Acestrocephalus sardina ( Fowler, 1913 )
|
publication ID |
https://doi.org/10.1590/S1679-62252006000400002 |
|
DOI |
https://doi.org/10.5281/zenodo.17095410 |
|
persistent identifier |
https://treatment.plazi.org/id/03B55E5E-2676-5A47-8C1D-F9CA4A9FFA61 |
|
treatment provided by |
Carolina |
|
scientific name |
Acestrocephalus sardina ( Fowler, 1913 ) |
| status |
|
Acestrocephalus sardina ( Fowler, 1913) View in CoL
Charax sardina Fowler, 1913: 566 View in CoL , fig. 2 ( type locality: Madeira River, about 200 miles east of W. long. 60º20’W, Brazil); Böhlke, 1984: 32 (catalog); Eschmeyer, 1998: 1507 (catalog).
Cyrtocharax sardina ; Fowler, 1950: 312 (listed).
Cynopotamus ( Acestrocephalus) sardina View in CoL ; Géry, 1972: 28 (diagnosis in key).
Acestrocephalus sardina View in CoL ; Menezes, 1976: 39 (in part, only specimens from rioAripuanã, MZUSP 10423); Eschmeyer, 1998: 1507 (catalog); Lucena & Menezes, 2003: 200 (in check list; maximum length, distribution).
Acestrocephalus ginesi Lasso & Taphorn, 2000: 443 View in CoL (type locality: Venezuela, Apure State, Río Cinaruco, playa en frente de Laguna Larga, Distrito Muñoz GoogleMaps ( 06º33’45"N 67º24’W); holotype not seen; topotypes examined); Lucena & Menezes, 2003: 200 (in check list; maximum length; distribution); Lasso, Lew, Taphorn, Nascimiento, Lasso-Alcalá & Machado-Allison, 2004: 148 (listed).
Acestrocephalus cf. ginesi View in CoL ; Lima et al., 2005: 166 (description; habitat; feeding habits; fisheries).
Material examined. ANSP 39307 About ANSP , holotype, 31.4 mm SL, (data taken by H.A. Britski; digital image and radiographs examined), Brazil, Mato Grosso: rio Madeira system, by Edgar A. Smith, 1912 ; MZUSP 10423 View Materials , 1 View Materials , 89 mm SL, Aripuanã: rio Aripuanã , 9º34’S 59º25’W GoogleMaps ; MZUSP 60396 View Materials , 1 View Materials , 94 mm SL, rio Aripuanã, Porto de Balsa on the road between Colmiza and Panela, 10º09’S 67º27’W GoogleMaps . MZUSP 29237 View Materials , 11 View Materials , 49-65 mm SL, Amazonas: ilha de Tamaquaré, rio Negro ; MZUSP 29239 View Materials and 29241, 36, 50-71 mm SL, Marauiá: rio Negro ; MZUSP 29240 View Materials , 5 View Materials , 51-66 mm SL, Massarabi: rio Negro ; MZUSP 31140 View Materials , 1 View Materials , 84 mm SL, Cachoeira de São Gabriel: rio Negro ; MZUSP 29238 View Materials and 31036, 6, 47.5-86 mm SL, Paraná do Jacaré: rio Negro . MZUSP 81209 View Materials and 81259, 4, 81-94 mm SL, rio Tiquié, indian village of Cururu , 0º16’N 69º54’W GoogleMaps . MZUSP 56558 View Materials , 2 View Materials , 62 View Materials and 65 mm SL, rio Jutaí , 2º57’S 67º0’W GoogleMaps ; MZUSP 35498 View Materials , 3 View Materials , 52-69 mm SL, Venezuela, Apure: Río Orinoco system, 1º55’N 67º04’W GoogleMaps . MZUSP 35499 View Materials , 5 View Materials , 53.7-82.6 mm SL, Rio Negro: Río Negro, near San Carlos do Rio Negro .
Diagnosis. Acestrocephalus sardina is most similar to A. nigrifasciatus , both having identical color patterns consisting of a triangular dark blotch on caudal-fin base, anteriorly continuous with a longitudinal dark stripe extending along lateral line ( Figs. 3 View Fig and 12 View Fig ) not found in any other species of the genus. In addition, both have the eye diameter greater than their congeners ( Fig. 4 View Fig ). Acestrocephalus sardina can be distinguished from A. nigrifasciatus by having fewer horizontal scale rows above lateral line (10-12 vs 13-14) and below (10-12 vs 12-13). See Tables 1 View Table 1 and 2 View Table 2 . Acestrocephalus nigrifasciatus also has the muscular hiatus over the anterior part of the swim bladder which is longer than and narrower than A. sardina and the fibers of the obliquus inferioris muscle, visible in the latter are entirely covered by the obliquus superioris muscle in the former (compare Figs. 5a and 5e View Fig ).
Description. Morphometrics of holotype and additional specimens presented in Table 3 View Table 3 . Meristic and morphometric data based on all lots for this species because no statistical differences found among population samples studied, in spite of wide distribution of species. Body elongate, relatively large (SL= 31.4-90 mm), compressed and not very deep; greatest body depth between occiput and dorsal-fin origin near middle of pelvic-fin length. Dorsal body profile nearly straight, but inclined ventrally from snout tip to base of supraoccipital spine, slightly convex between that point and dorsal-fin origin, nearly straight along dorsal-fin base, and straight or nearly so from base of dorsal-fin termination to caudal peduncle. Ventral body profile convex from tip of lower jaw to anal-fin origin, nearly straight and dorsally inclined along anal-fin base and slightly concave from base of posterior most analfin ray to origin of procurrent caudal-fin rays. Snout pointed. Lower jaw included in upper jaw when mouth is fully closed. Mouth angled posteroventrally from anterior tip of snout to posterior part of mandibular joint. Maxilla extending slightly beyond vertical line passing through posterior border of orbit.
Dorsal-fin rays ii, 9 in all specimens, n=76, including holotype. Posterior most ray unbranched, n=76. Adipose fin present. Anal-fin rays iv,32 (iv or v, usually iv unbranched, branched rays mean=30.3, range 28-33, n=76, posterior ray split to its base and counted as 1). Moderately developed anterior anal-fin lobe including anterior unbranched rays and first 7-8 branched rays. No hooks on anterior rays of males, but very few sexually mature males represented in examined sample. Pectoral-fin rays i,13 (anterior unbranched ray i, n=76) branched rays mean=13.6, range 12-15, n=76. Posterior tips of longest pectoral-fin rays reaching slightly beyond pelvic-fin origin. Pelvic-fin rays i,7, n=76. No hooks on pelvic-fin rays of sexually mature males. Distal tips of longest pelvic-fin rays extending to or slightly beyond anterior border of anus. Principal caudal-fin ray count 10/ 9 in all specimens, n=76.
Lateral line complete, perforated scales 73 (mean=71.3, range 67-74, n=76). Scale rows above lateral line 12 (mean=11.3, range 10-12, n=76). Scale rows below lateral line 10 (mean=10.3, range 10-12, n=76). Scale rows around caudal peduncle 20 (mean=20.7, range 20-22, n=76).
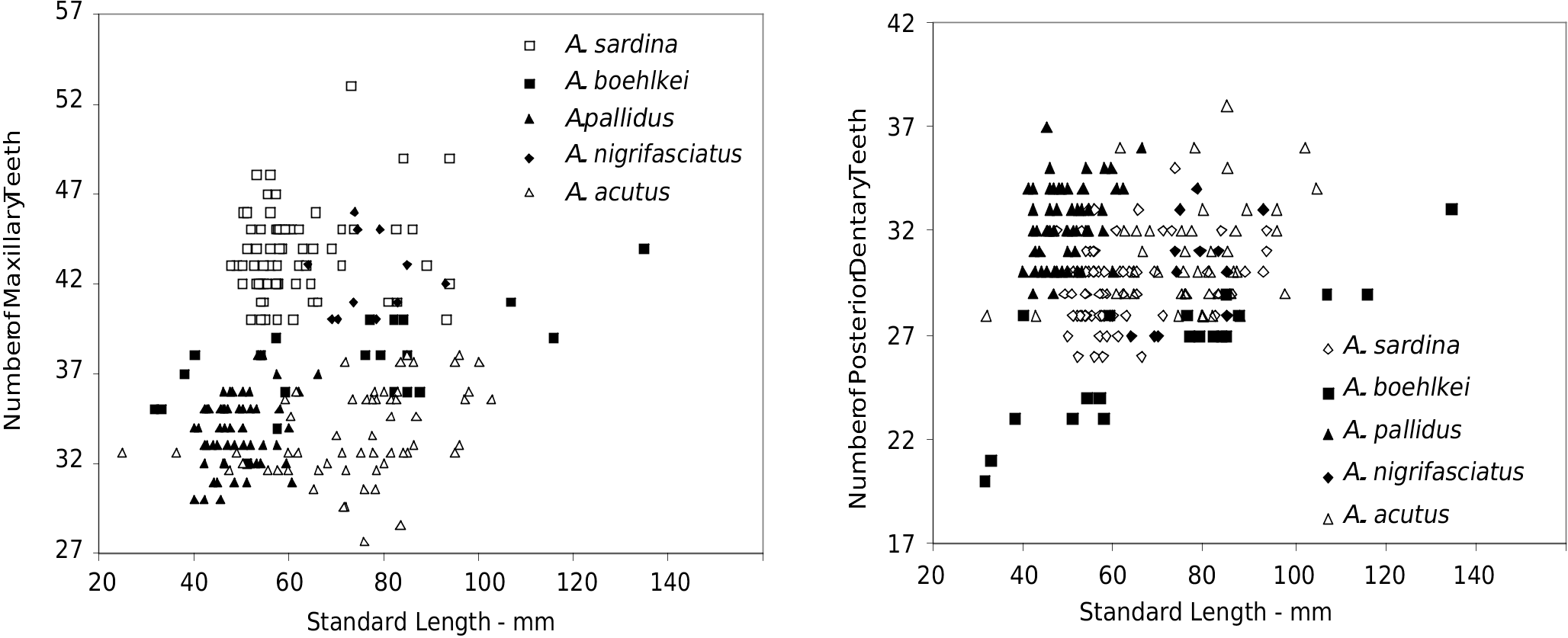
Premaxilla with two slightly enlarged inner conical teeth, an outer row with one anterior canine followed by row of small conical teeth and one posterior canine. Anterior canine outside anterior border of lower jaw when mouth closed. Outer row small conical teeth 8 (mean=7.7, range 7-9, n=75). Maxillary teeth 44 (mean=43.6, range 40-53, n=75). Maxillary teeth conical with tendency to increase in number according to increase in standard length ( Fig. 6 View Fig ). Dentary with outer row of conical teeth of which four anterior most spaced and first and third larger than third and fourth, these followed by posterior row of close-set smaller conical teeth that show tendency to increase in number with increase in standard length ( Fig. 7 View Fig ). Posterior dentary teeth 28 (mean=29.4, range 26-33, n=75). Teeth in inner row on dentary conical, very small, slightly inclined proximally to four anterior spaced larger conical teeth of outer row. Inner row dentary teeth 9 (mean=9.8, range 7-12, n=75).
Vertebrae 38 (mean=39, range 38-40, n=35). Total number of gill-rakers on first gill-arch 7 (mean=7, range 6-8, n=76).
Muscular hiatus of pseudotympanum ( Fig. 5a View Fig ) relatively large, dorsally limited by lateralis superficialis muscle, posteriorly by large exposed portion of second pleural rib, anteriorly by small exposed portion of first pleural rib, anteroventrally by large portion of the obliquus superioris and posteroventrally by small portion of obliquus inferioris muscle. Small opening anterior to first pleural rib visible.
Color in alcohol. Body pale to dark yellow, slightly darker dorsally than ventrally due to presence of dark chromatophores. Dark chromatophores on dorsal and lateral parts of trunk mostly concentrated along free border of scales. Dorsal part of head from tip of snout to supraoccipital region darker than remaining dorsal surface of body. Premaxilla, median part of maxilla and lower jaw densely pigmented with dark chromatophores. Scattered dark chromatophores on infraorbital and opercular bones. Lateral dark body stripe extending from upper opercle to caudal peduncle where fused with triangular dark blotch on caudal base, dark color extending to base of principal caudal rays. Lateral body stripe narrower anteriorly and posteriorly and sometimes obscured by guanine. Anterior part of lower jaw bordered with dark pigmentation. Mental area of lower jaw with diffuse dark blotch in some specimens. Dorsal-fin origin with black dot extending to base of first unbranched dorsal-fin ray. Pectoral, pelvic, dorsal, and caudal fins pale, with few scattered dark chromatophores.
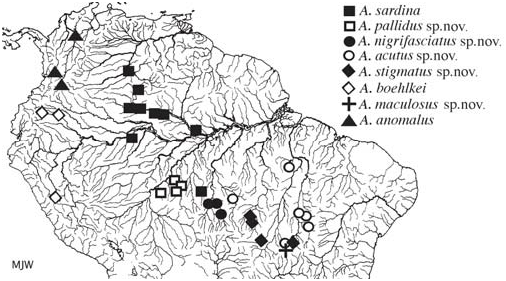
Distribution. This species is known from tributaries of the rio Madeira, Mato Grosso, rio Jutaí and rio Negro, Amazonas, Brazil and of the Orinoco Basin, Venezuela ( Fig. 8 View Fig ).
Remarks. Menezes’ redescription of A. sardina (1976) was based on the examination of two specimens, one of them from rio das Mortes near Xavantina, Mato Grosso. It was mentioned that no differences had been found among them and a small dark blotch at the humeral region was described for the species. Actually, this spot is present only in the specimen from rio das Mortes considered to belong to A. stigmatus herein described. A dark humeral spot is never present in specimens of A. sardina . At that time it was predicted that the taxonomic status of A. sardina would be cleared up only after a study based on a large series of specimens.
Acestrocephalus ginesi Lasso & Taphorn is herein considered a junior synonym of A. sardina . Five paraypes from Río Negro, near San Carlos de Río Negro, Venezuela (MZUSP 53499) and three specimens collected in the Apure State, near the type locality were studied and proved to be morphologically indistinguishable from specimens of the population samples representing A. sardina .
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Acestrocephalus sardina ( Fowler, 1913 )
| Menezes, Naércio A. 2006 |
Acestrocephalus cf. ginesi
| Lima, F & Ramos, T & Barreto, A & Cabalzar, G & Tenorio, A & Barbosa, F & Tenorio, A 2005: 166 |
Acestrocephalus sardina
| Eschmeyer, W 1998: 1507 |
| Menezes, N 1976: 39 |
Cynopotamus ( Acestrocephalus ) sardina
| Gery, J 1972: 28 |
Cyrtocharax sardina
| Fowler, H 1950: 312 |
Charax sardina
| Eschmeyer, W 1998: 1507 |
| Bohlke, J 1984: 32 |
| Fowler, H 1913: 566 |