pattern, snout profile and width of the head (dorsum brownish colored, rounded snout profile and wider head in S. littoreus ). It differs from S. melloi by its larger size (maximum SVL in males of S. melloi 17.00 mm and 18.7 mm in females; Peixoto 1988b) and by its dorsal color pattern typical of the S. perpusillus group (marble dorsal pattern in S. melloi ; Peixoto 1988b). Scinax cosenzai sp. nov. can be distinguished from S. peixotoi by having tibia larger than thigh (tibia same size as thigh in S. peixotoi ; Brasileiro et al. 2007a), slender body and by the distinct advertisement call. It differs from S. perpusillus by its slender body, less pronounced eyes, snout more acuminated and a distinct advertisement call. The new species is distinguishable from S. tupinamba by the larger size [SVL of 17.8 mm in S. tupinamba (16.1–19.2; SD=0.9; n=21); Silva & Alves- Silva 2008], loreal region less concave and by the absence of a yellow stripe on the head of the tadpoles of the new species (the tadpole of S. tupinamba has a distinct, large, and bright yellow stripe between the eyes and the nostrils; Silva & Alves-Silva 2008). Scinax cosenzai sp. nov. differs from S. v-signatus by its smaller SVL [21.4–23.5 mm (x=22.4; SD=0.7; n=20) in males of S. v-signatus ], less rugose dorsal skin texture, canthus rostralis less evident, loreal region less concave and with less tubercles. Besides, S. v-signatus has a dark pigmentation on the gular region organized in the form of a Y or V ( Lutz 1973; Peixoto 2002), and has a distinctive orange-flash color on the foot, lower leg, thigh, inguinal region, and armpit (H. R. da Silva, pers. com.). Scinax cosenzai sp. nov. has irregular dark pigmentation on the gular region and presents a yellow-flash color only on inguinal region and hidden surfaces of thigh and tibia, lacking on armpits and foot.
The advertisement call of S. peixotoi (3–5 notes and duration of 146–232 ms; Brasileiro et al. 2007a) is shorter than that of the new species (the calls of Scinax cosenzai sp. nov. with 3–5 notes have duration of 512.75–941.81 ms). The advertisement call of S. arduous (4–6 notes and duration of 198–328 ms; Pombal & Bastos 2003) is shorter than that of the new species (the calls of Scinax cosenzai sp. nov. with 4–6 notes have duration of 812.25–1395.75 ms). The territorial call of S. arduous differs from the vocalization of the new species in having only one note ( Pombal & Bastos 2003) (the vocalization of Scinax cosenzai sp. nov. has at least 2 notes). The advertisement call of S. perpusillus (3–6 notes and duration of 92–174 ms; Pombal & Bastos 2003) is shorter than that of the new species (the calls of Scinax cosenzai sp. nov. with 3–6 notes have duration of 512.75–1395.75 ms). Pombal and Bastos (2003) described a second type of call of S. perpusillus , also shorter than that of the new species, with 6–12 notes and duration of 314–400 ms (the calls of Scinax cosenzai sp. nov. with 6–12 notes have duration of 892.81–2066.75 ms). These authors also mentioned the existence of a third type of call with only one note and duration of 185s, 435s and 508s (the calls of Scinax cosenzai sp. nov. have at least two notes).
Description of holotype. ( Figs. 1 View FIGURE 1 , 2 View FIGURE 2 ) Male; body slender; head longer than wide; snout protruding in lateral view and acuminated in dorsal view; nostrils rounded; prominent medial process between the nostrils; canthus rostralis distinct; loreal region concave; eye slightly protuberant; tympanum distinct; discrete subgular vocal sack; tongue round and large; vomerine teeth in two arc shape series between choanae; medium-sized, elliptical choanae.
Arms slender; forearms slightly robust; fingers slender and medium-sized, relative lengths I<IV<II<III ( Fig. 1 View FIGURE 1 E); fingers not webbed; discs elliptical, slightly wider than long; outer metacarpal tubercle large, bifid; inner metacarpal tubercle elliptical, weakly developed; subarticular tubercles evident, rounded, single; subarticular tubercle in the fourth finger more developed than others.
Legs moderately robust; tibia longer than thigh; sum of thigh and tibia size smaller than SVL; toe disks elliptical, faintly wider than long, about the size of the finger disks; toes with relative lengths I<II<V<III<IV ( Fig. 1 View FIGURE 1 D); feet with round metatarsal tubercles (inner more developed than outer); subarticular tubercles evident, rounded and single; numerous tubercles on the posterior skin of the feet and tarsus. Webbing formula I – II 2 + – 3+ III 2 – 3 + IV 3 – 2 V; dorsal skin with a few tubercles; ventral skin rugose; belly and chest granular.
Dorsum light-gray with an evident interorbital dark bar; discreet dark line on the canthus rostralis; dark longitudinal stripe originating at edge of each eye and running towards and nearly reaching the inguinal region; transversal dark bars originating on each inguinal region and converging on the third portion of the dorsum in an inverted V-shaped figure; transversal dark bars along arms and legs until the tips of fingers and toes; gula region with some irregular dark patches; ventral surfaces white, but darker on the undersurfaces of tibia, tarsus, feet and cloacal region. In life, the hidden portion of thigh, calf and inguinal region with some irregular bright yellow colored patches. In preservative, these patches become white.
Measurements of the holotype (mm). SVL 19.34, HEL 7.05, HW 6.94, THL 8.87, TBL 10.01, FL 7.27, HL 5.18, ED 2.53, TD 1.05, IOD 2.25, END 2.42, IND 1.95.
nant frequency of Scinax cosenzai sp. nov.. *One of the calls with one pair of merged notes **Two of the calls with one pair of
merged notes.
7 (3)* 1338.48 (1247.06–1428.63; 183.68 (30.00–372.93; 4027.82 (3877.77–4316.09;
SD=90.79) SD=121.72; n=17) SD=249.72)
10 (2)** 942.75; 1132.19 68.53 (13.75–210.75; 3949.33; 4135.09 SD=54.74; n=16)
11 (3)* 1380.33 (1062.75–1580.25; 88.07 (18.18–403.43; 3984.85 (3941.26–4070.48;
SD=278.09) SD=98.73; n=29) SD=74.16)
12 (3)* 1748.08 (1266.12–2066.75; 106.81 (10.37–443.25; 4568.52 (4563.14–4571.21;
SD=242.26) SD=125.22; n=32) SD=4.66)
13 (1) 1790.50 94.24 (23.37–487.81; 4175.47 SD=134.43; n=12)
14 (1) 1784.69 87.16 (10.00–381.87; 4127.01 SD=109.69; n=13)
Variation. In preservative, as in life, coloration of dorsum varies from clear-gray to dark-gray ( Fig. 2 View FIGURE 2 ). Interorbital and dorsal bars are not evident in some individuals. Webbing formulae varies among specimens I – II 2 – 3 + III (2+ – 2-) – (3+ – 3) IV 3 + – 2 V. Females are larger than males. Range, mean and standard deviation of the measurements of 30 males and seven females are given in Table 2 View TABLE 2 .
Character Male (N=30) Female (N=7)
Natural history notes. Calling activity was more common during the rainy season at night. As observed in others species from the S. perpusillus group ( Oliveira & Navas 2004; Brasileiro et al. 2007a), Scinax cosenzai sp. nov. was more frequently found in clustered bromeliads. We observed a few spawnings, with one or two eggs each, stuck to lateral leaves (as also reported for other species of the group by Alves-Silva & Silva 2009) a few centimeters above the water accumulated by the bromeliad. Tadpoles were also observed in the water of bromeliads and frequently two individuals were observed sharing the same leaf axil. When we were noticed by the tadpoles they usually dived to the bottom of the accumulated water, as also reported for Scinax tupinamba by Silva and Alves- Silva (2009).
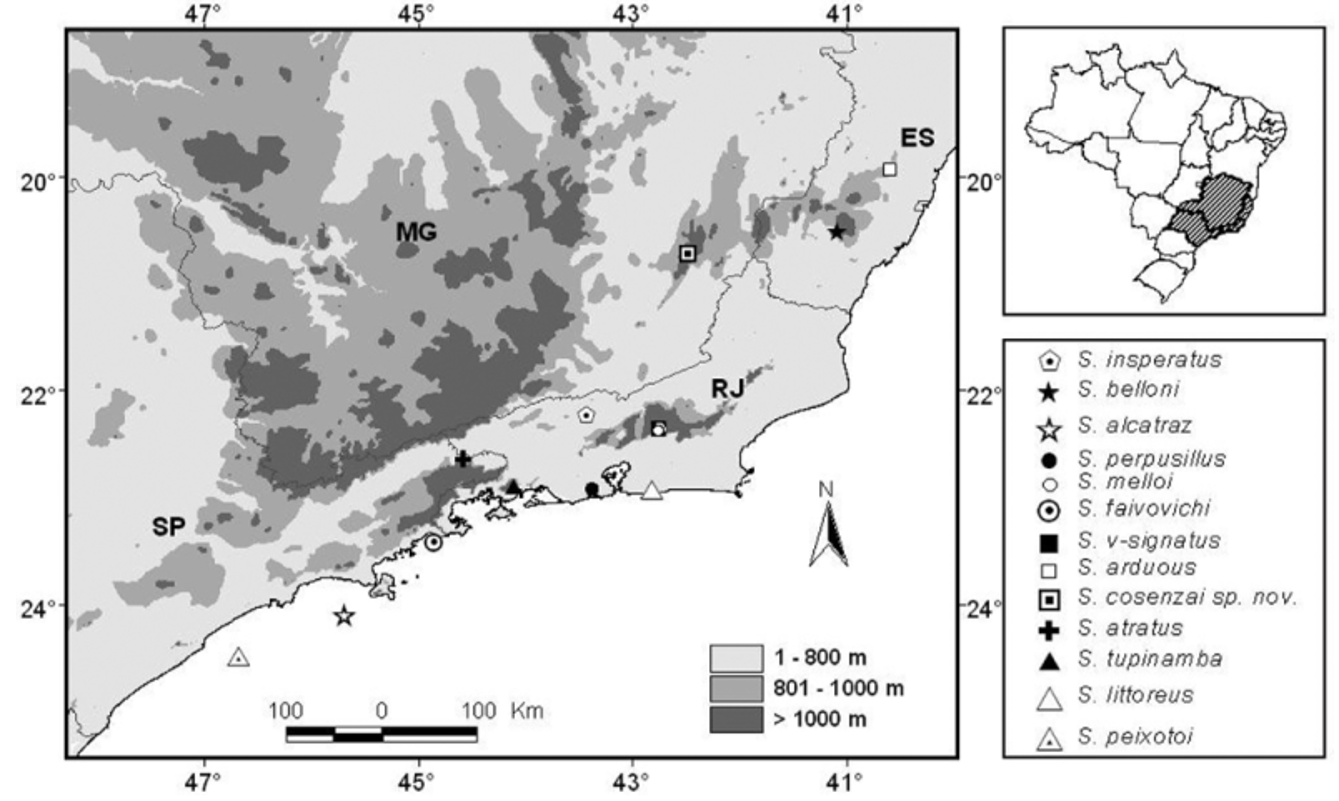
Distribution. The new species seems to be abundant in Alcantarea extensa (Bromeliaceae) , a ground bromeliad commonly found in the rocky outcrops from Parque Estadual da Serra do Brigadero—PESB ( Fig. 5 View FIGURE 5 ), a conservation unit with approximately 13,000 ha from the Atlantic Rain Forest biome managed by Instituto Estadual de Florestas—IEF, in the northern part of the Mantiqueira Mountain Range Complex, State of Minas Gerais ( Cruz et al. 2007; Caramaschi et al. 2008). We collected specimens from two rocky outcrops: the type locality, located in the PESB, and another outcrop, located in the surroundings of the PESB ( Fig. 5 View FIGURE 5 ). As Alcantarea extensa is commonly found in others rocky outcrops from the Zona da Mata region in the State of Minas Gerais ( Versieux & Wendt 2006), we believe that the new species may also occur in some of these other rocky outcrops and may not be in danger of extinction.
Etymology. The specific name honors the biologist Braz Antônio Pereira Cosenza for his contributions to the studies of flora and fauna of Serra do Brigadeiro.
Additional remarks. As observed by Silva and Alves-Silva (2008), species from the Scinax perpusillus group from outside the geographic limits sampled by Peixoto (1987), usually referred to as Scinax perpusillus , Scinax aff. perpusillus or Scinax gr. perpusillus (see Heyer et al. 1990; Pombal & Gordo 2004; Oliveira & Navas 2004; Carvalho-e-Silva et al. 2008; Feio et al. 2008; Bertoluci et al. 2009; Lacerda et al. 2009), may represent species not yet recognized and described. In spite of such problematic taxonomy, the advertisement call of only five species has been somewhat described ( Scinax arduous , S. insperatus , S. peixotoi , S. perpusillus and S. v-signatus ). However, the vocalization of these species has been described in two different manners: Heyer et al. 1990, Pombal and Bastos (2003) and Brasileiro et al. 2007a described each call of a call series while Alves-Silva and Silva (2009) and Silva and Alves-Silva (2008) referred to the entire call series as a single call, referring to notes and pulses what previous studies considered respectively as single calls and notes. We agree with Silva and Alves-Silva (2011) in believing that these previous studies did not consider the social interactions during vocalizations and that the described vocalization probably represents only a part of the entire call (herein referred to as call series). We did not compare the vocalization of S. cosenzai sp. nov. to those described for S. insperatus , S. perpusillus and S. v-signatus presented by Alves-Silva and Silva (2009) and Silva and Alves-Silva (2011) because these studies described only duets, or calls emitted when a female approached the male, while the recorded vocalizations from S. cosenzai sp. nov. were emitted by isolated males. We agree with Alves-Silva and Silva (2009) and Silva and Alves-Silva (2011) in considering further investigation necessary in order to establish the universality of call structure for the species from the S. perpusillus group.
As observed in Figure 6 View FIGURE 6 , the type localities of Scinax alcatraz , S. faivovichi and S. peixotoi are associated to coastal islands from the state of São Paulo (until now, considered endemic to these islands). Scinax littoreous , S.
perpusillus and S. tupinamba have type localities associated to lowland coastal areas in the state of Rio de Janeiro. Scinax arduous and S. belloni have distribution associated to mountain regions in the state of Espírito Santo, while S. insperatus , S. melloi and S. v-signatus are found in the state of Rio de Janeiro and S. atratus in the state of São Paulo. Scinax cosenzai sp. nov. is the first species belonging to the S. perpusillus group described in the State of Minas Gerais.
Bertoluci et al. 2009 provided the record of specimens referred to as Scinax sp. 3 (gr. perpusillus ) in the Estação Ambiental de Peti, in Minas Gerais state, but they only cited the field number, not mentioning the deposition of voucher specimens in any public collection. If this identification is confirmed, Bertoluci et al. 2009 represents the first record of a species belonging to the S. perpusillus group in the Espinhaço mountain range. The first record of a species belonging to the Scinax perpusillus group in the state of Minas Gerais was made by Feio et al. 2008 reporting the existence of a population of Scinax gr. perpusillus in the Parque Estadual da Serra do Brigadeiro. Lacerda et al. 2009 also referred to this population as species that has probably not yet been described. The present study describes this new species bringing to 13 the number of species included in the S. perpusillus group.
TABLE 1. Number of notes per call (number of recorded calls in parenthesis), call duration, interval between notes and domi-
| Notes/call (Number of calls) | Call duration (ms) | Interval between notes (ms) | Dominant frequency (Hz) |
|---|---|---|---|
| 2 (7) 3 (9) | 186.77 (177.62–220.12; SD=14.95) 583.33 (512.75–747.81; SD=71.81) | 100.232 (87.00–110.44; SD=8.14; n=7) 230.21 (815.00–516.50; SD=145.73; n=18) | 4073.41 (3872.33–4555.06; SD=231.97) 4073.41 (3872.33–4555.06; SD=231.97) |
| 4 (6) | 882.364 (812.25–1018.5; SD=80.61) | 244.73 (74.00–400.12; SD=120.98; n=18) | 3886.89 (3375.91–4127.01; SD=273.22) |
| 5 (3) 6 (4) | 967.58 (931.43–991.00; SD=31.75) 1148.07 (892.81–1395.75; SD=249.63) | 191.31 (77.62–397.00; SD=122.25; n=12) 185.65 (35.00–439.00; SD=135.64; n=20) | 3964.79 (3877.77–4127.01; SD=140.61) 4002.91 (3753.61–4250.25; SD=202.76) |
TABLE 2. Range, mean and standard deviation (SD) of some measurements (mm) of Scinax cosenzai sp. nov. males (N = 30) and females (N = 7): snout-vent length (SVL); head length (HEL); head width (HW); thigh length (THL); tibia length (TBL); foot length (FL); hand length (HL); eye diameter (ED); tympanum diameter (TD); interorbital distance (IOD); eye-nostril distance (END); and internostril distance (IND).
| Mean | SD | Range | Mean | SD | Range | |
|---|---|---|---|---|---|---|
| SVL | 18.99 | 0.92 | 17.29–20.97 | 23.37 | 0.44 | 22.65–24.02 |
| HEL | 7.20 | 0.34 | 6.64–7.85 | 8.54 | 0.36 | 8.14–9.02 |
| HW | 6.67 | 0.27 | 6.19–7.20 | 7.96 | 0.25 | 7.55–8.20 |
| THL | 8.67 | 0.59 | 7.33–9.73 | 10.52 | 0.52 | 9.73–11.15 |
| TBL | 9.65 | 0.54 | 8.74–11.29 | 11.76 | 0.90 | 10.54–13.15 |
| FL | 7.34 | 0.39 | 6.69–8.33 | 9.10 | 0.73 | 8.23–10.35 |
| HL | 5.10 | 0.27 | 4.62–5.55 | 6.11 | 0.61 | 5.47–7.35 |
| ED | 2.12 | 0.23 | 1.62–2.60 | 2.33 | 0.20 | 2.00–2.60 |
| TD | 0.99 | 0.08 | 0.82–1.20 | 1.15 | 0.10 | 1.04–1.32 |
| IOD | 2.53 | 0.18 | 2.24–2.96 | 2.74 | 0.24 | 2.25–3.05 |
| END | 2.39 | 0.13 | 2.12–2.80 | 2.59 | 0.25 | 2.26–3.04 |
| IND | 1.78 | 0.10 | 1.60–2.00 | 2.06 | 0.15 | 1.85–2.35 |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.