Cambarus ( Jugicambarus ) pauleyi, Loughman, Zachary J., Thoma, Roger F., Fetzner Jr, James W. & Stocker, G. Whitney, 2015
|
publication ID |
https://doi.org/10.11646/zootaxa.3980.4.4 |
|
publication LSID |
lsid:zoobank.org:pub:ABC1557C-BA0C-4D96-BEBA-6B5C696F1814 |
|
DOI |
https://doi.org/10.5281/zenodo.5681281 |
|
persistent identifier |
https://treatment.plazi.org/id/A004D13C-1588-4713-9BE6-BDFB5E402CA2 |
|
taxon LSID |
lsid:zoobank.org:act:A004D13C-1588-4713-9BE6-BDFB5E402CA2 |
|
treatment provided by |
Plazi |
|
scientific name |
Cambarus ( Jugicambarus ) pauleyi |
| status |
sp. nov. |
Cambarus ( Jugicambarus) pauleyi View in CoL , new species Loughman, Thoma, Fetzner, and Stocker
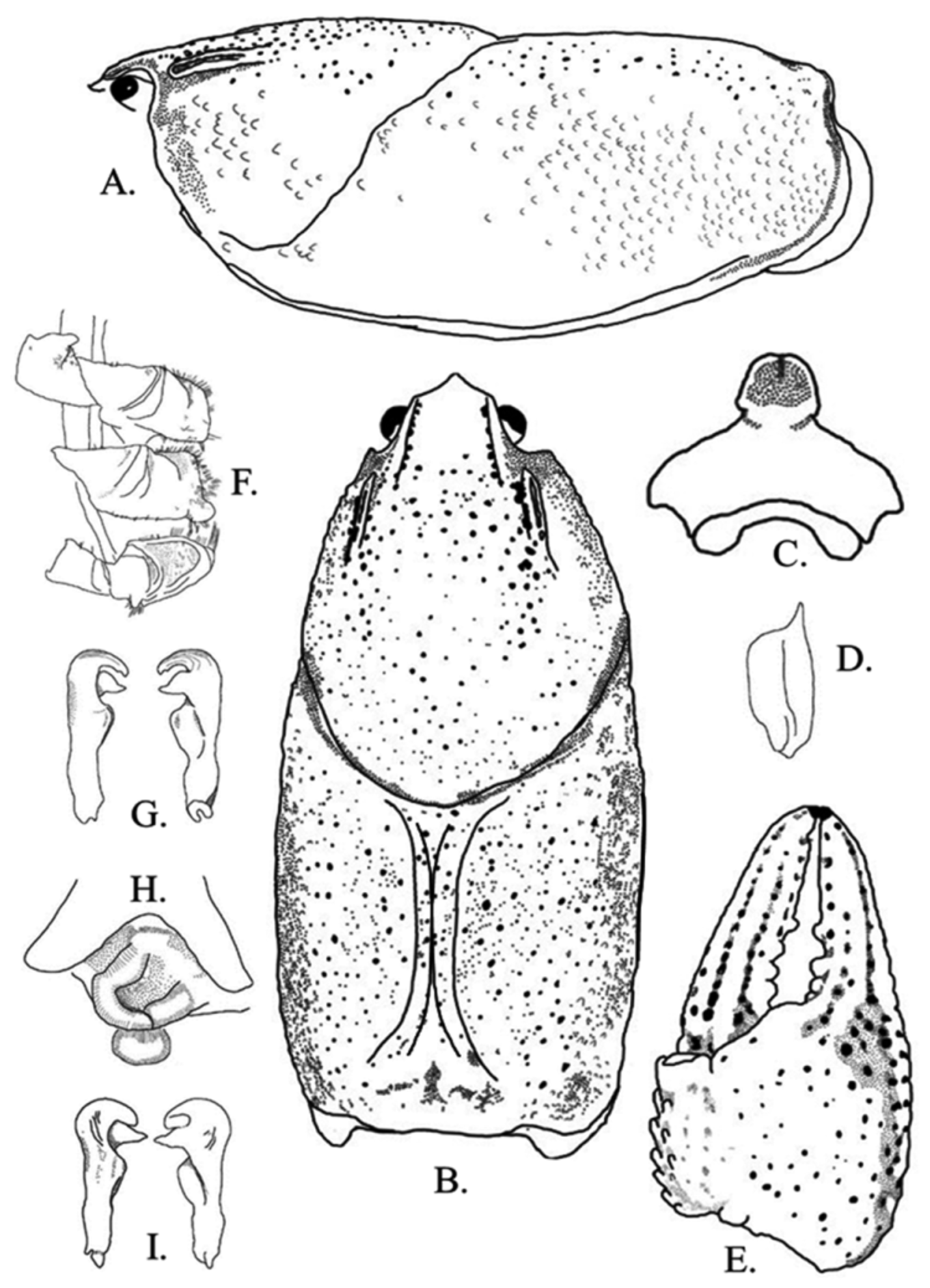
Figs. 5 View FIGURE 5 A–I, 6, 7; Table 1 View TABLE 1
Cambarus dubius Faxon, 1884:114 View in CoL [in part]. Faxon 1885:70, pl. 4: fig. 3; pl. 8: figs. 7, 7' [in part]; Dewees, 1972:1, figs. 1b–l, 2c–h, 3, 4, 5b–h, 6–16 [in part].
Cambarus carolinus dubius: Faxon, 1914:396 View in CoL , 425 [in part].
Cambarus (Jugicambarus) dubius: Hobbs and Bouchard, 1973:62 View in CoL [in part]; Hobbs, 1974:18, fig. 60 [in part]; Hobbs, 1989:22, fig. 78 [in part]; Jezerinac et al., 1995:121, fig. 61 [in part].
Diagnosis. Body and eyes pigmented. Posterior region of rostrum concave and deflected anteriorly. Rostral margins thickened, parallel to slightly converging at acumen. Rostrum excavated. Acumen distinctly triangular with prominent dorsally deflected spiniform tubercle at terminus. Areola obliterated. Cervical spines always absent. Mandibular, branchiostegal, and orbital regions of carapace with numerous well-developed tubercles. Postorbital ridges short; spiniform tubercle present in juveniles and subadults; adult postorbital ridge lacks tubercle. Suborbital angle acute to subacute. Carapace slightly vaulted in lateral profile. Antennal scale widest in middle, 1.3–3.6 ( x = 1.8, n = 35, SD = 0.5) times as long as wide. Total carapace length (TCL) 2.0–2.2 ( x = 2.1, n = 35, SD = 0.1) times longer than width. Form I and II males possessing hook on ischium of third pereiopods only; hook gently curved at apex, overarching basioischial joint in form I males, not reaching basioischial joint in form II males; hooks not opposed by tubercle on basis. Mesial surface of chelae with two rows of tubercles; mesial most row with 5–9 ( x = 6.7, n = 35, SD = 0.9) tubercles, second dorsal row with 2–7 ( x = 5.1, n = 35, SD = 1.1) tubercles. Tubercles do not extend from mesial surface of chela to body of palm. One or two subpalmar tubercles present at junction of dactyl with propodus. Longitudinal ridge of dactyl consisting of several well developed scattered tubercles. Dorsomedian ridge of fixed finger of propodus pronounced. Ventral margin of propodus costate. Chelae lack defined lateral impression at the junction of the fixed finger with the propodus base. Dactyl and fixed finger with sharp corneous subterminal tip forming spine. Form I male palm length 73.9–77.5% ( x = 75.7%, n = 3, SD = 3.5%) of palm width, form I male palm length 35.5–37.7% ( x = 36.6%, n = 3, SD = 1.6%) of total propodus length; female dactyl length 56.0–62.0% ( x = 58.6, n = 19, SD = 2.0%) of total propodus length. One or two large carpal spines present. First pleopod of form I male with short terminal elements. Central projection not tapering distally; recurved> 90º to main shaft of gonopod, with distinct subapical notch. Mesial process directed 90° to shaft, bent cephalolaterally; inflated cephalically, tapering to distinct caudal point beyond terminance of central projection; mesial process terminating in a split tip. Annulus ventralis immovable; distinctly asymmetrical posteriorly; cephalic portion with median trough leading to strongly sculptured central fossa; exaggerated “S” bend in sinus terminating at caudal edge. Fossa sclerotized.
Description of holotypic male, form I. ( Figs. 5 View FIGURE 5 A–G). Body somewhat compressed laterally ( Fig. 1 View FIGURE 1 A); carapace posterior to cervical groove slightly wider than abdomen. Carapace depth greater than carapace width at caudodorsal margin of cervical groove. Total carapace length 35.4 mm; post orbital carapace length 30.7 mm. Areola length 14.6 mm: areola 41.2% of TCL (47.6% of postorbital carapace length (PCL). Rostrum moderately excavated, more so proximally than distally; margins thickened, parallel and continuous to base of acumen where margins becomes subparallel; floor of rostrum with numerous punctations. Rostrum 1.4 times longer than wide. Acumen broad and triangular, ending in dorsally upturned corneous tip ( Fig. 5 View FIGURE 5 A). Postorbital ridges short and well developed, lacking cephalic tubercles or spines. Suborbital angle subacute, lacking tubercle ( Fig. 5 View FIGURE 5 B). Cervical spine absent. Mandibular, branchiostegal, and orbital regions of carapace punctated with well-developed tubercles; greatest tubercle density in hepatic region.
Abdomen supraequal in length to carapace, pleura rounded cephaloventrally, angled distoventrally. Lateral margin of terga angulate; lateral margin of second pleuron deeply furrowed. Cephalic section of telson with 2 large spines in each caudolateral corner. Proximal podomere of uropod with distal spine on mesial lobe; mesial ramus of uropod with median ridge ending distally in distomedian spine not overreaching margin of ramus; laterodistal spine pronounced. Distal margin of proximal segment of lateral ramus of right uropod having 13 immovable, small spines and 1 lateral, large, movable spine. Cephalomedian lobe of epistome subtriangular, zygoma moderately arched ( Fig. 5 View FIGURE 5 C); cephalolateral margins thickened, forming sharp angle at junction with endostyle ( Fig. 5 View FIGURE 5 C). Body of epistome possessing prominent cephalomedian fovea. Antennal scale broadest in middle; lateral margin thickened, terminating in large corneous spine; mesial margin setiferous ( Fig. 5 View FIGURE 5 D). Right antennal scale 4.9 mm long, 5.5 mm wide ( Fig. 5 View FIGURE 5 D). Tip of right antenna reaching posterior portion of carapace when adpressed.
Mesial surface of right chela with 2 rows of tubercles; mesial most row with 8 tubercles, second dorsal row with 5 tubercles ( Fig. 5 View FIGURE 5 E). Palm length 39.0 % of palm width; depth of palm 6.5 mm. Ventral surface of palm containing 2 subpalmar tubercles. Dorsal longitudinal ridge of dactyl developed and possessing moderate sized tubercles ( Fig. 5 View FIGURE 5 E); dactyl terminating in large corneous spine. Dorsomedian ridge of fixed finger of propodus pronounced. Propodus terminating in sharp, corneous tip. Lateral impression at junction of fixed figure and propodus absent. All measurements and counts from right chela.
Carpus with prominent dorsal furrow and 3 weak dorsomesial tubercles; remainder of surface with some setiferous punctations; mesial margin with 2 large, procurved spines at about midlength. Distodorsal surface of merus with 9 spiniform tubercles; ventrolateral ridge with 5 small spines and large, corneous distal spine; ventromesial ridge with 3 well-developed spines; ventrolateral margin of ischium with 2 small, spiniform tubercles. Hook on ischium of third pereiopods only ( Fig. 5 View FIGURE 5 F); hook gently curved at apex, overarching basioischial joint, not opposed by tubercle on basis. Form I gonopod as described in diagnosis ( Fig. 4 View FIGURE 4 G); tip reaching anterior margin of fourth caudomesial boss when abdomen is flexed.
Description of allotypic female. ( Fig. 4 View FIGURE 4 H, Table 1 View TABLE 1 ). Differing from holotype in the following respects; carapace height less than carapace width (12.6 and 13.1 mm, respectively, 96.2%); TCL 28.2 mm, PCL 24.9 mm. Areola 41.1% of TCL (46.6% of PCL). Posterior portion of rostrum more excavated than anterior portion; rostrum as long as wide. Abdomen length 25.0 mm. Mesial surface of chelae with 2 rows of tubercles; mesial most row with 7, and second dorsal row with 4. Palm length ( 6.7 mm) 75.2% of palm width ( 8.9 mm); depth of palm 5.6 mm. Antennal scale 4.0 mm long, 4.4 mm wide. All measurements and counts from right chela. Annulus ventralis as described in diagnosis ( Fig. 1 View FIGURE 1 H); width of postannular sclerite half total width of annulus ventralis; first pleopods uniramous, reaching central region of annulus ventralis when abdomen flexed.
Description of morphotypic male, form II. ( Fig. 4 View FIGURE 4 G, Table 1 View TABLE 1 ). Differing from holotype in the following respects: Carapace wider than abdomen (16.4 and 13.3 mm, respectively); carapace height less than carapace width (15.9 and 16.4 mm respectively, 97%); TCL 32.0 mm and PCL 28.6 mm. Areola length 40.0% of TCL (46.4% of PCL). Rostrum margins subparallel to base of acumen; rostrum 1.3 times as long as wide. Abdomen 30.5 mm long. Mesial row of tubercles on palm of chela with 7 tubercles; second dorsal row with 6. Palm length ( 7.8 mm) 98.7% of palm width ( 7.9 mm). Antennal scale 5.4 mm long, 4.9 mm wide. Gonopods reaching anterior margin of 4th pereiopod caudomesial boss. Central projection curved 90° to shaft, with complete apex; rounded ( Fig. 1 View FIGURE 1 C). Mesial process tapered, bulbous, directed caudolaterally. Hook on ischium of third pereiopod small, not reaching basioischial joint. All measurements and counts from right chela.
Size. Form I male (n = 3) TCL ranges in size from 32.5–35.4 mm (PCL 29.0– 30.7 mm) with a mean TCL of 33.6 mm. Form II male (n = 17) mean TCL is 31.9 mm and ranges in size from 28.0– 35.5 mm (PCL 24.3–31.4 mm). Non-ovigerous female (n =10) TCL mean is 30.3 mm and ranges from 28.2–40.2 mm (PCL 24.9–35.8 mm). Ovigerous female (n = 4) TCL ranges in size from 32.2–40.0 mm (PCL 28.2–35.8 mm) with a mean of 35.6 mm. The largest specimen examined was a nonovigerous female with TCL of 40.2 mm (PCL 35.8 mm).
Color. Carapace ground color deep blue to navy blue; posterior margin of carapace navy blue ( Fig. 6 View FIGURE 6. A ). Hepatic and antennal region of carapace punctuated with cream, light blue, or white tubercles. Postorbital ridge color blue, to navy blue. Rostrum margins and acumen maroon or red, rarely blue. Cephalic section of carapace immediately anterior to and including cervical groove blue to indigo; mandibular abductor scars ranging from indigo to blue. Lateral margin of antennal scale light blue to blue; body of antennal scale light blue to blue. Antennal flagellum and antennules light blue; dorsal surface of lamellae blue to light blue; ventral surface cream to light blue. Dorsal surface of chelae blue to navy blue with white highlights; mesial, second dorsal row, and dorsal surface of dactyl tubercles blue or navy blue. Denticles on opposable surfaces of fingers cream, white, or tan. Ventral surface of chelae white or cream. Dorsal surface of carpus blue, navy blue or light blue; occasionally light purple; region adjacent to and including furrow navy blue to blue; carpus spine cream. Merus indigo to blue, or light blue. Podomeres of pereiopods light blue, blue-green, or blue; joints of pereiopodomeres cream or white. Dorsal and dorsolateral surface of abdomen same color as carapace; anterior region of abdomen with slight indigo tint; tergal margins red to reddish brown. Uropods navy blue, blue, light blue, with purple tint; margins white to cream. Ventral surface of abdomen and carapace cream or white. Dorsal ridge of form I gonopod central projection amber; body of central projection, gonopod, and mesial process tan. Form II gonopod and all associated processes cream. Cephalic portion of annulus ventralis pink to pink-cream; ridge of fossa pink; caudal region of annulus ventralis ranges from pink to cream colored.
Type locality. Seepage stream adjacent to Moncove Lake State Park Playground, 2.0 km ( 1.3 mi) northwest of Roxalia, Monroe County, West Virginia ( 37.620745 N, - 83.352988 W). All animals were extracted from deep within their burrows, and did not rise to the surface during extraction efforts. Crayfish were collected from burrows in streambanks and seepage wetlands associated with a small headwater stream coursing through a mixed mesophyitc forest composed of American Beech ( Fagus grandifolia Ehrh. ), Red Oak ( Quercus rubra L.), Black Oak ( Quercus velutina Lam. ), and Tulip Poplar ( Liriodendron tulipifera L.). The holotype, allotype, morphotype, and paratypes were all excavated from burrows ranging from 0.6 to 1.3 meters deep in iron-rich clay-loam soil. Burrow architecture was simple, consisting of one or two surface tunnels unifying into a central tunnel, terminating in an enlarged resting chamber. The type series and paratypes were all collected on 28 June 2014 by Spencer Bell, Zachary Dillard, ZJL, Luke Sadecky, Nicole Sadecky, R. Katie Scott, Eric Tennant and Eric Tidmore.
Disposition of types. The holotype, allotype, and morphotype are deposited in the National Museum of Natural History ( USNM), Smithsonian Institution, Washington, D. C. (catalogue numbers USNM 1283506, 1283507, 1283508, respectively). Paratypes are deposited in the West Liberty University Astacology Collection, West Liberty, WV (WLU 2001).
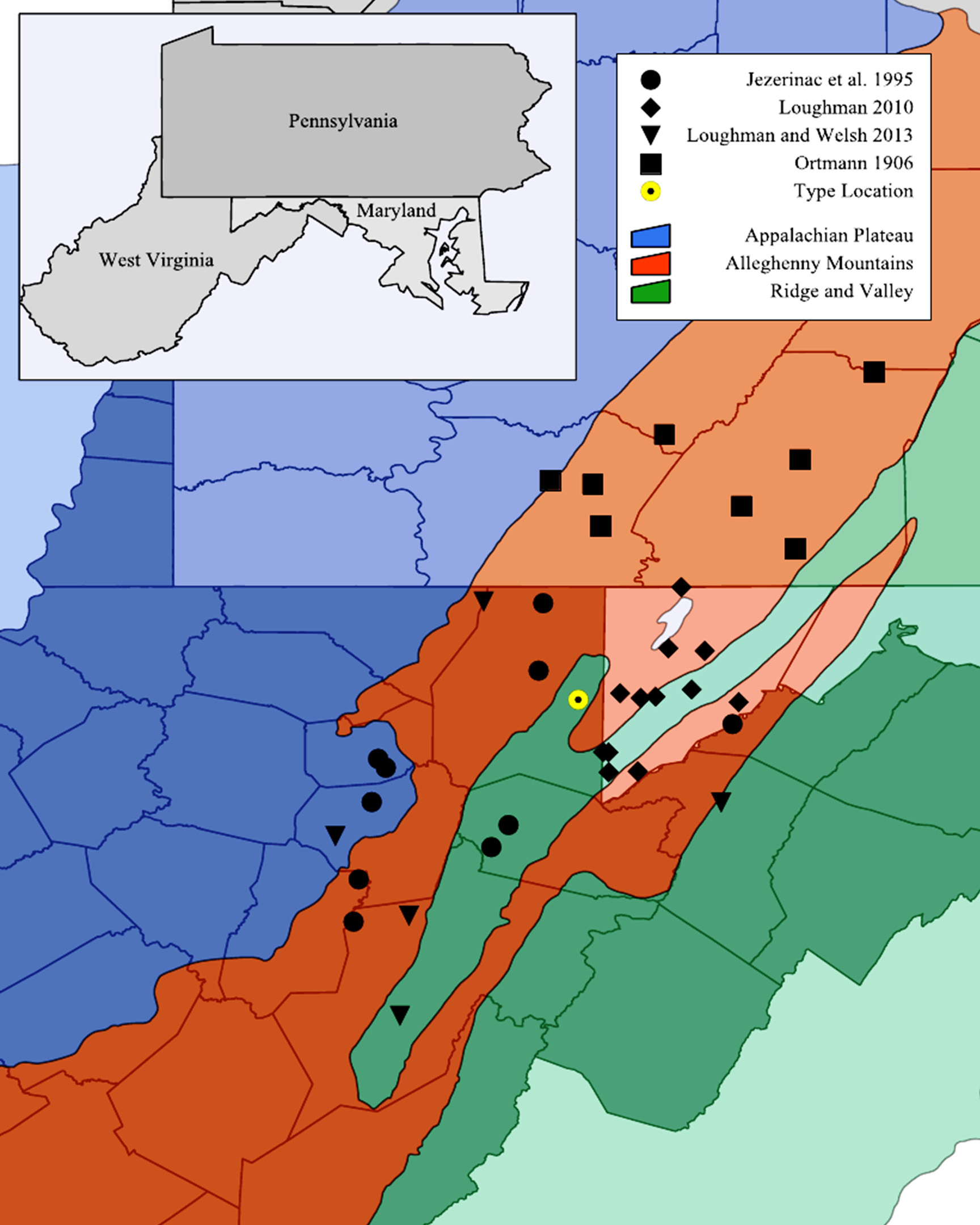
Range and specimens examined. Cambarus pauleyi is endemic to an 846 km 2/ 324 mi 2 section of the high elevation (> 762 m; 2,500 ft) Appalachian Plateau physiographic province in Greenbrier and Monroe counties, West Virginia and confined to the Meadow and Greenbrier River basins. The entirety of its range is close to the junction of the Appalachian Plateau, Allegheny Mountain, and Ridge and Valley physiographic provinces, with the eastern extent limited to the western front of the Ridge and Valley, and the northern extent limited to the southern foothills of the Allegheny Mountain provinces ( Fig. 7 View FIGURE 7 ). The distribution of Cambarus pauleyi also appears to be limited to elevations between 730 to 790 m ( 2,395–2,590 ft), with the majority of known extant occurrences in wetlands adjacent to Meadow River in Greenbrier County, West Virginia. Collecting efforts between the type locality and the apparent core of the C. pauleyi range only produced C. aff. dubius . Over 95% of historic wetlands in this region had been ditch-drained, and converted to agricultural fields. It is possible that populations persist in unsampled areas in this region, warranting future surveys for the species.
Due to its limited distribution, and potential conservation concerns associated with over collecting, specific locations will not be given following collection accession numbers, but are available from the primary author upon request. All West Liberty University collections (WLU) were made by ZJL and the 2014 West Liberty University Astacology Laboratory summer field crew, and are housed in the West Liberty University Astacology Collection, denoted with the prefix WLU. Additional specimens were examined from the Ohio State University Museum of Biological Diversity Crustacean Collection, denoted with the prefix OSU. MI = form I male, MII = form II male, F = female.
A total of 62 specimens were examined from 8 localities that compose the following 12 lots: WLU 1997, 26 June 2014, 6 MII, 3 F; WLU 1998, 25 June 2014, 3 MII, 1 F; WLU 1999, 27 June 2014, 1 MI; WLU 2000, 26 June 2014, 1 F ovig; WLU 2001, 28 June 2014, 6 MII, 2 F; WLU 2002, 26 June 2014, 1 F ovig, WLU 2003, 26 June 2014, 1 F ovig; WLU 2004, 27 June 2014, 2 MII, 2 F; WLU 2005, 26 June 2014, 1 MII; WLU 2006, 26 June 2014, 4 MII, 4 F; WLU 2007, 26 June 2014, 2 MII, 4 F; WLU 2008, 24 May 2008, 2 F, ZJL and Christopher G. Vopal; OSU 9183, 30 August 2013, 1 MI, 1 MII, 7 F, 7 Juv, R. F. Thoma and Mitchel E. Farley
Conservation status. Cambarus pauleyi should be listed as Endangered (E) according to the American Fisheries Society criteria ( Taylor et al. 2007), and assigned a G2 ranking according to the global conservation criteria ( Masters 1991) for conservation listing due to its limited range, and reduction of range. Cambarus pauleyi should be listed as near threatened (VU) using the International Union for the Conservation of Nature ( IUCN 2001) criteria due to its narrow distribution and reduction of its natural range.
Preferred habitats for Cambarus pauleyi appear to be upland wetlands, seepage stream banks, and wet meadows at the base of hillsides. These habitats are also prime agricultural sites in a state dominated by mountainous topography. Within the upper Meadow River Valley, and specifically within Meadow River Wildlife Management Area ( MRWMA), Putnam (1995) cited conversion of wetlands to agricultural sites as the number one threat to unique floristic communities in the area. Currently the MRWMA is the stronghold for the species, given the majority of wetlands and meadows that abut the MRWMA parcel have been converted to pasture and row crop agriculture ( Putnam 1995).
Given the rate of conversion of wetlands to farmland in the upper Meadow River Valley, and the lack of any specimens procured from this habitat by us, it is highly likely that C. pauleyi has experienced greater than 50% range reduction across its limited natural range in Greenbrier and Monroe County, West Virginia. West Virginia currently is experiencing a boom in natural gas extraction. Associated with this industry is a burgeoning network of natural gas pipelines, some of which have been proposed to run through the core range of C. pauleyi . The ultimate impact of this practice on C. pauleyi remains unknown.
Ecology and life history. Cambarus pauleyi should be classified as a primary burrower following Hobbs (1981) due to its utilization of burrows for all facets of its ecology and life history. No animals were observed in surface waters, though several colonies were in direct contact with such habitats (ZJL, personal observation). Seepage wetlands at slightly higher ( 10–15 m) elevations immediately adjacent to large Pin Oak/Maple swamps appear to be the preferred habitat for C. pauleyi . Forested seeps on hillsides and along ridgelines within the range of the species did not harbor any colonies of C. pauleyi , but we did find populations of an unnamed member of the C. aff. dubius complex, which we refer to as the black-body orange-claw ( BBOC) morph, in these habitats. In addition to seepage wetlands, C. pauleyi also occurs in low-gradient headwater seepage streams. In these environs, C. pauleyi burrows both in floodplains and occasionally in stream banks. Cambarus pauleyi does occur in anthropogenic habitats, although at much lower densities when compared to population levels in natural undisturbed situations (ZJL, personal observation). Anthropogenic habitats utilized by C. pauleyi included both residential yards and roadside ditches, with higher densities being found in the latter. In disturbed situations, C. pauleyi was subordinate to C. thomai , the latter being more prevalent in wetlands associated with such habitat.
Compared to other members of the C. aff. dubius complex, C. pauleyi burrow morphology is simplistic. Burrows maintain one or two entrances leading to vertical tunnels, which are frequently capped, plugged or possess well-constructed chimneys. Burrows with two openings maintained bifurcate tunnels that unify into single central tunnels after 25–30 cm. Central tunnels continue an additional 30–50 cm vertically, ultimately ending in simplistic resting chambers. Ancillary tunnels running laterally and along resting chamber floors are rarely encountered in this species (ZJL, personal observation). Burrows of syntopic C. thomai often maintain several ancillary tunnels extending from both the central tunnel and resting chambers of the burrow (ZJL, personal observation). Burrow morphology for the BBOC morph differs from C. pauleyi in the orientation of the initial burrow tunnel, which runs laterally into hillsides for 10–30 cm before adopting a more vertical orientation (ZJL and RFT, personal observation).
Surface activity in this species was not relegated solely to periods of darkness. On three occasions, C. pauleyi was observed positioned within 5 cm of burrow entrances at midafternoon; two of these observation occurred during periods of bright sunshine. Though observed during periods of daylight, C. pauleyi surface activity reaches its highest levels during the first three hours following twilight (ZJL, personal observation). Fifty plus nocturnal observations were made of this species resting at the entrance to its burrow (ZJL, personal observation). Observed surface behaviors included plugging burrows, maintaining chimneys, grooming, and foraging. Foraging behavior included positioning of antennae at both 45° and 135°, with chelae slightly elevated and agape. Animals encountered in this position frequently lunged at any object that made contact with their primary antennae (ZJL, personal observation). After a brief period following grasping of an object, objects deemed edible were drug down into the confines of the burrow. Defensive behaviors were also observed for C. pauleyi resting at burrow portals. Animals engaged at the burrow entrance that were not interested in food would most frequently flee down their burrows, but occasionally would position their antennae over their carapace, at the same time gaping their chelae. Once this posture was adopted, animals deliberately moved towards stimuli, slashing their agape chelae at the perceived threat. After two or three bouts of this behavior, most animals would quickly fall down their burrows by pulling their pereiopods tightly against their bodies. Individuals that displayed this behavior were not observed at burrow openings for the remaining period of a collection effort (ZJL, personal observation).
Cambarus pauleyi View in CoL life history is poorly understood and in need of further research. Based on specimens captured with this effort, as well as Jezerinac et al. (1995), males appear to molt from form II to form I during August through September, and maintain a form I condition from fall through mid-May to early June, when they molt from form I back to form II. This is evidenced by only one form I male (5.5 %) collected in June 2014, compared to 17 form II males (94.5%) from across 6 sites. Carapaces were immaculate for several males captured during this time indicating recent molting activity (ZJL, personal observation). Jezerinac et al. (1995) collected form I males on 19 August 1989, further supporting this contention. The holotype molted from form II to form I on 17 September 2014, adding support. Glared females were observed in August, September and October (ZJL, personal observation). Glare glands of two females maintained in captivity steadily developed beginning in August, and reached full development by early October (ZJL, personal observation). It is theorized that egg deposition in this species occurs April through June. A single ovigerous female with 31 eggs was excavated from a burrow on 26 June 2014. Two females maintaining instars excavated from burrows on 26 and 28 June 2014; average instar compliment for the females was 26.0 instars (SE = 5.67 instars). Both females were collected when chimneys were removed from burrows. In both instances females were resting inside the chimney, and did not attempt to flee once observed (ZJL, personal observation). The third female responded aggressively to plunging of her burrow, rising from the confines of the resting chamber with chelae agape. In several instances, juveniles were encountered inside burrows alongside females, but never alongside males. This behavior has been observed for several primary burrowing crayfish (Richardson 2007; Loughman 2010b) including nominate C. dubius View in CoL ( Dewees 1972; Loughman 2010b).
Crayfish associates. Cambarus (J.) pauleyi View in CoL has been collected in areas also harboring Cambarus ( C.) carinirostris Hay, 1914 View in CoL ; Cambarus ( T.) thomai Jezerinac, 1993 View in CoL ; and Procambarus View in CoL ( O.) a. acutus Fowler, 1912.
Variation. Very little morphological variation was observed between sites and life stages, with the only noticeable variation being juvenile carapaces that are less laterally inflated than adults. Tubercle shape and degree of supination did not display ontogenic differences in this species; both juvenile and adult C. pauleyi lack postorbital, cervical, and hepatic spines.
Relationships and comparisons. Cambarus pauleyi is placed in the subgenus Jugicambarus based on the presence of cristiform tubercles on the mesial margin of the chelae ( Hobbs 1969). Among described members of the subgenus, C. pauleyi is most similar to nominate C. dubius and several undescribed members of the C. dubius complex. Color in life, differences in palm depth/palm length and rostral width/rostral length ratios, as well as morphological differences in the chelae and carapace anatomy can all be used to distinguish C. pauleyi from C. dubius s.s., as well as undescribed members of the complex. The dorsal carapace, chelae, and abdomen of C. pauleyi all display various shades of blue in living specimens, whereas C. dubius s.s. is predominately composed of oranges and orange-reds. Cambarus pauleyi palm depth/palm length ( x = 79.8%; range 74.7–84.9%) is larger than C. dubius s.s. ( x = 71.0%, range 64.0–77%). Cambarus pauleyi rostral width/rostral length ratios ( x = 66.7%; range 60.9–73.8%) are smaller than C. dubius s.s. (rostral length/rostral width ( x = 73.5%; range 67.3–79.6%).
The number of subpalmar tubercles is an important character that separates C. pauleyi from both typical and undescribed members of the complex. Cambarus pauleyi always has at least 1, and sometimes as many as three ( x = 1.2) subpalmar tubercles; all other populations of C. aff. dubius lack subpalmar tuberlces. This character is particularly important when dealing with preserved specimens once they have been bleached following preservation (ZJL, personal observation). In addition to the subpalmar tubercles, the primary carpal spines frequently (82% of specimens examined) are bifurcated into two large spines in C. pauleyi . To date, no other C. dubius population analyzed in West Virginia, or elsewhere by the authors, possess either of these characters states. Finally, the C. pauleyi carapace is more dorsally flattened and less vaulted compared to the laterally compressed, vaulted carapace of C. dubius .
The BBOC morph is the closest geographic member of the C. aff. dubius complex to C. pauleyi , with populations occurring within 1.0 km of a site harboring C. pauleyi (ZJL, unpublished data). Cambarus pauleyi can be differentiated from the BBOC morph by overall coloration, palm depth/palm length ratio, and rostral and carapace morphology. The dorsal coloration of the carapace and abdomen of the BBOC morph ranges from black to brown, with lateral carapace colors ranging from orange to red, which differs from the blue carapace of C. pauleyi . Dorsal surfaces of the palm in the BBOC morph are composed primarily of black/browns, with both the dactyl and propodus primarily consisting of oranges and reds. Cambarus pauleyi chelae coloration always consists of blues. Unlike comparison with C. dubius s.s., the palm length/palm depth ratio in C. pauleyi is smaller ( x = 79.8%; range 74.7–84.9%) than the BBOC morph ( x = 83.1%; range 80.7–88.0%, n = 22) ratio. Morphologically, both the BBOC morph and C. dubius s.s. lack subpalmar tubercles and possess a more dorsolaterally compressed carapace compared to C. pauleyi , the latter of which always maintains subpalmar tubercles as well as a dorsolventrally compressed carapace. The rostrum of Cambarus pauleyi is moderately excavated, while the BBOC morph possesses a deeply excavated rostrum.
The dorsal and lateral carapace and abdominal coloration in Cambarus monongalensis Ortmann, 1905a is similar to C. pauleyi , consisting of several shades of blue. The latter can be distinguished from C. pauleyi by chelae and rostral morphology. The margins of the propodus in Cambaurs pauleyi are costate compared to the smooth margins of C. monongalensis . Cambarus pauleyi also has two rows of tubercles on the mesial margin of the chelae, compared to a single row in C. monongalensis . Rostral anatomy can also be used to differentiate these species; with C. pauleyi having a more elongate rostrum while in C. monongalensis the rostrum is truncated, and short.
Furthermore, the two species can be separated in the field by the location of collection, given that the ranges of C. pauleyi and C. monongalensis do not overlap. Cambarus pauleyi is relegated to the high elevation Appalachian Plateau in Greenbrier and Monroe County, West Virginia. Cambarus monongalensis has a range that consists of two disjunct populations, one in the Allegheny Mountains of West Virginia and Virginia, and another present in the low elevation Appalachian Plateau of West Virginia and Pennsylvania ( Ortmann 1905b; Jezerinac et al. 1995; Loughman and Welsh 2013).
Etymology. It is with great pleasure, specifically from the primary author, that this crayfish be named in honor of Dr. Thomas K. Pauley, Professor Emeritus, Marshall University. As ZJL’s graduate advisor, you let his little “crayfish distraction” away from the herpetological focus of his thesis occur without objection, thus changing his professional focus, and academic life forever. Furthermore, it is also fitting that C. pauleyi bare Professor Pauley’s name, given C. pauleyi burrows provide refuge and safety for countless reptiles and amphibians that share its range, much like Pauley’s research efforts to conserve West Virginia’s reptiles and amphibians have undoubtedly aided in the conservation of reptiles and amphibians statewide.
Common name. Meadow River Mudbug.
TABLE 1. Measurements (mm) of Cambarus pauleyi, n. sp.
| Carapace | Holotype | Allotype | Morphotype |
|---|---|---|---|
| Total carapace length | 35.4 | 28.2 | 32.0 |
| Postorbital length Length cephalic section | 30.7 20.7 | 24.9 16.4 | 28.6 18.7 |
| Width | 14.5 | 13.1 | 16.4 |
| Depth | 16.5 | 12.6 | 15.9 |
| Length rostrum Length acumen | 6.4 2.3 | 5.5 1.9 | 6.0 1.8 |
| Length areola | 14.6 | 11.6 | 12.8 |
| Width areola | 0 | 0 | 0 |
| Antennal scale | |||
| Width | 5.5 | 4.4 | 4.9 |
| Abdomen | |||
| Width | 12.1 | 10.8 | 13.3 |
| Cheliped Length mesial margin palm | 8.4 | 6.7 | 7.8 |
| Width palm | 3.2 | 8.9 | 7.9 |
| Depth palm Length dactyl | 6.5 14.3 | 5.6 9.4 | 6.4 11.5 |
| Length carpus | 11.1 | 7.3 | 9.7 |
| Width carpus | 6.6 | 5.7 | 6.7 |
| Length dorsal margin merus Depth merus | 14.3 6.8 | 11.2 5.8 | 14.3 6.7 |
| Gonopod length | 8.2 | N.A. | 5.8 |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Cambarus ( Jugicambarus ) pauleyi
| Loughman, Zachary J., Thoma, Roger F., Fetzner Jr, James W. & Stocker, G. Whitney 2015 |
Cambarus (Jugicambarus) dubius:
| Hobbs 1989: 22 |
| Hobbs 1974: 18 |
| Hobbs 1973: 62 |
Cambarus carolinus dubius :
| Faxon 1914: 396 |
Cambarus dubius
| Dewees 1972: 1 |
| Faxon 1885: 70 |
| Faxon 1884: 114 |