Balanodytes taiwanus Utinomi, 1950
|
publication ID |
https://doi.org/ 10.11646/zootaxa.3694.3.3 |
|
publication LSID |
lsid:zoobank.org:pub:216E5E96-9535-45E7-9044-E60B48882632 |
|
DOI |
https://doi.org/10.5281/zenodo.6163735 |
|
persistent identifier |
https://treatment.plazi.org/id/03A687D6-EB63-051E-FF3B-FA60FB47D365 |
|
treatment provided by |
Plazi |
|
scientific name |
Balanodytes taiwanus Utinomi, 1950 |
| status |
|
Balanodytes taiwanus Utinomi, 1950 c
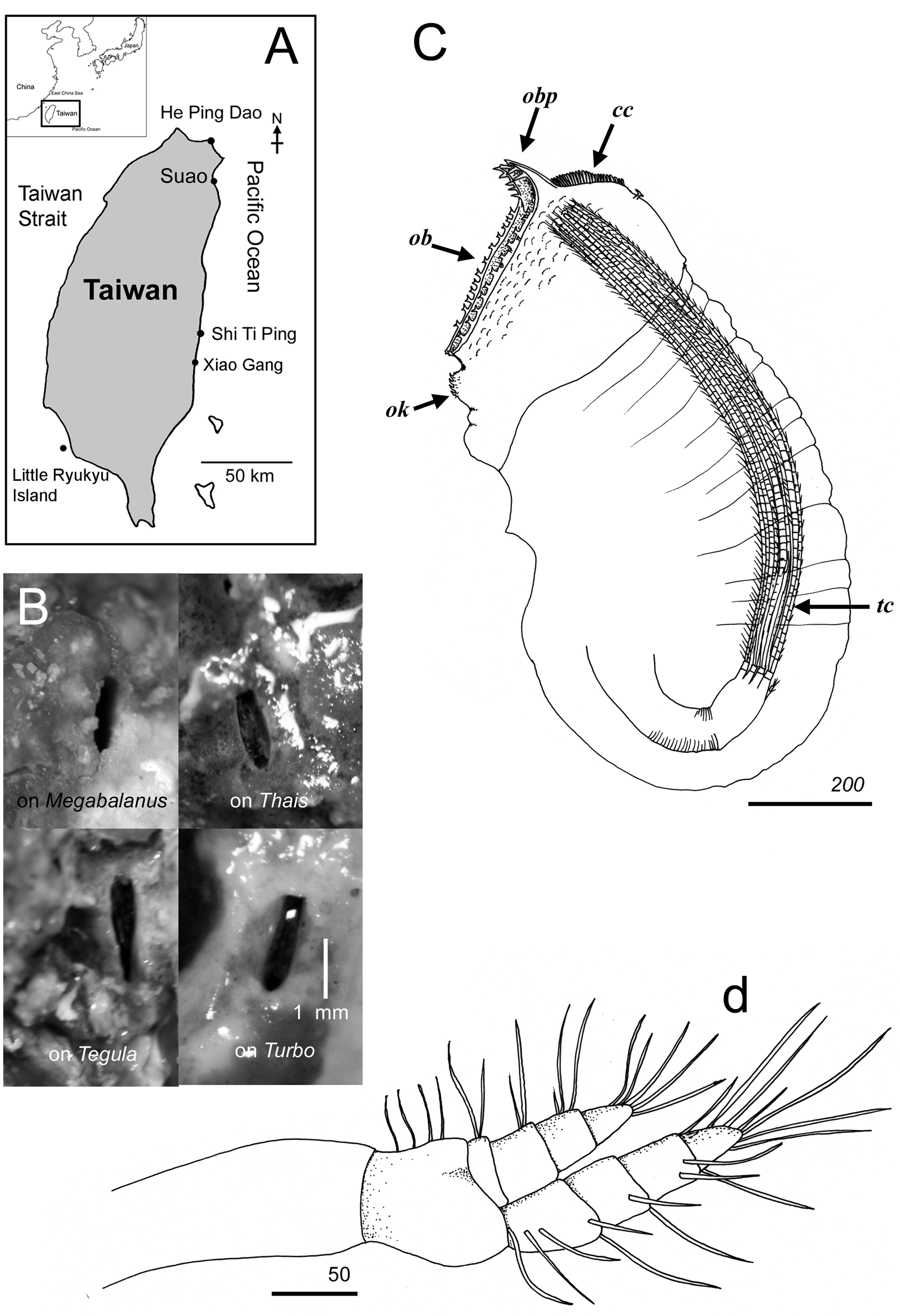
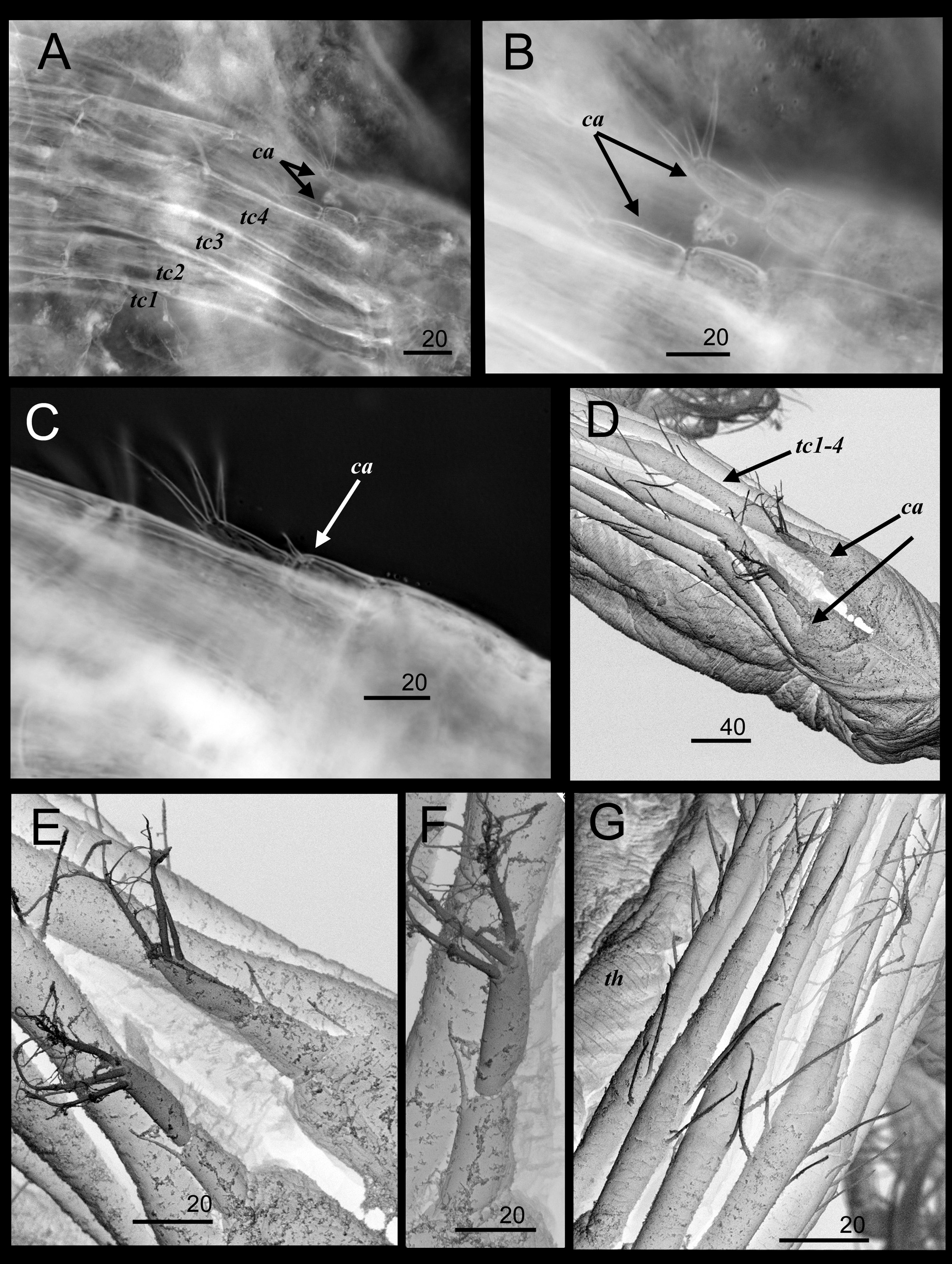
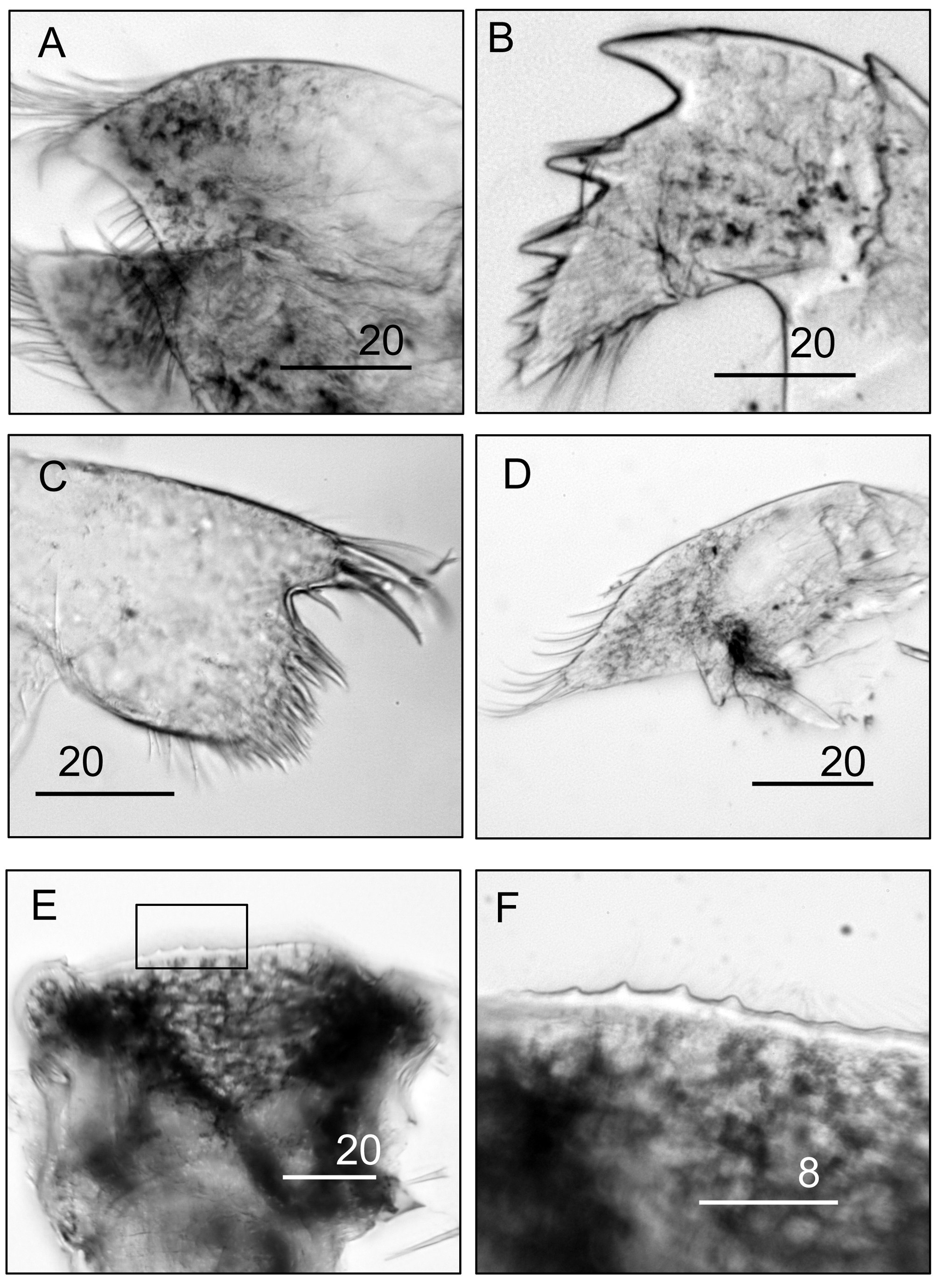
Figures 1 View FIGURE 1 A–G, 2B–D, 3A–L, 4A–G, 5A–F
Balanodytes taiwanus Utinomi, 1950c: 454 , 459, fig. 16. — Utinomi 1950a: 1, figs 1–2. Kochlorine habei Tomlinson, 1963: 270 , figs 7–17.
Lithoglyptes habei . — Tomlinson 1969: 61–65, fig. 12.
Armatoglyptes habei . — Kolbasov & Newman 2005: 47–51, figs 5a–f, 6a, d. — Kolbasov 2009: 301, fig. 97. Armatoglyptes taiwanus . — Chan et al. 2012: 5–19.
Material examined. Syntypes, SMBL type 52. 2 specimens, in the base of Megabalanus tintinnabulum, Takao (=Kaoshiung), 1938. CEL- Acro-RYU-01, 1 specimen, in a gastropod shell, Lobster Cave, Siaoliouciou, Pingtung, Taiwan, 16 August 2010. CEL-Acro-RYU-02, same data as CEL-Acro-RYU-01. CEL-Acro-RYU-03, same data as CEL-Acro-RYU-01. CEL-Acro-sp-8, 1 specimen, in Megabalanus shell, He-Ping-Dao, Keelung, Taiwan, 17 January 2010. CEL-Acro-sp-6, 1 specimen, in Megabalanus shell, He-Ping-Dao, Keelung, Taiwan, 14 January 2010. CEL-Acro-sp-16, 1 specimen, in Turbo setosus , Shi-Ti-Ping, Hualien, Taiwan, 21 January 2010. CEL-Acrosp-15, 1 specimen, in Turbo setosus , Shi-Ti-Ping, Hualien, Taiwan, 21 January 2010. CEL-Acro-sp-3, 1 specimen, in dead coral skeleton, He-Ping-Dao, Keelung, Taiwan, date unknown. CEL-Acro-sp-10, 1 specimen, in Thais clavigera , He-Ping-Dao, Keelung, Taiwan, 18 January 2010. CEL-Acro-sp-31, 1 specimen, in the base region of Cyphastrea , He-Ping-Dao, Keelung, Taiwan, 25 February 2010. CEL-Acro-sp-13, 1 specimen, in Turbo setosus , Shi-Ti-Ping, Hualien, Taiwan, 21 January 2010. CEL-Acro-sp-6, 1 specimen, in Megabalanus shell, He-Ping-Dao, Keelung, Taiwan, 14 January 2010. CEL-Acro-sp-4, 1 specimen, in Thais clavigera shell, He-Ping-Dao, Keelung, Taiwan, 13 Jan 2010. CEL-Acro-sp-14, 1 specimen, in Thais clavigera shell, He-Ping-Dao, Keelung, Taiwan, 21 January 2010.
Diagnosis. Opercular bars with bifid teeth, posterior projections of opercular bars pronounced, recurved with 1–3 pairs of sharp spine-like teeth, lateral bars feeble, orificial knob distinct, with curved teeth, terminal cirri consisting of 4 pairs, caudal appendages 2-segmented, pedestals absent.
Descriptions. (based on syntype and our own material). Female ( Figs 1 View FIGURE 1 C–G, 2C, D, 3, 4, 5). Shape of burrow opening varying from narrow oval or drop-shaped to oval ( Fig. 2 View FIGURE 2 B). Opercular bars approximately 400 Μm long, armed with series of bifid teeth and simple setae ( Figs 1 View FIGURE 1 C, 2C, 3B, C, F, G); posterior processes pronounced, recurved (anteriorly bent), with small bifid teeth along their length (teeth number variable, Figs 1 View FIGURE 1 C, E, 3B, C), simple spine-like teeth apically ( Figs 1 View FIGURE 1 E, 3B, C, F, G); comb collar long ( Fig. 3 View FIGURE 3 B), with feather-like cuticular projections fused at bases ( Fig. 3 View FIGURE 3 I, J), pair of bifid teeth located at posterior end of comb collar ( Fig. 3 View FIGURE 3 B). Lateral surface of opercular area deep-orange when alive ( Fig. 3 View FIGURE 3 A), color off-white in ethanol, covered with sparse setae and massive multifid scales ( Fig. 3 View FIGURE 3 K), lateral bars feeble. Orificial knob distinct, globular-shaped, surface with setae and curved, simple or bifid teeth ( Figs 1 View FIGURE 1 D, 3D, E, H, L). Attachment disc (removed in preparation) composed of several layers of striated cuticle.
Terminal cirri consisting of 4 pairs ( Fig. 4 View FIGURE 4 A), orange color when alive, white when preserved in ethanol; anterior surfaces of annuli of terminal cirri armed with 2 pairs of plumose setae, posterior surfaces with sparse serrated setae ( Fig. 4 View FIGURE 4 D, G). Caudal appendages 2-segmented, pedestal absent, basal segment with simple distal seta, terminal segment with 4 distal plumose setae ( Figs 1 View FIGURE 1 F–G, 4B, C, E, F). Mouth cirri ( Fig. 2 View FIGURE 2 D) with 4- segmented posterior ramus shorter than 4-segmented anterior ramus, both rami with long plumose setae.
Labrum saddle-like, with small, fine, blunt teeth on anterior margin ( Fig. 5 View FIGURE 5 E, F). Mandible with 3 teeth in upper two-thirds, lower one-third with row of 4 sharp denticles, inferior angle with sparse setae ( Fig. 5 View FIGURE 5 B). Mandibular palp trapezoid, simple setae distally along exterior margin ( Fig. 5 View FIGURE 5 D). Maxillule with 2 long upper cuspidate setae, notch with short sharp seta, lower two-thirds of cutting edge with numerous short sharp setae ( Fig. 5 View FIGURE 5 C). Maxilla triangular ( Fig. 5 View FIGURE 5 A), long, dense setae on exterior margin, sparse setae at inferior margin.
Geographical distribution. Widespread: Maldives, Seychelles, Red Sea, Aden and Persian Gulf, Japan, Vietnam, Java, New Guinea, northern, eastern and southern Australia, Taiwan, Thailand, Philippines, Hawaii and San-Felix ( Chile), from tidal to 220 m depth (Tomlinson 1969; Kolbasov 2009; Jones et al. 1990; Poore 2012).
Remarks. In the present study, we have re-examined one of the syntypes of Balanodytes taiwanus (note the second syntype had only the opercular bar left), revealing that this species has a pair of caudal appendages (without pedestal). Thus, the redescription by Utinomi (1950a) stating that B. taiwanus lacks caudal appendages is incorrect. This is further supported by additional collections of Balanodytes taiwanus from various locations in Taiwan (including Siaoliouciou Island, which is very close to the type locality at Kaoshiung, Taiwan). All Balanodytes specimens collected in the present study have four pairs of terminal cirri and a pair of caudal appendages. The presence of caudal appendages was also observed in a very small juvenile specimen (CEL-Acro-RYU-03), suggesting that the caudal appendages are present during ontogenetic development. The definition of Balanodytes is, therefore, emended as “lithoglytid species having four pairs of terminal cirri and with a pair of caudal appendages (without pedestals)”.
Kolbasov (2000a) originally described Lithoglyptes balanodytes . Kolbasov & Newman (2005) placed this species in Armatoglyptes , herein treated as a junior synonym of Balanodytes . Newman (2011) treated B. balanodytes as a junior synonym of B. taiwanus , followed by Poore (2012). However, B. balanodytes differs from B. taiwanus in having recurved (hooked posteriorly) posterior projections of the opercular bars in the adult female (decurved – hooked anteriorly in B. taiwanus ) and by the absence of a distinct and armed orificial knob, whereas B. taiwanus has a number of curved teeth and setae on its distinct orificial knob. Therefore, we follow Kolbasov (2000a) and conclude that B. balanodytes is a different species from B. taiwanus . Tomlinson (1969) recorded B. taiwanus from the Marshall Islands, Fiji Sea (a fungi-damaged specimen) and described this species as having four pairs of terminal cirri but no caudal appendages. We have contacted the United States National Museum and confirmed Tomlinson had not deposited his material. We follow the suggestion in Kolbasov (2000a) that B. taiwanus identified by Tomlinson (1969) should be Lithoglyptes balanodytes Kolbasov, 2000 (now Balanodytes balanodytes ) and that the absence of caudal appendages in Tomlinson’s specimen might be a result of the specimen being damaged by fungi. Specimens described by Tomlinson (1963) as Kochlorine habei and later as Lithoglyptes habei (Tomlinson 1969) , and other subsequent descriptions of this species (Kolbasov 2009), correspond to A. taiwanus . Therefore, Armatoglyptes habei is treated as a junior synonym of Balanodytes taiwanus .
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.