Poeppigia procera (Poepp. ex Spreng.) C. Presl. (1830: 16)
|
publication ID |
https://doi.org/ 10.11646/phytotaxa.513.3.1 |
|
persistent identifier |
https://treatment.plazi.org/id/0399878C-984C-FF86-D6C9-FCF0FB4EE811 |
|
treatment provided by |
Felipe |
|
scientific name |
Poeppigia procera (Poepp. ex Spreng.) C. Presl. (1830: 16) |
| status |
|
Poeppigia procera (Poepp. ex Spreng.) C. Presl. (1830: 16) View in CoL . ( Figures 1 View FIGURE 1 and 2 View FIGURE 2 ).
≡ Basionym: Caesalpinia procera Poepp. ex Spreng. (1825: 343) . ≡ Poeppigia procera (Poepp. ex Spreng.) C. Presl. var. procera . Benth in. Mart. (1870: 54). Type:— CUBA. July-1822 –1824, Poeppig. s.n. HAL barcode HAL120570 (Lectotype designated here: HAL!; possible isolectotypes in BR!; JE!; MO!; P!).
= Poeppigia excelsa A. Rich. (1846: 484) . ≡ Ramirezia cubensis A. Rich. (1846: plate XXXIX). nom. nud. Type:— CUBA. s.d. Ramon de La Sagra s.n. P barcode P02771615 (Lectotype designated here: P!) .
= Poeppigia ferruginea Tul. (1844: 120) . ≡ Poeppigia brasiliana Tul. nom. ined. Type:— BRAZIL, Minas Gerais. 1838-1840. Claussen 967 P barcode P02771622 (Holotype: P!; isotypes in F!; P!; NY!; RB!) .
Trees (3–) 10–30 (–45) m tall, very rarely flowering as 3–5m shrubs; up to 1.6m in diameter; bark brown, grayish or whitish; flowering branches usually long, with (7–) 12–34 (–50) cm long. Canaliculate leaf rachis, (7–) 10–16 (–19) cm long, pubescence generally concentrated on the upper face, rarely glabrous, trichomes generally curved; petioles (0.3–) 0.5–1 (–1.5) cm long; petiolules about 1–2 mm long; (28–) 38–68 (–76) leaflets, (0.6–) 1–1.7 (–2.3) × (0.2–) 0.3–0.5 (–0.7) cm; stipules lanceolate to elliptical, caducous, two by axillary bud; axillary bud lanceolate to elliptical. Inflorescences thyrsoid, distichous, emitted from the apex of the branch and from the axils of many leaves, usually in half or more than half of the leaves of a branch, with thyrsoids gradually smaller towards the apex, forming lax synflorescences of triangular shape, with (5.5–) 8–18 (–24) × (4–) 7.5–14 (–21) cm, the basal most and largest inflorescences with (2.5–) 3–10 (–12.5) cm long; bracts caducous, without bracteoles. Flowers with connate sepals, with 3–5 × 1–2 mm, lobes about 1–2 mm long; petals with 6–9 (–10) × 2–4 mm; stamens with anthers measuring 1–1.2 × 0.5–0.7 mm, filaments with 4–8 × 0.5 mm; ovary with 3–6 × 1 mm, glabrescent or pubescent, stipe with 2–3 mm long. Samaroid fruit, all or most of the fruits elongated, the largest with (4.2–) 6–12.5 (–14.5) × 0.9–1.5 × 0.2–0.4 cm; stipe generally glabrous, less commonly pubescent, pubescence uniformly distributed; calyx generally caducous, less commonly persistent in the fruit. (1–) 2–4 (–6) seeds, with 6–8 × 4–6 × 1–2 mm. Seedlings with first pair of eophils lanceolate.
Diagnosis: — Poeppigia procera are median to large trees, with generally longer flowering branches, while P. densiflora are usually shrubs or rarely small trees, with generally shorter flowering branches; P. procera has longer leaf rachis with generally 38–68 leaflets generally longer, while P. densiflora has shorter leaf rachis with generally 16–26 leaflets generally shorter; P. procera has stipules and bracts almost always caducous, while P. densiflora has stipules almost always persistent, at least in the upper part of the branch, those stipules commonly leafy, smaller stipules commonly along the rest of the branch and a variable number of persistent bracts along the primary and secondary axes of the inflorescences; P. procera has inflorescences emitted from the apex of the branch and also from the axils of many leaves along the branch, with thyrsoids gradually smaller towards the apex, forming lax triangular synflorescences with more flowers, the more basal inflorescences larger, while P. densiflora has inflorescences only terminal or terminal and axillary, but the axillary ones are almost always few and emitted only from the axils of the most distal leaves of the apex of the branch, forming small and congested synflorescences with less flowers and generally umbelliform, or when, in rare cases, inflorescences are emitted from leaves of the entire branch, basal and apical inflorescences are small and of similar size; P. procera has all or most of the fruits of the same branch elongated and larger with, generally, 2–4 seeds, while P. densiflora has fruits elliptical and small with generally 1 seed and less commonly 2.
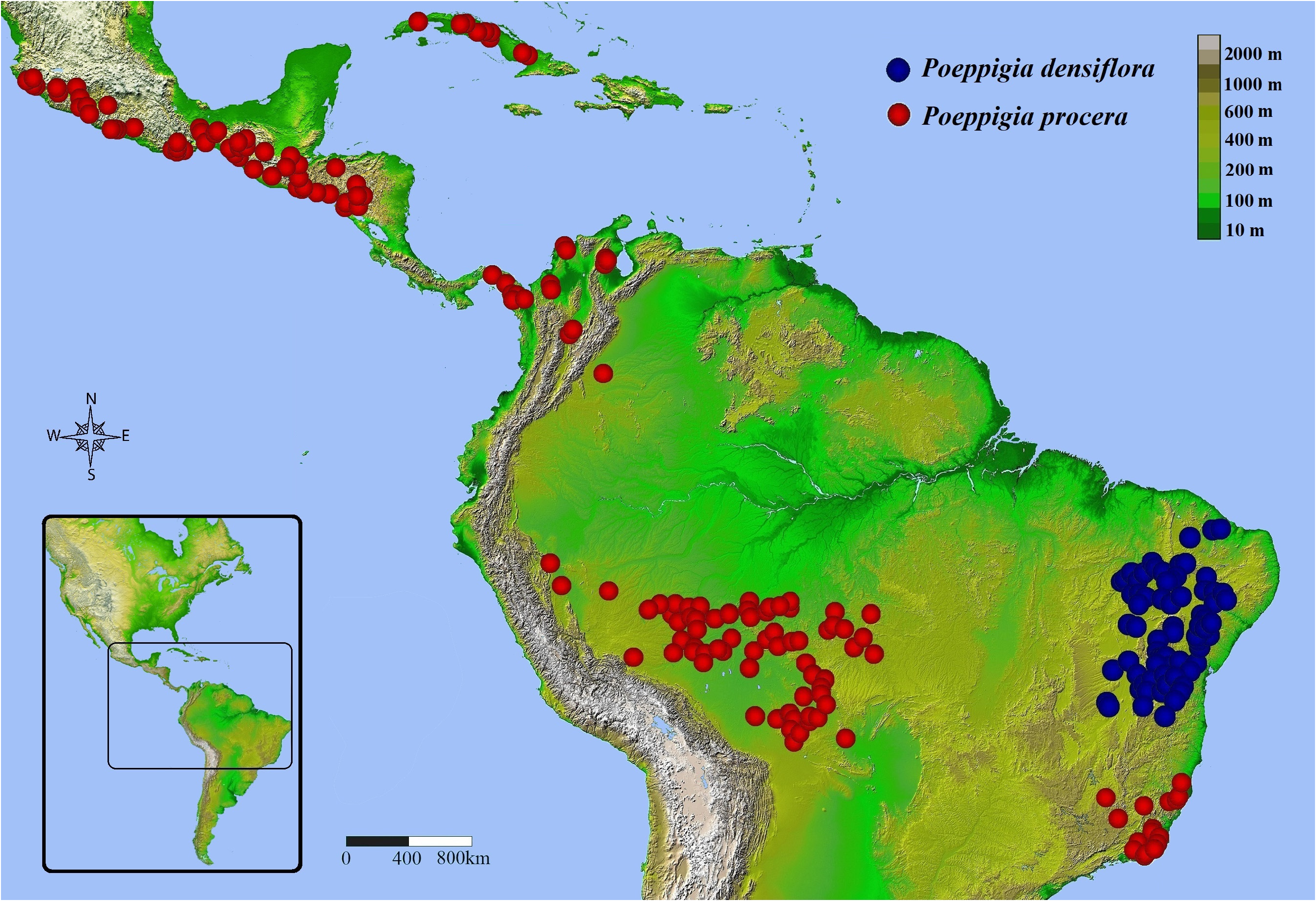
Distribution and Habitat: — Poeppigia procera occurs in primary and secondary forests in the following countries: Cuba, Mexico (states: Chiapas, Oaxaca, Guerrero, Michoacán, Colima and Jalisco), Guatemala, El Salvador, Honduras, Nicaragua, Panama, Colombia (departments: Chocó, Córdoba, Tolima, Meta and Magdalena), Venezuela (department of Zulia), Peru (departments: San Martin, Madre de Dios and Ucayali), Bolivia (departments: Santa Cruz, Beni and Pando) and Brazil (states: Acre, Rondônia, Mato Grosso, Rio de Janeiro, Espírito Santo and Minas Gerais) ( Fig. 3 View FIGURE 3 ). The occurrence of the species in Belize and Costa Rica has not been confirmed here, but we consider it possible (See biogeographical notes). The species is found in the following phytophysiognomies: open ombrophilous forest, seasonal evergreen forest, seasonal semi-deciduous forest and savannah, usually in upland forests, very rarely in Igapó forests, developing in clayey or clayey-sandy soils. In Brazil, P. procera is found in the Amazon and Atlantic Forest biomes.
Conservation: — A great EOO of 12,809,221 km 2 was estimated for P. procera . The species occurs in 12 countries and in several different phytophysiognomies, including some protected areas as the Reserva Ríos Blanco y Negro and Parque Nacional Noel Kempff Mercado in Bolivia; the Uru-Eu-Wau-Wau indigenous land in Brazil; and the Parque Nacional del Manu in Peru. Thus, a preliminary least concern category can be suggested to the species. However, the existence of several small and isolated populations along the distribution of this species (See biogeographical notes), notably the small populations of Cuba and of the Brazilian Mata Atlântica may indicate the reduction or absence of gene flow between populations and the existence of local extinction threats, mainly in areas composed of small fragmented forests as in Southeast Brazil. We should also note that P. procera , even though widely distributed and present in officially protected areas, is not considered common in any of the environments where there are records of its occurrence (present work) and that high deforestation rates have been observed recently even in protected areas, notably in Brazilian Amazon ( Potapov et al. 2017; Montibeller et al. 2020; INPE, 2020). Thus, population genetics studies are necessary to better understand the diversity within this species and the possibility that some of its isolated populations are facing some kind of threat or will face it in the near future.
Phenology: —With its widespread occurrence throughout the Neotropics, P. procera may have its populations clearly separated into two main groups with respect to phenology: one to the north and one to the south of the Equator, with flowering concentrated in the summer of each hemisphere. The populations of Mexico, Central America and Venezuela flourish in May–July (–August) and bear fruits in (July–) September–March. The populations of Peru, Bolivia and Brazil flourish in (August–) November–January (–April) and bear fruits in (January–) March–September. The main exceptions are the populations of Colombia, a possible transition between the phenologies of the northern and southern hemispheres, with examples of flowering in January, May, July, October and November.
Etymology: —Presl possibly referred to the height of the tree when proposing the specific epithet “procera”, from the Latin “procerus”, which means tall. However, compared to the majority of tree species of the subfamily Dialioideae in the Neotropics that commonly exceed 30 meters in height and can reach up to 40–50 m, P. procera is only a medium sized tree, generally reaching an average of 20–25 m with very rare specimens reaching 35– 45 m.
Uses: —The wood is considered quite hard, as well as in many other genera of Dialioideae such as Apuleia , Martiodendron , Dicorynia and Dialium , being used in the manufacture of various objects and also for construction and as fuel. It is also reported as medicinal in Bolivia, with the leaves used in the manufacture of teas to treat colds. In Central America, it is considered sacred by some communities (information obtained from specimen labels).
Vernacular Names: —Pintadinho ( Brazil: Northern region), Coco d’óleo ( Brazil: Southeastern region); Isnepa, Cari-cari, Cari-cari blanco, Pajarillo, Toco, Tasaa ( Bolivia); Tepemisque, Tepemiste, Plumillo ( Guatemala); Harrino ( Panama); Memble, Memble-amarillo, Memblón de bajío, Frijolillo ( El Salvador); Cedro-pashaco ( Peru); Guayacán, Llovizno, Hoja menuda ( Colombia); Abey-hembra, Abey-heurbra, Palo-tranquilo, Tengue ( Cuba), Corazón bonito, Guaje, Quebracho blanco, Quebracho and Quiebra-hacho ( Mexico). The latter two names are also reported for the related genus, Dialium , which also occurs in Mexico and Central America and for several other exceptionally hardwood tree genera.
Taxonomic Notes: —In the description of P. ferruginea, Tulasne (1844) specifies some of the distinctive floral characteristics of the new species such as the median narrowing of the calyx tube being rare, the most unequal calyx and less pubescence of the ovaries. Tulasne describes his new species as having broader and less acute fruits. The seeds are described as less flattened and less quadrilateral. When checking all these characteristics, we see that there is a great variation in all of them throughout Neotropical populations, without even a differentiated frequency between the typical plants of Cuba and the continental plants, so that the characters punctuated by Tulasne do not seem strong enough to support any taxon in this genus. Tulasne also mentions some variations, also in degree and in a very subjective way, in shapes, colors and angles of the hilum, radicle, raphe, embryo and cotyledons of the seeds as differentiating the two species. However, among the many Cuban specimens analyzed personally by us, we were unable to obtain any whose fruits had seeds suitable for analysis. However, it also seems to us that the idea of separating two taxa, even in an infra-specific rank, by characters such as “mediocre raphe”, “green cotyledon”, “straight embryo”, among others, lacks robustness, and these characteristics are commonly found both in P. procera and P. densiflora . So, with the data obtained so far, we can consider P. ferruginea as a synonym of P. procera . P. excelsa ( Richard 1846) , as already mentioned, does not even have comments on any specific character that differentiates it from P. procera ; in the description and illustration no character seems to denote any difference between the two taxa or could, when scrutinized by us, show some morphological pattern differentiating some population, so that we also consider P. excelsa as a synonym for P. procera .
When we compare the populations of P. procera from the Atlantic Forest of southeastern Brazil (Rio de Janeiro, east of Minas Gerais and Espírito Santo) with those populations from the Amazon, Central America and Mexico, largely isolated from each other (see biogeographic notes), a slight variation can be observed in relation to the fruits. In the Amazon and Central American populations, it is quite common for the narrow wing that forms from the suture on one side of the fruit to be conspicuous from the junction of the fruit to the stipe. On the other hand, in southeastern Brazil populations, the wing usually only begins to become conspicuous shortly after the fruit base ( Fig. 4 View FIGURE 4 ). In P. densiflora , you can find both types of wing with similar frequencies. The pubescence of carpels, in turn, is a stable character in P. densiflora , with practically all specimens presenting the carpel mostly glabrous, with pubescence in the stipe and, more slightly, along the carpel suture. In P. procera , although the most common pubescence pattern is the same as in P. densiflora , specimens with pubescence in the stipe and along almost the entire carpel are also common, notably in southeastern Brazil (example: Spada , 105 [RB]), a character not found in P. densiflora . As for the campanulate calyx, P. densiflora has it almost always persistent in the fruit, as P. procera may also have persistent calyx in some populations, notably in southeastern Brazil and Central America, however it is more common in the species that the calyx is senescent in fruit. As, in all these cases, there are many exceptions to the rules, it is not possible to infer the existence of any differentiated taxon in southeastern Brazil, even in an infra-specific rank. However, it is possible to speculate that this is an evidence of the beginning of a speciation process between the isolated populations of these regions.
The height of P. procera individuals varies considerably in relation to their distribution, while in South and Central America individuals up to 30m in height are common, with cases up to 45m in Peru (Foster, R. 12608 [ US]), fertile specimens of less than 10m are very rare and flowering shrubs of less than 5m are practically nonexistent. However, in Mexican savannas, such smaller specimens become more common, with the country containing about half of the 35 specimens with 5-10m in height and almost all 10 fertile specimens under 5m observed in this work (out of a total of 250). It should be noted that in this country, several specimens of 20-30m and even a specimen of 40m are also found (Méndez, A. 7781 [NY]). Despite the variation in size, there are no variations in reproductive characteristics associated with this, and the species remain distinct from P. densiflora . The latter has a much more stable size throughout its distribution, with only three specimens observed (of a total of 160) exceeding 10m (12-15m: Santos, W. 12 [RB]; Queiroz, L. 3726 [HUEFS]; Lewis, G. 1082 [NY]), which can be related to its more restricted distribution in more similar habitat, while P. procera has a widest distribution throughout more diverse habitat.
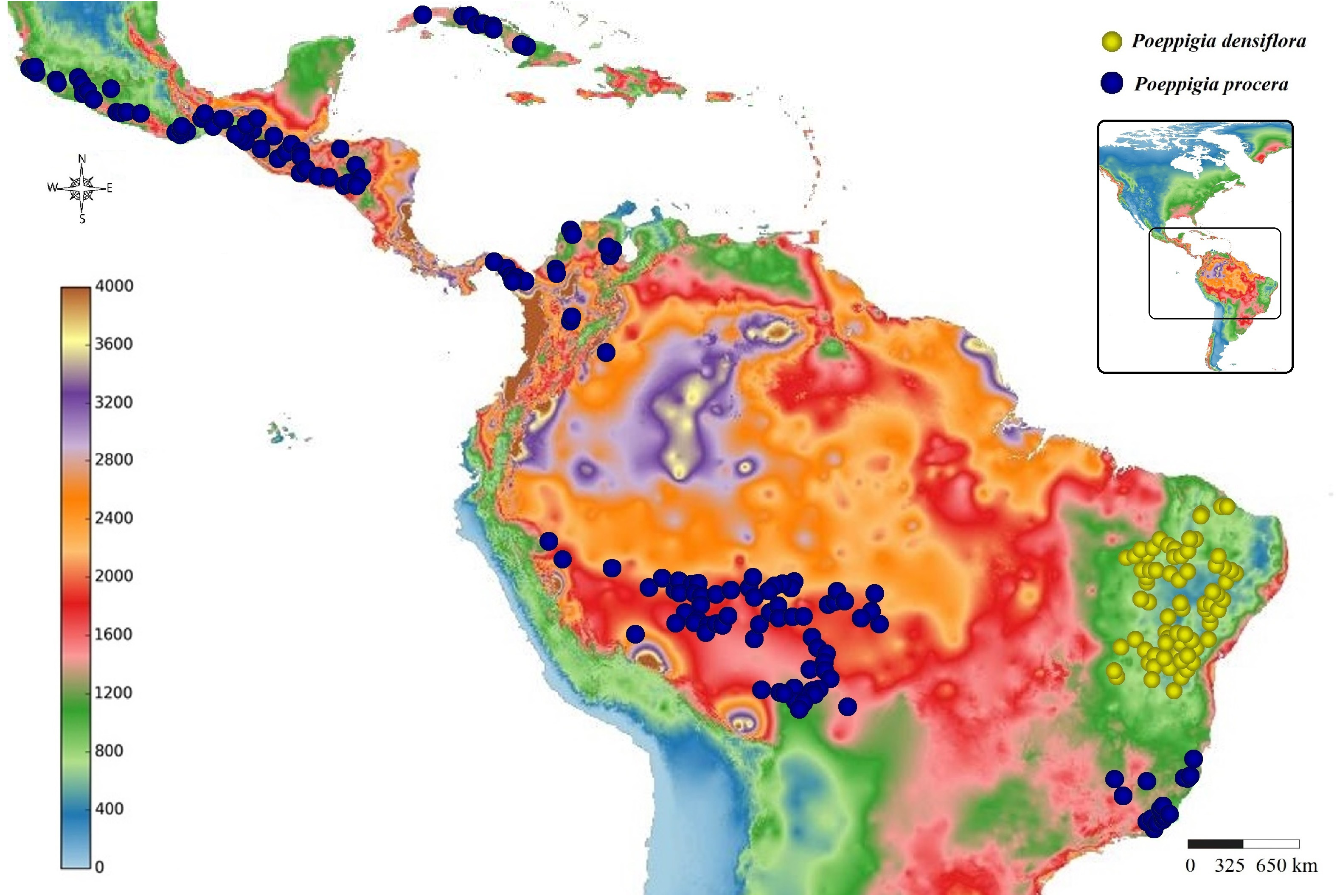
Biogeographical Notes: —The geographical distribution of Poeppigia is Neotropical, with P. densiflora restricted to the Brazilian Caatinga and P. procera widely distributed from Mexico and Cuba to Rio de Janeiro. The distribution of this second species stands out, not only when compared to the other species of the genus, but also among all Neotropical Dialioideae . While all species of the genus Dicorynia , Martiodendron , Apuleia and almost all american species of Dialium have mostly continuous distributions and a single species of Dialium : Dialium guianense (Aubl.) Sandwith (1939: 184) , is distributed in two main disjunct areas, P. procera is the only species with populations concentrated in numerous groups strongly isolated for distances ranging from 800 to 1500km. It has four major disjunctions (Caribbean Sea; Amazon Basin; Cerrado; West Panama –East Nicaragua) and five isolated distribution areas ( Cuba; from Mexico to western Nicaragua; from eastern Panama to Colombia; from eastern Peru to Mato Grosso; part of Southeast Brazil). When we compare this distribution to a topographic map of the Neotropics ( Fig. 5 View FIGURE 5 ), we see that, in addition to the oceanic barrier separating the Cuban plants from the others, we have the species occurring mostly in areas with elevations above 100 meters, absent from the low elevations of the central and eastern Amazon basin. When we associate this distribution with average rainfall ( Fig. 6 View FIGURE 6 ), we see more possible correlations, with the species occurring almost always in areas with average annual precipitation less than 2200 mm, being the most humid areas of the Amazon basin and Costa Rica a possible barrier to the occurrence of the species. We can also think about the possibility of a continuous distribution in drier periods of the past in which these less humid areas could be connected ( Bush & Oliveira 2006, Haffer 2008), and the species not being able to persist later in those regions with moister climate. As for the disjunction of P. procera in the Brazilian Cerrado region, although a relatively smaller average annual precipitation arc and slightly higher elevations between the states of Goiás and Minas Gerais may contribute to the isolation between the Amazonian and Southeastern Brazil populations, these variables alone do not seem to explain the observed distribution since, in western Mexico, northern Colombia, and eastern Minas Gerais, P. procera appears to withstand average rainfall levels similar to the areas of non-occurrence in Cerrado of eastern Mato Grosso, Goiás and Tocantins states. So, it is possible that there is a seasonal relationship with the lowest precipitation in the driest months of the year in these regions of Cerrado. In addition, as we could not find stable differences between the populations of Southeast Brazil and the Amazon, we can consider the hypothesis of relatively recent connections between the forests of the Amazon and the Atlantic Forest ( Sobral-Souza & Lima-Ribeiro 2017). As in Poeppigia , the related genus Dialium , with similar distribution (disjunction in the Cerrado, with populations in the Amazon and in the Atlantic Forest from the southeast to the Northeast of Brazil), had different morphotypical patterns observed in the plants on each side of the disjunction, although not enough to distinguish different taxa ( Falcão et al. 2016). Thus, it seems possible that, like the related Dialioideae genera Dialium , Apuleia and Martiodendron , the genus Poeppigia has part of its evolutionary history associated with the expansion and contraction of the seasonally dry forests of the Neotropics and with the conservation of ecological niches (Banda et al. 2016, Falcão et al. 2016, 2020b, in prep).
Although there are small pockets of dry forests in Costa Rica, notably in its northwest region, in the province of Guanacaste ( Fig. 6 View FIGURE 6 ), similar to the environments in which P. procera occurs in the other Central American countries, we were unable to find any specimens collected in this country, despite having accessed some of the largest and most complete collections on Central American flora, such as those of the MO, P, US and NY herbaria. It should be noted that, although a large number of specimens exist for almost all countries on the continent, only three specimens for Nicaragua were observed, all from regions close to the border with Honduras and none close to Costa Rica. Likewise, all specimens observed from Panama are concentrated in the east of the country, far from Costa Rica. In addition, the species is not mentioned for the country in the flora of Costa Rica ( Standley 1937) and Guatemala ( Standley & Steyermark 1946). Only two related citations were found: the first in the book Árboles de Centroamérica ( Barrance 2003) where the area of potential occurrence for the genus reaches northwestern Costa Rica, but the occurrence of the taxon in the country or the existence of collected specimens is not cited. Even in this potential distribution, the aforementioned disjunction for most of Costa Rica, eastern Nicaragua and western Panama is also indicated; Second, in the checklist of tree species in El Salvador ( Bridgewater et al. 2006) the occurrence in Costa Rica is mentioned, but again, no specimen is indicated. Having this in mind, due to the presence of some dry forest environments in the northwest of the country, we consider the occurrence of P. procera in Costa Rica possible in such regions, perhaps with great rarity.
No specimen of Poeppigia from Belize was observed in this work and the only citations referring to the country are in the book Árboles de Centroamérica ( Barrance 2003) where, again, the map for the genus indicates Belize as a potential occurrence area but does not mention the existence of collected specimens. In the List of the Dicotyledoneae of Belize ( Dwyer & Spellman 1981), a single specimen is cited (Leino 355 [MO]), and it is actually a Lysiloma latisiliquum (L.) Benth (1875: 534), a Mimosoid legume. The extensive checklist for the Chiquibul forest ( Bridgewater et al. 2006), a large and representative region in the south of the country, the flora of Guatemala ( Standley & Steyermark 1946) and the tree flora of El Salvador (Berendshon et al. 2009) does not mention the presence of Poeppigia for Belize. However, observing the average precipitation and elevation of this region and the distribution of the specimens observed in this work ( Figures 5 View FIGURE 5 and 6 View FIGURE 6 ), we can infer that Belize is, in relation to these parameters, potentially habitable for the genus and that some specimens in Guatemala occur close to the border with the country. Thus, although without confirmation, we consider it possible that Poeppigia also occurs in the south of Belize, albeit rarely.
Representative specimens examined (50 of 250 specimens): — BOLIVIA. Pando: Triunfo , 11°10’S 69°7’W, mixed seasonal forest on undulating land. Tree, 26m × 29cm, 28-VII-1988. Pennington, R. T GoogleMaps . 50 ( K); Manupuri, municipio de Puerto Rico, comunidade Irak , bosque alto inundado, 11°21’57”S 67°16’49”W. 11-V-2014. Carrion Cuellar, A. M GoogleMaps . 1011 (NY); Beni: Alto Ivo, Chacobo, Vaca Díez , 11°45’S 66°02’W, tropical wet forest. Tree, 12m × 28cm, 7-XII-1993. Boom, B. M GoogleMaps . 4097 ( NY); Vaca Díez, Reserva Ecológica El Tigre, km 42 de la carretera Riberalta- Guayaramerín, bosque secundário. Árbol , 12m × 21cm, 4-VI-98. Peña , M . 826 (U); Santa Cruz: Reserva ecológica El Refugio: a 1400m al SW del campamento Toledo. Arbol , 16m. 12-V-1995. Guillén, R . 3605 ( NY); Nuflo de Chavez: Perseverancia. Vecindad del Rio Negro, tributario del Rio Baures , 14°38’S 62°37’W, Árbol, 20m, 18-V-1991. Mostacedo, B GoogleMaps . 133 (F); BRAZIL. Acre: Manoel Urbano, rio Purus, margem direita, seringal Santa Cruz , 9°04’46”S 69°36’05”W, floresta de bambu com subosque densamente ocupado por colmos. Árvore, 30m, 26-XI-1996. Silveira , M GoogleMaps . 1594 ( NY); Xapuri, rio Acre , 10°45’S 68°20’W, moist forest. Tree, 30m × 30cm, 6-XI-1991. Daly, D. C GoogleMaps . 7165 ( NY); Bujari, riozinho do Andirá. Árvore , 7m, 28-V-2009. Obermuller, F . 455 ( NY); Rio Branco , km 8 R . 364, 18- VI-1987. João, A. S . 71 (INPA); Espírito Santo: Reserva Florestal de Linhares, mata de tabuleiro. Árvore. 21m, 14- XII-1972. Spada , J . 133/72 ( RB); estrada 154, talhão 507, mata de tabuleiro. Árvore , +- 25m × 12cm, 29-XI-1972. Spada , J . 105 ( RB); Colatina, Alto Moacir, Pedra do Cruzeiro , 19°18’39”S 40°31’75”W, borda da mata com vegetação rupestre. Árvore, 16m × 36cm, 22-II-2006. Magnago, L. F. S. 729 ( RB); Mato Grosso: Jaturaha river, Machado river region. Tree 65ft, XII-1931. Krukoff. B. A. 1591 ( F); Aripuanã : BR-174, estrada para o aeroporto, mata de terra firme. Árvore , 8m, 1-VI-1979. Silva , M. G. 4741 ( NY); margem direita rio Juruena , fazenda Ariel , floresta mista com babaçu. Árvore , 30m × 1.60m, 23-VI-1977. Rosa , N. A. 2168 ( RB); Comodoro, mata seca e floresta ombrófila, 13°59’41”S 60°20’37”W. 24-IV-2012. Antunes, E. C. 280 A ( RB); GoogleMaps Minas Gerais: Estação experimental de Água Limpa. Árvore com 20m, 10-X-1968. Gomes , V. 2793 ( UB); Caratinga-Jaó. Árvore, 20-VI-1984. Lopes, M. A. 437 ( BHCB); Paraopeba, em frente ao vivieiro de mudas de EFLEX. Árvore, ca. 15m, 22-III-1999. Valente, G. E. 394 ( VIC); Rio de Janeiro: Quisamã, fazenda São Miguel, ponto 276, 22°04.27’S 41°31.35’W, mata em área de canavial. Árvore , 12m × 60cm, 14-II-2007. Lima, H. C. 6509 ( RB); Rio de Janeiro: arboreto JBRJ, em frente ao prédio de sistemática. Árvore, 6m, 28-I-2005. Ribeiro, R. B. 368 ( RB); arboreto JBRJ, canteiro 34 A. Árvore, 9m, 19-VIII-2015. Silva, F. R. s.n. RB 631116; GoogleMaps Rondônia: near Mutumparaná, Madeira-Mamoré railroad, km 167-169, disturbed forest on terra firme. Tree , 8m × 5cm, 6-VII-1968. Prance , G. T. 5616 ( F); Porto Velho : 15.5 km E of Campo Novo on road to Ariquemes , 10°35’S 63°30’W, forest on slopes. Tree , 14m × 15cm, 25-IV-1987. Nee , M. 35030 ( K); Parna Mapinguari. Árvore com 12m, 13-XII-2013. Silveira , A. 521 ( RON); margem direita do rio Madeira , 5km W do porto da balsa para vila São Lourenço , parcela T5 P1 , 9°35’25”S 65°2’51”W, floresta ombrófila aberta. Árvore, 20m × 30cm, 22-XI-2013. Simon, M. F. 2027 ( CEN); GoogleMaps COLOMBIA. Chocó: Riosucio, Parque Nacional Natural Los Catios , alrededores del departamento de Tilupo. Árbol, 12m, 1-IV-1976. Forero, E. 1700 ( U); Córdoba: Montería, km24 vía a Planeta rica. Arbol 15m, 24-X-1987. Leguízamo , I. 490 ( MO); Chinú , Aguas Vivas , 9°03’00.0870”N 75°29’59.7595”W. Árbol, 7m, 10-III-2015. Quintero, D. 193 ( COL); GoogleMaps Meta: Villavicencio, prédios UNILLANOS. Árbol , 8m, 25-I-1994. Rincón, R. 224 ( COL); Tolima: Mariquita 2-IV-1976. Mahecha 6 ( UDBC); CUBA. Pinar del Río: 1-VII-1905. Donovan, O. 5289 ( NY); Santiago de Cuba: Sevilla, near Santiago. 31-VIII-1906. Taylor, N. 130 ( NY); Villa Clara: Santa Clara in valleys at higher altitude Trinidad mts, near San José. 40’tree, V-1941. Howard , R. A. 5162 ( NY); Trinidad Mountains , San Blas, Buenos Aires, open field at Naranjal. 20’tree. VIII-1941. Howard, R. A. 6429 ( NY); EL SALVADOR. Morazán: Jocoro. 1924. Calderón , S. 2139 ( NY); San Vicente: National Highway, vcty. Kil. 72. Tree , 75ft. 30-VII-1958. Allen, P. H. 6884 ( NY); GUATEMALA. Escuintla: frecuente en potreros e a la orilla de la carretera cerca de Azuchio. Árbol, 8-15m, 20-V-1963. Standley, P. C. 12485 ( F); HONDURAS. El Paraíso: c. 5 mi S of Yuscarán, xeric parkland-scrub. Tree , c. 10m, not common, 5-VII-1962. Grady, L. 12013 ( U); MEXICO. Colima: 1 km en línea recta al SE de Chandiablo, Manzanillo. Árbol, 10m. 30-VIII-2002. Manríquez, G. I. 5898 ( NY); Chiapas: Chiapilla, a 12 km SE de Acala por camino a Venustiano Carranza, bosque tropical caducifolio. Arbusto , 3m, 23-VII-1984. Hernández, H. M. 558 ( MO); Michiocán/ Guerrero: Chumbítaro, Malpaso. 7-XI-1935. Hinton, D. B. 8042 ( P); Oaxaca: Pochutla, San Miguel del Puerto , 1km al S de Rancho San Agustín, 15°58’52”N 96°5’47”W, selva mediana subperennifolia. Árbol, 6m. 28-VI- 2001. Saynes, A. 2262 ( NY); GoogleMaps NICARAGUA. Chinandega: 7.2 km E of Cinco Pinos along road to San Francisco del norte, fencerows and dry forest patches, 13°12’49”N 86°48’48”W. Tree, 15m. 8-I-2013. Stevens, W.D. 33975 ( MO); sobre el camino hacia el municipio de San Francisco del Norte , 13°12’55”N 86°48’54”W. Tree, 15m. 21-XII-2011. Quezada , J. B. 214 ( MO); GoogleMaps PANAMA. Darien: Yaviza, along rio Chucunaque, Quebrada Borbua Trail , approx. 2km W of Yaviza pan-highway. Unarmed tree, 75ft × 16”, 5-VI-1959. Stern, W. L . 91 (P); PERU. San Martín: Alto Rio Huallaga. XII-1929. Williams, L . 5817 (F); VENEZUELA. Zulia: Perijá, between the ríos Yasa and Tucuco , along the Machines and Los Ángeles de Tucuco road, 9°50-56’ N 72 °40-44’W, edge of forest remnant along Caño. Tree , 20m, 21-VI-1980. Davidse, G . 18383 ( MO); 20-30km al oeste de La Carretera Machiques-La Fría. Arbol , 20m. 4-V-1982. Bunting, G. S . 11541 (NY).
List with summary data of the other specimens examined: (200 of 250 specimens): — BOLIVIA: Meyer, G . 139 ( NY); Jardim , F . 593 ( F); Vargas , I . 634 ( NY); 1694 ( NY); 1798 ( NY); 1820 ( NY); Meneces , E . 815 ( MG); Killen , T . 1468 ( F); 4354 ( F); 6686 ( MO); 7147 ( P); 7575 ( F); Guillén , R . 1853 ( F); 3132 ( F); 3327 ( F); Saldias , M . 2727 ( F); 2805 ( NY); 3366 ( F); Anderson , W . 11939 ( US); Nee, M . 38797 ( NY); 41421 ( NY); Gentry , A . 73689; BRAZIL: Oliveira, A . 236 ( K); Medeiros , H . 335 ( RB); 365 ( RB); Rego , I . 448 ( MO); Magalhães , F . 530 ( INPA); Krukoff , B . 5708 ( MO); Lima , H . 6767 ( RB); Daly, D. 8143 ( MO); 8418 ( NY); 13163 ( MO); Croat , T . 85469 ( RB); 85859 ( NY); Silva , I . 334 ( RB); Folli, D. 1332 ( CVRD); Gomes , J . 3715 ( VIES); Duarte , A . 14047 ( RB); Monteiro , O . 1045 ( INPA); Bisby , F . 1279 ( NY); Krukoff , B . 1524 ( F); Nave, A. G . 1841 ( RB); Ivanauskas , N . 1996 ( ESA); Silva , M . 3380 ( INPA); Árbocz , G . 4254 ( ESA); Hatschbach , G . 65398 ( K); Rocha , M . 132 ( RB); Felix , D. 368 ( BHCB); Mello Barreto 1715 ( F); Vincent 1900 ( L); Rabelo, G . 1 ( HUENF); 92 ( HUENF); 96 ( HUENF); Nascimento , M . 115 ( HUENF); s.n. HUENF6576 ( HUENF); s.n. HUENF9217 ( HUENF); Gaglianone , M . 187 ( RB); Ribeiro , R . 1069 ( RB); 1070 ( RB); s.c. s.n. RB 354638 ( RB); Lima , H . 5331 ( RB); 6507 ( RB); Equipe do arboreto sn RB 439736 ( RB); RB 434473 ( RB); sc sn HUENF8291 ( HUENF); Glaziou A . 10649 ( P); Duarte , A . 14047 ( NY); Alves. F . 0112-030- 1996 ( RON); Silva , N . 443 (IAN); Vieira , M. 850 ( INPA); Leonaldo 1103-182-1997 ( RON); Bigio , N . 1185 ( RON); Lobato , L . 1241 (MG); Pereira , H. 1309-043-1996 ( RON); Teixeira , L . 1578 ( MG); Simon , M . 1590 ( CEN); Krukoff , B . 1591 ( K); Matos , J . 2107-080-1997 ( RON); Vinha , E . 3101-311-1997 ( RON); Cid, C. A . 4910 (INPA); Rosa , N. 4979 ( MG); Silva, M. G . 5846 ( INPA); 6120 ( MG); Thomas , W . 6461 ( NY); Prance , G . 8948 ( NY); Nee , M . 34566 ( MO); Paulino Filho H . 83-41 ( INPA); Crater s.n. P2771659 ( P) ; COLOMBIA: Villota, A . 252 ( UDBC); Smith , H . 261 ( P); Mutis , J . 2351 ( US); 4283 ( MA); 4684 ( US); Bartton? 4684 ( F); Callejas , R . 12214 (MO); CUBA: Ramon de la Sagra 679 ( P); sn P02771616 ( P); P02771637 ( P); P02771655 ( P); Roiy , J . 12 ( NY); Wright , C ; 145 ( NY); 1596 ( NY); sc 157 P02771656 ( P); Combs , R . 322 (NY); Luna , A. 392 ( NY); van Hermann 991 ( NY); Hioran ?, B . 2204 ( NY); Clemente , B . 2960 ( US); Shafer, J . 2962 ( NY); 5049 ( US); Leon 5309 ( NY); 9112 ( NY); 20071 ( US); Errers? 5426 ( P); Ekman , E . 5889 ( P); 10918 ( NY); Jack , J . 6891 ( NY); 6903 ( US); 7983 ( NY) ; EL SALVADOR: Marrínez, V . 184 ( MO); Sandoval , E . 564 ( MO); sn MO5889593 ( MO); Lewis , G . 1721 (NY); Rosales , J. 1757 ( MO); Calderón , S . 1975 ( US); 2165 ( US); Villacorta, R . 2152 ( MO); 2166 ( MO); Monro , A . 2974 ( MO); Molina , A . 12572 ( US); Standley, P . 20859 ( US); 21413 ( US); GUATEMALA: Tendy, G . 11-6 ( MO); Hughes , C . 1481 ( MO); Castillo , J . 1554 ( MO); Harmon , W . 5669 ( MO); Steyermark , J . 47876 ( US); HONDURAS: Yuncker, T . 8649 ( MO); Molina , A . 13032 ( US); 14562 ( US); Sousa , M . 13294 (MO); MEXICO: Gamon, J . 7 ( US); Nakamura, K . 40 ( US); Palmer, E . 66 ( US); Eggler, W . 188 (MO); Drake , E. 227 ( P); Velázquez , E . 232 ( MO); Merrill , R . 503 ( NY); Koeppen , R . 632 ( US); Magallanes , J . 248 ( MO); Sánchez , F . 777 ( US); Germán, M . 893 ( MO); Laughlin , R . 1162 ( US); Mcvaugh, R . 1384 ( NY); 15478 ( NY); Elorsa , M . 1417 ( NY); 7348 ( NY); Hughes , C . 1484 ( NY); Rivera , J . 2004 ( NY); King , R . 2741 ( NY); 4976 ( NY); Nelson , E . 2797 ( US); Shilom Ton, A . 3025 ( NY); 3188 ( MO); Salas , S . 3288 ( NY); 3474 ( NY); Steinmann , V . 3400 (NY); Re?o, B. 3455 ( US); Ventura, E . 3777 ( NY); Lott , E . 3816 (MO); 4099 (MO); Sousa , N. 4249 ( US); Matuda, E . 4453 ( US); 16845 ( NY); 17941 ( NY); Hinton , G . 4556 ( NY); 5703 ( US); 6106 ( NY); 9173 ( US); Martínez, S . 5143 ( US); 23879 ( NY); Enríquez , O . 6455 ( US); Cabrera , E . 7339 ( US); Sousa , M . 7385 ( NY); 7628 ( NY); 7652 ( NY); 7664 ( NY); 12556 ( MO); Méndez , A . 7781 ( NY); Nuñez , J . 9672 ( NY); Soto , J . 20233 ( NY); Breedlove, D. 25976 ( NY) 48491 ( NY) ; NICARAGUA: Coronado, I . 7979 (MO); PANAMA: Cohn, G . 4 ( MO); Duke , J . 5110 (MO); 13040 (MO); PERU: sc 1304 ( F); Tessmann , G . 3420 ( NY); Foster , R . 12608 (MO); VENEZUELA: Zambrano, O . 1337 (MO).
Poeppigia densiflora Tul. (1844: 122) . ≡ Poeppigia procera (Poepp. ex Spreng.) C. Presl. var. conferta Benth. in Mart. (1870: 54). Type:— BRAZIL. Piauí. 1845, Gardner 2142 K000054916 (Lectotype designated here: K! Isolectotypes: F!; L!; NY!; P!). ( Figures 7 View FIGURE 7 and 8 View FIGURE 8 ).
Shrubs to small trees, (0.8–) 2–8 (–15) m tall; up to 30 (–50) cm in diameter; bark whitish, gray, orange or reddish, soapy sap; flowering branches usually short, (2.7–) 5–16 (–22) cm long, very rarely reaching up to 35 cm. Canaliculate leaf rachis, measuring (2.2–) 3.8–8 (–11.5), with pubescence generally concentrated on the upper face, rarely glabrous, curved trichomes; petiole 3–8 mm long; petiolule about 1–2 mm long; (12–) 16–26 (–38) leaflets, measuring 0.7–1.4 (–2) × (0.2–) 0.4–0.5 (–0.7) cm; stipules lanceolate to elliptical, almost always persistent and commonly leafy in the axils near the base of the synflorescence, smaller stipules commonly along the rest of the branch, two per axillary bud; axillary bud lanceolate to elliptical. Inflorescences thyrsoid, distichous, terminal or terminal and axillary, the axillary inflorescences being almost always few and emitted only from the axils of the most distal leaves of the branch apex, generally in less than half the leaves of a branch, forming small and congested synflorescences, generally umbelliform, with (2.5–) 3–7 (–8) × 3–8 cm, the basal and apical inflorescences of similar size, the largest measuring 1.5–3.2 (–6) cm long, in rare cases inflorescences extending along almost the entire branch, 10–20 × 5–8 cm, all of similar size; persistent bracts, arranged in variable numbers along the primary and secondary axes of the inflorescence, without bracteoles. Flowers with sepals connate to about half the length of the calyx, with 4–6 (–7) × 1–2 mm, lobes about 1–2 mm long; 5 petals with (5–) 7–10 × 2–4 mm; stamens with anthers measuring (0.8–) 1–2 × 0.5–1 mm, filaments with 5–10 × 0.5 mm; ovary 5–8 × 1–2 mm, glabrous to slightly pubescent only along the suture. Samaroid fruits, elliptical to slightly lanceolate, (1.3–) 1.8–4.2 (–5.7) × (0.6–) 1–1.4 × 0.2–0.4 cm; pubescent stipe, pubescence commonly concentrated in the median region; calyx persistent in the fruit; 1 (–2) seeds with 4–7 × 3–5 × 1–2 mm. Seedlings with first pair of eophils elliptical and long petiolate.
Diagnosis: — Poeppigia densiflora are usually shrubs or rarely small trees, with generally shorter flowering branches, while P. procera are median to large trees, with generally longer flowering branches; P. densiflora has shorter leaf rachis with generally 16–26 leaflets generally shorter, while P. procera has longer leaf rachis with generally 38–68 leaflets generally longer; P. densiflora has stipules almost always persistent, at least in the upper part of the branch, commonly leafy, smaller stipules commonly along the rest of the branch, and a variable number of persistent bracts along the primary and secondary axes of the inflorescences, while P. procera has stipules and bracts almost always caducous. P. densiflora has only terminal or terminal and axillary inflorescences, but the axillary ones are almost always few and emitted only from the axils of the most distal leaves of the apex of the branch, forming small and congested synflorescences with less flowers and generally umbelliform, or when, in rare cases, inflorescences are emitted from leaves of the entire branch, the basal and apical inflorescences are small and of similar size, while P. procera has inflorescences emitted from the apex of the branch and also from the axils of many leaves along the branch, with thyrsoids gradually smaller towards the apex, forming lax triangular synflorescences with more flowers, and the more basal inflorescences are larger; P. densiflora has fruits elliptical and small with generally 1 seed and less commonly 2, while P. procera has all or most of the fruits of the same branch elongated and larger with, generally, 2–4 seeds.
Distribution and Habitat: — Poeppigia densiflora is endemic to Brazil, found in the following states: Minas Gerais, Bahia, Pernambuco, Piauí and Ceará ( Fig. 3 View FIGURE 3 ). It occurs mostly in the Caatinga of the Northeast region and of the far north of Minas Gerais. Occurs more rarely in ecotone areas between the Caatinga and the Cerrado in western Bahia, being observed in shrub or tree Caatinga, in “carrasco” vegetation and, very rarely on a single coastal tableland in northern Ceará; it grows on clayey, sandy soils or on rocky outcrops.
Conservation: — An EOO of 574,428 km 2 was estimated for P. densiflora . The species is endemic of a single region of Brazil and was recorded in few protected areas as Chapada do Araripe and Parque Nacional da Chapada Diamantina. There are many recent collections of this species in several areas of the Caatinga, however, some collectors indicate that the individuals are sparsely distributed in the areas of occurrence (Vanilda, M. 132; Bandeira, I. 115 [HUEFS]). It is also worth mentioning the numerous mentions about the use of the species to manufacture tools, or even firewood, even in the vernacular names of the species ( Queiroz 2009; present work). As stated for P. procera , the EOO and the amount of recent records may indicate a preliminary least concern status for P. densiflora , but the mentioned common uses of the species and the high deforestation rates in Caatinga ( Beuchle 2015; Santos 2014) indicate the need for more analysis to understand the threats this species may face now or in the near future.
Phenology: —Flourish from (October–) November–March (–August); bear fruits from (November–) December– June (–August). While flowering is related to the rainy season of the Caatinga, it is likely that individuals of P. densiflora lose their leaves in part of the dry season, since this species occurs abundantly in areas of extreme climate where most of the vegetation loses its foliage in the dry season. However, there is no collection of this species indicating it as a deciduous plant, which can be caused by the absence of reproductive structures in this period and, consequently, of collections, unlike the related genus Apuleia , which presents synchronized flowering in the leafless season. Some other deciduous trees are found in Dialioideae such as some species of Martiodendron (Falcão et al., in prep) and Dialium ( Rojo 1982) .
Etymology: —Although there is no explanation for the name described in Tulasne’s work, the specific epithet “densiflora” must refer to the arrangement of inflorescences concentrated at the apex of the branches forming a dense ensemble.
Uses: —The wood is considered resistant and used in the manufacture of various objects as tool handles and also as fuel; several specimens and also some of the vernacular names indicate that an extract used as soap is made from its exudate.
Vernacular names: —Pau-branco, Pau-de-machado, Lava-cabelo, Lava-cabeça, Aroeira-branca, Sabonete, Saboeiro, Sabão, Saboneteira, Coronheiro, Coração–de-negro, Caracu, Cabo-de-machado, Cabo-enchó, and Cabo-deinchó.
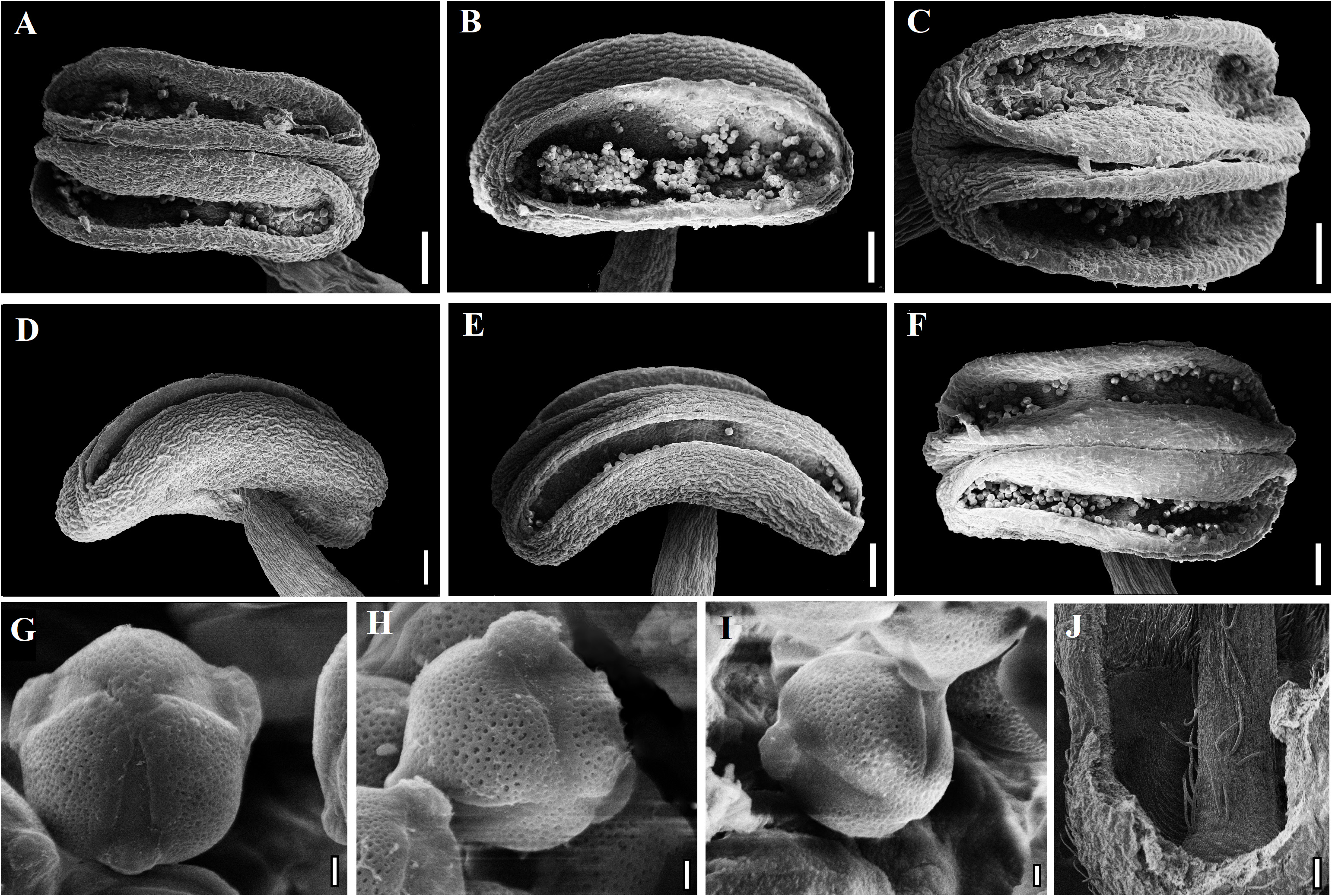
Taxonomic Notes: —The two species appear to be differentiated also by the seedling morphology in the shape of the first pair of unifoliolate eophils, which is elliptical and long petiolate in P. densiflora ( Figures 9A–C View FIGURE 9 ) and lanceolate, with the limbus almost indistinguishable from the petiole in P. procera ( Figures 9E–F View FIGURE 9 ). Subsequently, the first compound leaves emitted, also differ by an additional pair of leaflets in P. procera ( Figures 9E–G View FIGURE 9 ) in relation to P. densiflora ( Figures 9C–D View FIGURE 9 ) in an equivalent stage, a difference that will be accentuated in an increasing number difference on the following leaves. However, as seeds for germination were obtained from only one specimen of each species, these characters should be taken with caution as possible taxonomic distinctions. No significant difference was observed between the two species concerning seeds and embryos morphology ( Figures 9H–R View FIGURE 9 ). Regarding the morphology of anthers, their dehiscence and the morphology of pollen grains, the stamens of P. procera from continental America, P. procera from Cuba and P. densiflora were checked with SEM. No consistent morphological variation was found between the three groups with the exception of the anther shape, which is commonly straight in P. procera and slightly hawk in P. densiflora ( Fig. 10 View FIGURE 10 ). Again, due to the low sampling, such differences should be taken with caution.
The interior of the hypanthium of both Poeppigia species does not present uncinate trichomes or stomata ( Fig. 10J View FIGURE 10 ) typical of some genera of Dialioideae , such as Dialium , Uittienia C. G. G. J. van Steenis (1948: 416) , Distemonanthus , Androcalymma Dwyer (1958: 295) , Dicorynia and Apuleia ( Tucker 1998, Zimmerman et al. 2013, Falcão et al. 2020a, in prep). Although Britton & Rose (1930) mention that the Poeppigia stipe is adnate to the hypanthium inner wall, in all specimens observed in this work, the stipes forms from its central region ( Fig. 10J View FIGURE 10 ), although sometimes, somewhat oblique, as also observed by other authors ( Presl 1830, Richard 1846, Bentham 1870, Standley & Steyermark 1946, Zimmerman et al. 2013), which differs it, for example, from Apuleia , which has the carpel adnate to the abaxial wall of the hypanthium along its entire length in its monoclinous flowers ( Falcão et al. 2020a). Poeppigia stands out from the other Neotropical genera of Dialioideae for having very poorly developed and hardly visible leaf primordia in its seeds ( Figures 9Q and R View FIGURE 9 ), while in the other genera such structures are much more conspicuous (Falcão et al. in prep). And also for having the first pair of eophils narrow and with a wedged base, and the following eophils already with several leaflets and paripinnate while Dialium , Dicorynia , Martiodendron and Apuleia (Falcão et al. in prep) Have the first eophils wide and with a cordate to truncated base, being followed by unifoliolate to trifoliolate eophils and, subsequently, imparipinnate leaves with alternating leaflets. More comparisons between seedlings of some of the genera of the former subtribe Dialiinae and current subfamily Dialioideae were made by Hartmann & Rodrigues (2014).
Bentham (1870) reduced P. densiflora to the varietal rank, renaming it as P. procera var. conferta . The description was vague and disregards several important morphological characters of P. densiflora , in addition to indicating individuals from Piauí, Bahia and Rio de Janeiro, suggesting the occurrence of the variety throughout eastern Brazil and P. procera var. procera occurring in Minas Gerais, Central America and the Amazon, a distribution not consistent with the observed in this work, where P. densiflora occurs only in the Caatinga and the populations of Rio de Janeiro, southeastern Minas Gerais and Espírito Santo being similar to those found in the Amazon and Central America. In the illustration of P. procera var. conferta presented in Bentham (1870), a long fruit like those of P. procera is observed, however, this is possibly due to the fact that Bentham considers as belonging to this variety the plants of Rio de Janeiro that have elongated fruits and belong to the species P. procera . Even little elongated fruits are rare in P. densiflora . In the few plants where they are found, a little elongated fruit is observed among dozens of small fruits typical of the species. The opposite happens very more commonly in P. procera where, in branches with many fruits, those more peripheral in the structure of some of the subunits of the synflorescence are often smaller and shorter, not having fully developed as the central ones and can sometimes resemble P. densiflora fruits ( Figures 2G–H, J View FIGURE 2 ). However, in all specimens observed with several developed fruits, all or most of the fruits in the synflorescence are elongated, a character never reached in P. densiflora , which has more uniform sized fruits ( Fig. 8G View FIGURE 8 ). Some rare specimens of P. procera in Mexico, Central America and Minas Gerais (Lewis, G. 1721 [NY]; Molina, A. 13032 [ US]; Rero, B. 3455 [ US]; King, R. 4976 [NY]; Cabrera , E. 7339 [ US]; Valente, G. 394 [VIC]) present only a few poorly developed fruits with reduced size, possibly due to a very weak fruiting period. In these anomalous cases, the species remain clearly discernible due to the vegetative characteristics. The stipe of the fruit is always pubescent in P. densiflora , this pubescence being very commonly concentrated in a tuff on the middle portion of the stipe ( Fig. 7S View FIGURE 7 ), while P. procera has more commonly glabrous stipes ( Fig. 1T View FIGURE 1 ) and less commonly pubescent stipes, with this pubescence being uniformly distributed throughout the stipe. Again, is noticeable the greater morphological variation in P. procera , also more widely distributed geographically and ecologically, than in P. densiflora , more restricted geographically and ecologically and less variable morphologically.
Unlike P. procera , where, in addition to the terminal thyrsoid inflorescence, several inflorescences are emitted from the axils of many leaves on a branch, forming a triangular synflorescence, a large lax set where the thyrsoids produced are gradually smaller towards the apex of the branch. In P. densiflora , there is a terminal thyrsoid inflorescence that can be unique, or be added to a small number of other inflorescences emitted from only a few more distal leaves on the branch, forming a small, very congested and generally umbelliform synflorescence, so that the number of inflorescences per branch, the number of flowers per branch, the length of the synflorescence and the length of the most basal thyrsoids of the synflorescence are smaller in P. densiflora than in P. procera . It is important during the observation of herborized specimens, to pay attention to the possible breakage of part of the inflorescence or poorly developed inflorescences that, in a few specimens give the impression of small inflorescences in P. procera . It should be also noted that these variations in structure and shape of the inflorescence can play an important role in the interaction with pollinators, which, in turn, can result in reproductive isolation processes and in the speciation of taxa (Abbot et al. 2008, Van der Niet & Johnson 2014). Rare individuals of P. densiflora (9 out of 160 analyzed: Ramalho, F. 69 [RB]; Castro, A. s.n. EAC27452[EAC]; 654 [EAC]; 927 [EAC]; Nunes, E. s.n. EAC 7483 [EAC]; Falcão, M. 78 [RB]; Andrade-Lima, 68-5180 [F]; Bezerra, P. s.n. EAC5124 [EAC]; Cardoso, D. 921 [HUEFS]), present an exception regarding the disposition of their inflorescences, not having them concentrated at the apex of the branch as is typical in the species, but extending to the base or more than half of the branch ( Fig. 11 View FIGURE 11 ). However, such individuals remain distinct from P. procera for the other floral and vegetative characteristics, as well as for the inflorescences themselves, which, even produced in the axils of many leaves, have similar size and number of flowers, giving the synflorescence a considerably uniform width along its length. Unlike P. procera , where the inflorescences increase considerably in size and in number of flowers from the apex to the base of the branch, giving the synflorescence a triangular shape. It is worth mentioning that, in some of these 9 specimens, the anomalous branches are interspersed with branches with typical inflorescences.
Another less rare variation (15 out of 160 specimens analyzed: Bezerra, P. s.n. [ EAC5124 View Materials ]; Laurênio, A. 78 [ NY]; Carvalho 3866 [ RB]; Cardoso, D. 921 [ HUEFS]; 257 [ HUEFS]; Falcão, M. 78 [ RB]; Nunes, E. s.n. [ EAC7483 View Materials ]; Queiroz , L. 16154 [ HUEFS]; 14700 [ HUEFS]; Fernandes , A. s.n. [ EAC 11386]; Melo , E. 3179 [ HUEFS]; França, F. 1955 [ HUEFS]; Jardim , J. 1211 [ NY]; Moraes , M. 624 [ HUEFS]; Alves , L. 13 [ HUEFS]), is that of specimens who have long flowering/fruiting branches, with 22–35 cm in length, in contrast to that found in most individuals of P. densiflora , which has branches more commonly up to 16 cm and less commonly up to 22 cm. It is noteworthy that in most of these anomalous individuals, there are typical short branches, interspersed with an unusual long branch. It should be also noted that several of the individuals with abnormally long branches are also individuals with anomalous inflorescences. However, both cases where variation in the diagnostic characters of the species can be found, they appear to be individual anomalies and not any taxonomic indications, due to the strong variation of the anomaly in degree among these individuals, its great rarity, its instability in the same individual and to its appearance without any geographic pattern throughout the species distribution .
Biogeographical Notes: —As for the relationship between P. procera and P. densiflora , to try to understand the causes of reproductive isolation that may have led to speciation events in the genus, we can also correlate the distribution of the two species with the topography ( Fig 10 View FIGURE 10 ). We observed that, throughout South America and most of Central America, P. procera almost always occurs in areas of lower elevations, from 100–300 (– 600 m), with rare exceptions (7 specimens in 170 analyzed in these regions) above 600m in Cuba, Honduras, Guatemala and the Brazilian state of Minas Gerais. The main variation of this pattern are the populations of Mexico and El Salvador, which, although most of them also occurring below 600m, have a considerable number of specimens above this mark (17 specimens out of 80 analyzed in these regions). P. densiflora , on the other hand, almost always occurs in areas of higher elevations, with (300–) 500–1100m, with rare specimens in areas bellow 300m on a single coastal tableland in Ceará state, some valleys in the interior of Piauí state and in the east of Bahia state. So that this factor may be involved in the reproductive isolation between the two taxa, a fact also observed in the related genus Dialium ( Falcão et al. 2016). When we correlate the distribution with rainfall data ( Fig. 6 View FIGURE 6 ), the difference between the two species becomes even clearer, with P. procera occurring in wetter areas (1600-2200mm) of Central America, southern Amazonia and southeast from Brazil, with few specimens in less humid areas in Minas Gerais state, northern Colombia and western Mexico (1000-1600mm) and P. densiflora inhabiting much drier regions, contained within the average 1000 mm per year isohyet, so that two species live in very different environments in multiple aspects. It is also worth mentioning that the morphological differences of the taxa are maintained even when the environment in which the species occurs is modified, since seedlings of both species cultivated with similar soil types, humidity and luminosity, maintained, since very young, the distinctive vegetative characteristics ( Figures 9A–G View FIGURE 9 ). If we compare this event of reproductive isolation between P. procera and P. densiflora with, for example, that among the aforementioned populations of P. procera or the Neotropical species of Dialium ( Falcão et al. 2016), we can suggest possible distinct steps in speciation processes in each of them. In Dialium and among the populations of P. procera we have possible recent disjunctions with still considerably similar environments (Amazon and Atlantic Forest) and subtle variations in morphological frequencies, which may indicate the beginning of speciation processes, while, in relation to the populations of Poeppigia in Northeast Brazil, we have isolation in environments strongly different, so that the great difference in habitats and ecological conditions between the northeastern Caatinga and the Atlantic Forest / Amazon may have led to varied selective pressures.Among them, we can mention the low precipitation, soil type, high temperatures and solar irradiation of the Caatinga ( Queiroz 2009). Such pressures may be related to some of the morphological differences of P. densiflora as the smaller leaves contributing to less water loss in an environment with strong solar irradiation, smaller fruits and reduced habit due to poor soils and thicker stipules and bracts to protect leaf and inflorescence buds, resulting on these structures to be persistent. These selective pressures, added to the geographic isolation, may have led to a process of allopatric speciation that resulted in the two current species of Poeppigia .
Representative specimens examined (40 of 160 specimens): — BRAZIL: Bahia: Morpará, Morro da Antena, 11°33’25”S 43°16’40”W, Caatinga. Árvore , ca. 4m, 17-XII-2007. Conceição, A. A GoogleMaps . 2719 ( HUEFS); Milagres, morro do Couro or morro São Cristóvão , 12°54’S 39°52’W. Shrub to 3m, 6-III-1977. Harley, R. M GoogleMaps . 19414 ( MG); Jeremoabo, 10°13’S 38°19’W, Savana / Estepe. Árvore 3-4m, 4-VI-1981. Orlandi, R. P GoogleMaps . 428 ( NY); Abaíra, estrada nova de Abaíra- Catolés, 13°14’S 41°40’W, Caatinga. Árvore , até 5m, 28-XII-1991. Harley, R. M GoogleMaps . 50497 ( K); Manoel Vitorino , Rod. M . Vitorino Caatingal, km14, Caatinga. Árvore , 9m × 20cm, 16-II-1979. Mattos Silva, L. A . 285 ( CEPEC); Pilão Areado: Sítio do Poção. XI-2009. Prata, A. P . s.n. ASE 18363 ( ASE); Caminho para brejo do Zacarias, 10°S 42°47’59”W, Vegetação dunas. Árvore , 3.5m, 9-XII-2005. Conceição, A. A GoogleMaps . 1540 ( HUEFS); Tucano: Povoado da Pedra Grande, Serra do Pai Miguel , 11°7’24”S 38°46’25”W, Caatinga arbustiva. Árvore , 4-I-2006. Cardoso, D, 921 ( HUEFS); sítio do Mandacaru , ca 11km de Tucano, 10°52’51”S 38°46’36”W, Caatinga arbóreo-arbustiva, solo arenoso e pedregoso. Árvore ca 3,5m, 8-IV-2004. Cardoso, D. 6 ( HUEFS); ca. 23 km na estrada de Tucano para Euclides da Cunha , Caatinga. Árvore , ca. 6m, 22-III-1992. Carvalho, A. M GoogleMaps . 3930 ( CEPEC); Feira de Santana , Horto da UEFS , 12°16’7”S 38°56’22”W. Arbusto , 3-4m, 16-XI-2017. Falcão, M. J GoogleMaps . 78 (RB); Ceará: Estr. Mossoró-Aracati, pouco além da divisa, solo arenoso. Árvore 7-8m, 26-I-1968. Andrade-Lima 68-5180 ( F); Próximo à Varzinha Itaiçaba, 7km da cidade. Árvore, 14-I-2001. Castro, A. S. F . 927 ( EAC); Aracati, tabuleiro litorâneo. Árvore mediana, 3-IV-1977. Fernandes, A . s.n. EAC3129 View Materials ( EAC); Rodagem Boqueirão do Cesário–Aracati, tabuleiro litorânio. Árvore mediana, 18-I-1975. Fernandes, A . sn EAC2536 View Materials ( EAC); 2-4 km of Campas Salas, 7°14’S 40°25’W, Caatinga. Tree , 8m, 15-II-1985. Gentry, A. H GoogleMaps . 50126 ( MO); Aiuaba, Gameleira de baixo, Caatinga. Árvore , 12-IV-1991. Figueiredo, M. A . 138 ( EAC); Estação Ecológica Aiuaba , 6°36-44’ S 40 °7-19’W, 24-XII-2004. Lemos, J. R . 278 (EAC); Minas Gerais: Manga, Caatinga. Árvore, 8m. 27-I-1990. Pequeno, P. H. A . s.n. RB 746785 ( RB); Jaíba, mata seca. Árvore , 22-XI-1982. Teixeira s.n. RB 363983 ( RB) ; Pernambuco: Santa Maria de Boa Vista, Caatinga. Arvoreta , + 5m, 4-II-1998. Ribeiro, W . s.n. SP 329792 ( SP); Inajá, Reserva Biológica da Serra Negra, floresta aberta. Árvore , ca. 5-6m, 5-VI-1995. Laurêncio, A . 78 ( NY); Araripina. Arvoreta , ca. 3m, 13-XII-1997. Miranda, A. M . 2943 ( HST); Margem da estrada Afrânio-Petrolina , 15 km de Afrânio. Árvore, 5m, 24-IV-1971. Heringer, E. P . 336 ( RB); Buíque, fazenda Laranjeiras, nas vertentes a sotavento de Chapada de São José , 8°37’S 37°10’W, caatinga arbustiva densa. Árvore , ca. 8m, 5-V-1995. Rodal, M. J. N GoogleMaps . 536 ( NY); fazenda Laranjeiras, nas vertentes a sotavento de Chapada de São José, 8°37’S 37°10’W, Caatinga arbustiva densa. Árvore , ca. 6m, 16-VI-1995. Figueiredo, L GoogleMaps . 90 ( K); Mirandiba, sítio Chacal, Caatinga arbustiva densa. Árvore , 4-6m, 6-I-2008. Córdula , E . 342 ( UFP); estrada para Cacimba Nova, 8°07’50”S 38°40’26”W, Caatinga arbustiva densa. Árvore , ca. 4m, 32- III-2006. Córdula , E GoogleMaps . 25 ( NY); Serrotinho, 8°7’39”S 38°42’29”W, Caatinga arbustiva densa. Árvore , 2-4m, 10-II-2007. Córdula , E GoogleMaps . 215 ( RB); Ipubi, Serrolândia. Árvore ca 15m x 15,5cm CAP, 21-V-2018. Santos, W. B . 12 ( RB); Ibimirim, Estrada Ibimirim-Petrolândia, Caatinga arbustiva densa, solo arenoso alaranjado. Árvore ca 5m, 5-VI-1995. Gomes, A. P. S . 38 (NY); Piauí: Jaicós, margem estrada BR-316, 5 km depois da junção com BR-230, canga residual e areia. Árvore, 4-5m, 5-XII-1971. Andrade-Lima, D. 1170 ( UFS); Caracol , Serra das Confusões, mata subcaducifolia .
Árvore mediana, 7-XII-1980. Fernandes, A . s.n. EAC9094 View Materials ( EAC); Picos, BR-316, carrasco. Árvore , 16-XII-1978. Fernandes, A . s.n. EAC5124 View Materials ( EAC); Simões, Chapada do Araripe. 3-XII-1979. Fernandes, A . s.n. EAC7583 View Materials ( EAC); Belém do Piauí, estrada Padre Marcos. Árvore , 10-I-1999. Castro, A. S. F . 654 ( EAC); São Raimundo Nonato: Árvore pequena, 8-IV-1979. Fernandes, A . s.n. EAC6119 View Materials ( EAC); km 6 da Fundação Ruralista (Sede), estrada para Vitorino, 9°00’S 42°0W, open-branched canopy. Tree to 10m × 15cm, 19-I-1982. Lewis, G. P GoogleMaps . 1125 ( RB); Cel josé Dias, Mirante , PARNA Serra da Capivara , próx. a BR , Caatinga , 10-V-2008. Castro, A. S. F . 2024 ( EAC); São Raimundo Nonanto, Caatinga arbustiva densa. 4m de altura, 5-I-1985. Laure Emperaire 2672 ( P); entre Itaueiras e Canto do Burití. Árvore de flores amarelas, 17-XII-1978. Bezerra, P . s.n. EAC5140 View Materials ( EAC) .
List with summary data of the other specimens examined: (120 of 160 specimens): — BRAZIL: Bahia: Alves, L. 13 (HUEFS); 154 (ALCB); 361 (ALCB); Maia , H. 19 (ALCB); Costa, T.F. 21 (ALCB); Monteiro, V. 26 (HUEFS); Schulte, T. 32 (RB); Cardoso, D; 33 (HUEFS); 257 (HUEFS); 3518 (HUEFS); Lima, A. 50-438 (RB); Casaes, M. 60 (ALCB); Klitgaard, B. 65 (RB); Ramalho, F. 69 (RB); Falcão, M. 79 (RB); Bandeira, I. 115 (HUEFS); Vanilda, M. 132 (HUEFS); Silva, S. 132 (RB); 202 (RB); Carvalho, A. 138 (CEPEC); 3866 (CEPEC); sc 216 (ALCB); Gusmão, E. 257 (CEPEC); Neves, S. 281 (HUEFS); Silva, F. 301 (HUEFS); Santana, D. 326 (ALCB); Costa, G. 461 (HUEFS); Carvalho-Sobrinho J. 534 (HUEFS); Sarmento, A. 620 / 80 (RB); Moraes, M. 624 (HUEFS); 1115 (RB); Meireles, J. 650 (RB); Cavalcanti, F. 660 (EAC);? 668 RB6254 (RB); Nunes, T. 683 (HUEFS); Lana , J. 709 (CEPEC); Orlandi, R. 718 (MG); Silva-Castro, M. 910 (HUEFS); Conceição, A. 1025 (HUEFS); 2253 (HUEFS); 2598 (HUEFS); 1128 (HUEFS); 3111 (HUEFS); Jardim, J. 1211 (NY); Queiroz, E. 1345 (SPF); Melo, E. 1355 (HUEFS); 1432 (ESA); 1524 (ASE); 3179 (HUEFS); 10632 (HUEFS); 11107 (HUEFS); 11861 (HUEFS); Cardoso, L. 1521 (RB); Neves, D. 1549 (RB); G.C.P.P. 1556 (VIES); Santos, R. 1642 (HUEFS); Giulietti, A. 1714 (HUEFS); Ferreira, F. 1759 (RB); Blanchet 2667 (P); 2796 (NY); 3099 (K); 8612 (SP); França, F. 1955 (HUEFS); Hatschbach, G. 50072 (MBM); 50159 (RB); Silva, L. 2296 (CEPEC); Macedo, G. 2498 (HUEFS); Prata, A. 2546 (ASE); Santos, T. 2553 (CEPEC); Guedes, M. 2685 (CEPEC); 2873 (ALCB); 6997 (ALCB); 13089 (ALCB); 13316 (ALCB); 13460 (ALCB); 13484 (ALCB); 14474 (ALCB); 30140 (ALCB); 30534 (ALCB); Noblick, L. 3610 (HUEFS); Lewis, G. s.n. F2018344 (F); Queiroz, L. 3641 (RB); 3726 (HUEFS); 3850 (HUEFS); 9914 (HUEFS); 14700 (HUEFS); 16154 (HUEFS); Fernandes, A. s.n. EAC3757 (EAC); sn EAC11386 (EAC); s.n. EAC14116 (EAC); Hind, N. 4542 (ALCB); Saar, E. 5101 (ALCB); Amorim, A. 5518 (CEPEC); Arbo, M. 5659 (NY); Sobral, M. 5880 (ICN); 5884 (ICN); Fontana, A. 6387 (EAC); Miranda, A. 6451 (HST); Pinto, G. s.n. F1886034 (F); Harley, R; 27027 (NY); 54737 (HUEFS); 55353 (HUEFS); 56925 (HUEFS); sc sn RB275837 (RB); Ceará: Fr. Allemão 454 (P); Pernambuco: Lima, A. 61-3613 (RB); Laurênio, A. 115 (NY); 366 (NY); Piauí:? 61 US 2999427 ( US); Lewis, G. 1082 (NY); Emperaire, L. 2178 (RB); 2343 (P); Lima- Verde, L. 3139 (EAC); Nunes, E. s.n. EAC7483 (EAC); Fernandes, A. s.n. EAC10879 (EAC); Lima, D. 13310 (HST); Alencar, M. s.n. EAC19714 (EAC); s.n. EAC10631 (EAC); Castro, A. s.n. EAC27452 (EAC);
| P |
Museum National d' Histoire Naturelle, Paris (MNHN) - Vascular Plants |
| F |
Field Museum of Natural History, Botany Department |
| NY |
William and Lynda Steere Herbarium of the New York Botanical Garden |
| RB |
Jardim Botânico do Rio de Janeiro |
| R |
Departamento de Geologia, Universidad de Chile |
| T |
Tavera, Department of Geology and Geophysics |
| K |
Royal Botanic Gardens |
| A |
Harvard University - Arnold Arboretum |
| M |
Botanische Staatssammlung München |
| B |
Botanischer Garten und Botanisches Museum Berlin-Dahlem, Zentraleinrichtung der Freien Universitaet |
| C |
University of Copenhagen |
| S |
Department of Botany, Swedish Museum of Natural History |
| J |
University of the Witwatersrand |
| L |
Nationaal Herbarium Nederland, Leiden University branch |
| G |
Conservatoire et Jardin botaniques de la Ville de Genève |
| N |
Nanjing University |
| E |
Royal Botanic Garden Edinburgh |
| UB |
Laboratoire de Biostratigraphie |
| BHCB |
Universidade Federal de Minas Gerais |
| VIC |
Universidade Federal de Viçosa |
| H |
University of Helsinki |
| RON |
Universidade Federal de Rondônia |
| W |
Naturhistorisches Museum Wien |
| CEN |
EMBRAPA Recursos Geneticos e Biotecnologia - CENARGEN |
| U |
Nationaal Herbarium Nederland |
| I |
"Alexandru Ioan Cuza" University |
| MO |
Missouri Botanical Garden |
| COL |
Universidad Nacional de Colombia |
| UDBC |
Universidad Distrital |
| O |
Botanical Museum - University of Oslo |
| MG |
Museum of Zoology |
| INPA |
Instituto Nacional de Pesquisas da Amazonia |
| CVRD |
Reserva Natural da Vale |
| VIES |
Federal University of Espírito Santo |
| ESA |
Universidade de São Paulo |
| MA |
Real Jardín Botánico |
| V |
Royal British Columbia Museum - Herbarium |
| HUEFS |
Universidade Estadual de Feira de Santana |
| EAC |
Universidade Federal do Ceará |
| CEPEC |
CEPEC, CEPLAC |
| ASE |
Universidade Federal de Sergipe |
| UEFS |
Laboratorio de Ictiologia |
| SP |
Instituto de Botânica |
| UFP |
Universidade Federal de Pernambuco |
| UFS |
Nyabyeya Forestry College |
| BR |
Embrapa Agrobiology Diazothrophic Microbial Culture Collection |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |