Domitius culsu Ballarin, 2020
|
publication ID |
https://doi.org/10.4311/2019lsc0103 |
|
DOI |
https://doi.org/10.5281/zenodo.4323816 |
|
persistent identifier |
https://treatment.plazi.org/id/03BCA012-1E63-FFC6-FF2D-624FC584FD33 |
|
treatment provided by |
Plazi |
|
scientific name |
Domitius culsu Ballarin |
| status |
sp. nov. |
Domitius culsu Ballarin View in CoL sp. nov.
Figures 1 View Figure 1 A‒G, 2A‒E, 3A‒D.
Nesticus speluncarum Brignoli, 1979: 214 View in CoL (misidentification)
Type material. Holotype ♂. ITALY, Toscana: Garfagnana area, Lucca Province , Coreglia Antelminelli Municipality , Tana delle Fate di Coreglia Antelminelli cave, 141/T/LU , 260m a.s.l., 44.046336°N 10.523525°E, 21.VIII.2018, leg. F. Ballarin and D. Avesani ( MSNV). GoogleMaps Paratypes. Same locality as the holotype, 1♀, 04.IX.1967, leg. A. Vigna Taglianti ( MSNV) ( Brignoli 1979, sub Nesticus speluncarum ); GoogleMaps 1♀, 15.VIII.2015, leg. F. Ballarin and M. Gaiga ( MSNV) GoogleMaps ; 4♀♀, 24.VI.2017, ( 3♀♀ collected as juveniles and raised in captivity, adults: 20.VIII.2017, 15.IX.2017 and 28.VI.2018 respec- tively), leg. F. Ballarin and R. Ballarin ( MSNV); 4♀♀, 21.VIII.2018, leg. F. Ballarin and D. Avesani ( MSNV, MSNB) .
Examined comparative material. Domitius speluncarum (Pavesi, 1873) : ITALY: Liguria: 1♂, 1♀, (topotypes), La Spezia Province, Grotta Bocca Lupara cave, 74/Li/SP, 120m a.s.l., 05.III.1969, leg. P.M. Brignoli (MSNV) ( Brignoli, 1971); Toscana: 1♀, Lucca Province, Garfagnana area, Villa Collemandina Municipality, Canigiano village, Tana di Magnano cave, 162/T/LU, 653m a.s.l., 44.177285°N, 10.38803°E, 03.XI.1967, leg. A. Vigna Taglianti (MSNV) ( Brignoli, 1971), 1♀, 01.VIII.1975, leg. P. Magrini (MSNV), 4♀♀, 14.VIII.2015, leg. F. Ballarin and M. Gaiga (MSNV); 1♀, Forno- volasco (not reported in the label but very likely from Grotta del Vento cave), 700m a.s.l., 16.VI.1970, leg. O. Osella (MSNV) ( Brignoli, 1971); 1♀, Grotta del Buggine cave, 166/T/Lu, 315m a.s.l., 07.X.1967, leg. G. Castellini (MSNV) ( Brignoli, 1971); 1♀, Massa-Carrara, Buca della Freddana cave, 230/T/Ms, 550m a.s.l., 05.VI.1977, leg. C. Bonzano (MSNV) ( Brignoli, 1985); 1♀, Buca del Bacile cave, 226/T/Ms, 10.III.1975, leg. unknown (MSNV) ( Brignoli, 1985).
Domitius menozzii (di Caporiacco, 1934) : ITALY, Liguria, 1♂, 4♀♀, north-east of Genova town, Tanna da Vulpe cave, 264/Li/GE, 23.XI.1969, leg. A. Vigna Taglianti (MSNV) ( Brignoli, 1971); 2♂♂, 2♀♀, Creto, Tanna de Fate cave, 17/ Li/GE, 30.X.1971, leg. G. Gardini (MSNV); 2♀♀ (topotypes), Prati di Bavari locality, Tanna da Suja cave, 5/Li/GE, 582m a.s.l., 44.422894°N, 09.035239°E, 30.V.2013, leg. F. Ballarin, A. Trotta, G. Gardini, and S. Zoia.
Domitius sbordonii ( Brignoli, 1979) : ITALY, Lazio, 1♂ (holotype), Frosinone Province, Supino, Valle Serena, Grot- ta della Croce cave, 01.IX.1977 leg. V. Sbordoni (MSNV) ( Brignoli, 1979); 1♀ (paratype), 08.II.1976, leg. V. Sbordoni (MSNV) ( Brignoli, 1979).
Kryptonesticus eremita (Simon, 1880) : ITALY, Liguria: 2♂♂ (sub. Nesticus menozzii ), Creto, Tanna de Fate cave, 17/Li/GE, 30.X.1971, leg. G. Gardini (MSNV); Emilia-Romagna: 2♀♀ Ravenna Province, Riolo Terme, Borgo Rivola, Grotta del Re Tiberio cave, 36/Er/RE. 19.II.1951, Leg. Denis (MSNV) ( Zangheri, 1966, sub. Nesticus speluncarum ); Toscana: 1♀ Garfagnana area, Lucca Province, Coreglia Antelminelli Municipality, Tana delle Fate di Coreglia Antel- minelli cave, 141/T/LU, 260m a.s.l., 44.046336°N, 10.523525°E, 15.VIII.2015, leg. F. Ballarin and M. Gaiga (MSNV); 3♀♀, 24.VI.2017, Leg. F. Ballarin and R. Ballarin (MSNV-MSNVRAr/m 0007); 1♀, 21.VIII.2018, leg. F. Ballarin and D. Avesani (MSNV); Campania: 1♀, Avellino Province, Bagnoli Irpino, Mt. Piacentini, Grotta Giovannino cave, 16.VI.1956, leg. S.Ruffo (MSNV) ( Kritscher, 1958, sub. Nesticus speluncarum ).
Etymology
The name of the new species is derived from the Etruscan goddess Culsu who, according to the Etruscan mytholo- gy, ruled the cave-like entrance of the underworld. Noun in apposition.
Diagnosis
Species closely re- lated to D. speluncarum and D. menozzii. Males of D. culsu sp. nov. can be separated from males of all other Italian species of the genus Domitius by the different shape of the apophyses of the para- cymbium (Figs. 1A‒C, 2B,C vs. Fig. 4A‒I). D. culsu sp. nov shows a robust, S-shaped dorsal apophysis 1 in contrast with a large, flat and axe- like Da 1 in D. speluncar- um (Figs. 1A‒C, 2B, C vs. Fig. 4A‒C); a short and stumpy Da 1 in D. me- nozzii (Figs. 1A‒C, 2B, C vs. Fig. 4D‒F) or a long and thread-like Da 1 in D. sbordonii (Figs. 1A‒C, 2B, C vs. Fig. 4G‒I). Addi- tionally, males of D. culsu sp. nov. have a well-de- veloped, triangular medi- an apophysis, absent in males of the other three species (Figs. 1A, 2A vs. Fig. 4A, D, G).
Female D. culsu sp. nov. can be easily distin- guished from female D. speluncarum by the nar- rower, trapezoid-shaped median septum with slant- ing edges, in contrast with the larger, lobate Ms with rounded edges in D. spe- luncarum (Figs. 1E, 2D vs. Fig. 5A). Female D. culsu sp. nov. are separated from female D. morisii by the absence of a bulge on the Ms (clearly visible in D. morisii, Figs. 1E, 2D vs. Fig. 5C). Additionally, they can be distinguished by the different shape of copulatory ducts when the vulva is observed dorsally: with a rather uniform diameter in D. culsu sp. nov. and bearing a large, flattened middle trait in D. morisii (Figs. 1G, 2E vs. Fig. 5D). Female D. culsu sp. nov. are easily sepa- rated from those of D. sbordonii by the trapezoid-shaped Ms with slanting edges, in contrast with the squared Ms with vertical edges in D. sbordonii (Figs. 1E, 2D vs. Fig. 5E). They can further be distinguished by the different position of spermathecae, located in the lower half of the vulva and below the vulval pockets in D. culsu sp. nov., in contrast with S located in the upper half of vulva and above Vp in D. sbordonii (Figs. 1G, 2E vs. Fig. 5F). Description. Male (holotype). Total length 4.19. Carapace: 1.81 long, 1.56 wide.
Habitus as in Fig. 3A View Figure 3 . Carapace uniformly pale yellow with some sparse setae (more reddish while alive, see Fig. 3A View Figure 3 ). Cephalic region not clearly differentiated from the rest of carapace. Eyes reduced, AM missing, reduced to black maculae. Eye diameters: AM -, AL 0.079, PM 0.080, and PL 0.078. Thoracic grooves and fovea distinct. Mouthparts and sternum uniformly colored as the carapace. Promargin of chelicera with three teeth approximately of the same size, retromargin with several small denticles. Legs uniformly light yellowish. Legs measurements as follows: I 17.40 (4.89, 0.90, 4.78, 4.90, 1.93), II 14.03 (3.92, 0.80, 3.67, 3.76, 1.88), III 10.35 (3.19, 0.69, 2.53, 2.79, 1.15), IV 13.47 (4.34, 0.84, 3.53, 3.49, 1.27). Leg formula: I, II, IV, III. Opisthosoma gray-yellowish colored (lighter than carapace while alive, see Fig. 3A View Figure 3 ), covered with long hairs.
Palp as in Figs. 1 View Figure 1 A‒C, 2A-C. Cymbium oval, covered with short, sparse setae, with a tuft of longer hairs in the pro- lateral distal area near the tip. Embolus filamentous, slender in the terminal part. Conductor complex with three distinct processes: Cp 1-3. Cp 1 stocky and roughly triangularly shaped, Cp 2 and Cp 3 located at the distal part of the bulb and diagonally protruding (approx. 2 o’clock seeing the left palp ventrally), their tips curved towards each other ( Figs. 1A, C View Figure 1 , 2A, C). Median apophysis well-developed, shaped as a long, sharp triangle, heading prolaterally ( Figs. 1A View Figure 1 , 2A). Para- cymbium large with well-developed, sclerotized dorsal, distal and ventral processes. Two dorsal apophyses, Da 1-2: Da 1 robust and long, ending sharply, S-shaped when the palp is observed dorsally, Da 2 stocky, triangularly-shaped. Distal apophysis triangularly shaped. Ventral apophysis lobate, dorso-ventrally flattened and heading toward the cym- bium ( Figs. 1 View Figure 1 A‒C, 2A-C).
Female (based on 4 paratypes). Total length 3.65–5.27. Carapace: 1.71–1.98 long, 1.54–1.64 wide.
Habitus as in Fig. 3B, C View Figure 3 . Carapace uniformly yellowish with some sparse setae (often more reddish while alive, see Fig. 3B View Figure 3 ). Cephalic region not clearly differentiated from the rest of the prosoma.Eyes reduced, AM strongly reduced and bare- ly visible, reduced to small, dark maculae in some specimens. Eye diameters: AM (when present): 0.032, AL: 0.087, PM: 0.078, and PL: 0.77. Thoracic grooves and fovea distinct. Mouthparts and sternum uniformly colored as in the carapace. Teeth of chelicera as in the male. Legs uniformly light yellowish. Leg measurements as follows: I 17.07 (4.38, 1.01, 4.95, 4.90, 1.83), II 13.69 (4.04, 0.92, 3.57, 3.64, 1.52), III 10.46 (3.43, 0.80, 2.50, 2.56, 1.17), IV 13.89 (4.64, 0.92, 3.59, 3.41, 1.33). Leg formula: I, IV, II, III. Opistho- soma yellowish-gray (often lighter colored than carapace while alive, see Fig. 3B View Figure 3 ), covered with long hairs.
Epigyne as in Figs. 1E, F View Figure 1 , 2D, E. Medi- an septum short, not protruding, shaped as an inverted trape- zoid with a narrower base. Vulval pockets and copulatory ducts externally visible by transparence trough the tegument. Copu- latory openings at the lower, lateral side of median septum. Vul- va as in Figs. 1G View Figure 1 , 2E. Spermathecae small and round, located in the lower-half of the vulva, below vulval pockets and being partially covered by them. Vulval pockets wide and rounded, sac-shaped, located above spermathecae. Copulatory ducts with a wider diameter in the ventral trait and narrower in the dor- sal trait, rolling up around the lower part of vulval pockets and reaching spermathe- cae with some turns ( Figs. 1F View Figure 1 , 2E). Insem- ination ducts begin- ning from the lower part of spermathe- cae and following the same course of copu- latory ducts.
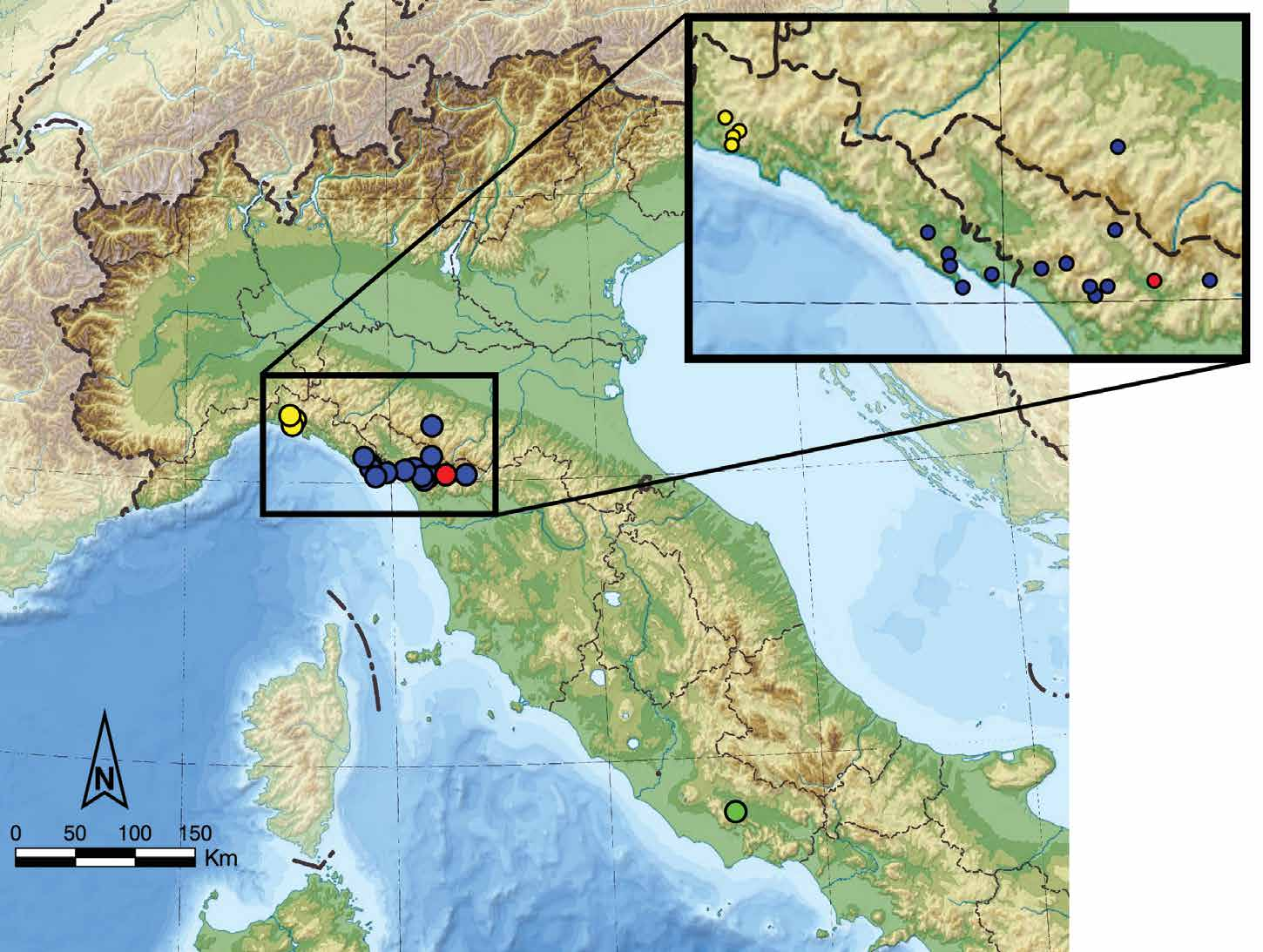
Distribution
Italy, endemic to the northern Apennines. Known only from the type locality; Tana delle Fate di Coreglia Antelminelli cave ( Fig. 6 View Figure 6 ). Biospeleological and ecological notes
The entrance of Tana delle Fate di Coreglia Antelminelli cave (Italian National Caves Registry number: 141/T/LU; Fig. 3E View Figure 3 ) opens in the left bank of the narrow valley of Segone Creek in the Province of Lucca (Toscana region) at an elevation of 260 m a.s.l. The cave occurs in the limestone of the Maiolica formation (lower Tithonian‒lower Aptian, ~150–120 Ma), which is particularly rich in flint nodules. After an initial steep slope (approximately 10 m deep), the cave continues with a long and sub-horizontal spatial development and a general NW‒SE orientation ( Fig. 3F View Figure 3 ). It branches with several, sub-circular tunnels as a result of ancient groundwater flows. The cave has an estimated total extension of 1100 m, although the deeper segments are still unexplored, as they are either filled with water or ending with sumps. The inner section is generally humid, with mud often covering the bottoms of the tunnels. The cave hosts a rich sub- terranean fauna including some endemic or locally protected species, e.g. the carnivorous land snail Oxychilus sp. ( Gastropoda, Oxychilidae ), the cave cricket Dolichopoda laetitiae Minozzi, 1920 ( Orthoptera , Rhaphidophoridae ), the blind subterranean beetle Duvalius apuanus lanzai Straneo, 1943 ( Coleoptera , Trechinae), the Italian cave salamander Speleomantes italicus (Dunn, 1923) ( Amphibia, Plethodontidae ), and three species of bats: the greater horseshoe bat Rhinolophus ferrumequinum (Schreber, 1774) , the lesser horseshoe bat Rhinolophus hipposideros (Bechstein, 1800) ( Chiroptera , Rhinolophidae ), and the common bent-wing bat Miniopterus schreibersii (Kuhl, 1817) ( Chiroptera , Miniop- teridae). Other animals known from the cave from the literature ( Lanza, 1961) or directly observed by the author include: Octodrilus complanatus (Dugès, 1828) , O. hemiandrus ( Cognetti, 1901) , O. transpadanus (Rosa, 1884) , and Aporrectodea rosea (Savigny, 1826) (Anellida, Lumbricidae ); Chaetophiloscia cellaria (Dollfus, 1884) ( Isopoda , Philosciidae ), Androniscus dentiger Verhoeff, 1908 , and Spelaeonethes mancinii (Brian, 1913) ( Isopoda , Trichoniscidae ); Euscorpius carpathicus (Linnaeus, 1767) ( Scorpiones , Euscorpiidae ); Trogulus sp. ( Opiliones , Trogulidae ), Ischyropsalis a damii Canestrini, 1873 ( Opiliones , Ischyropsalididae ); Lithobius tylopus Latzel, 1882 ( Chilopoda, Lithobiidae ), Gryllomorpha dalmatina (Ocskay, 1832) ( Orthoptera , Gryllidae ), Hypaena sp. ( Lepidoptera , Noctuidae ), Stenophylax permistus Mc- Lachlan, 1895 (Tricoptera, Limnephilidae ), and a large population of limoniid crane flies ( Diptera , Limoniidae ). Near the entrance and in the early section of the cave, numerous spiders were also observed: Amaurobius ferox (Walckenaer, 1830) , A. pesarinii Ballarin and Pantini, 2017 (Amaurobiidae) , Kryptonesticus eremita (Simon, 1880) (Nesticidae) , Meta menardi (Latreille, 1804) , Metellina merianae (Scopoli, 1763) (Tetragnathidae) , Pholcus phalangioides (Fuesslin, 1775) (Pholcidae) , and Tegenaria sp. ( Agelenidae ). The new species was found in the initial segments of the cave, but at some distance from the entrance ( Fig. 3F View Figure 3 ). During summer, when the cave was visited, adults, subadults, and juveniles of D. culsu sp. nov. were observed together, with a substantially higher number of adults and subadults during the month of August. Most of the juveniles collected in the cave and bred in captivity became adults after 2‒3 months of captivity, while it took approximately one year for the youngest specimens to reach sexual maturity.
Two different species of nesticid spiders, K. eremita and D. culsu sp. nov., were collected together in the Tana delle Fate di Coreglia Antelminelli cave. These species cover a different spatial distribution within the cave ( Fig. 3F View Figure 3 ), coexisting without overlapping despite occupying approximately the same ecological niche. Cohabitant nesticids, in particular involving D. menozzii or D. speluncarum together with K. eremita , have been previously observed in several occasions in Italian caves, and sometimes collected at short distances from each other ( Brignoli, 1971). However, no clear species overlap are reported within the same cave. Such distinct spatial partition can be explained by the different grade of adaptation to the hypogean environment showed by these arachnids. In fact, K. eremita appears to be a less specialized cave-dweller, lacking extreme morphological adaptations to subterranean life. Therefore, it mostly occurs near the entrance of caves or inside artificial tunnels, including, occasionally, shadowed epigean habitats with constant temperature and high relative humidity ( Brignoli, 1971 and personal observations by the author). On the other hand, all Domitius species show a greater degree of adaptation to the subterranean habitat, as suggested by reduction of the eyes and body depigmentation. Such strong adaptation allows Domitius to occupy deeper segments of the caves, thus avoiding direct competition with K. eremita .
Conservation Notes
Since caves are a unique and delicate ecosystem, they are highly susceptible to external disturbance ( Culver and Pipan, 2009). Its visible entrance and sub-horizontal extension makes Tana delle Fate di Coreglia Antelminelli cave easily accessible to visitors even with limited experience in speleology, and the cave is often used for training purpos- es by local speleological clubs. Although not threatened, D. culsu sp. nov. should be considered potentially at risk in case of frequent and long-lasting human disturbance due to its strict habitat requirements, its reduced population, and its extremely limited distribution, which appears to be confined to a single cave. Therefore, the new species is a good candidate for species conservation, deserving a place in the list of locally protected species.
Phylogenetic Analysis
A total of 16 nesticid species were used in this study, including representatives of the main nesticid genera present in Europe and all the species distributed in the Italian peninsula. Taxon sampling comprised the wide majority of Domitius species. Only D. murgis (Ribera and De Mas, 2003) from Spain was excluded from the analysis due to the absence of available sequences and fresh samples. The final dataset was formed by 1975 pair bases (bp) distributed as; COI = 1197 bp, 16S = 469 bp, and H3 = 309 bp. The resulting phylogenetic tree is illustrated in Figure 7 View Figure 7 , and the uncorrected pairwise distance between the species is reported in Table 2 View Table 2 . The European nesticids cluster into five different clades corresponding to the main genera Carpathonesticus , Domitius , Kryptonesticus , Nesticus , and Typhlonesticus , each of them highly supported (bootstrap support value = 100%). Each lineage represents a different and well-defined evolu- tionary line. These results concur with the outcomes of recent morphological and phylogenetic studies on the family Nesticidae ( Pavlek and Ribera, 2017; Ribera, 2018; Ballarin and Li, in prep.), supporting the validity of the newly-es- tablished genera. According to these results, Domitius represents the sister lineage of the monophyletic clade formed by the genera Carpathonesticus , Kryptonesticus , and Nesticus , with which it shares a common ancestor. The analysis supports Typhlonesticus as a basal clade within the European Nesticidae , as also suggested by recent molecular stud- ies ( Ballarin and Li, 2018; Ribera, 2018). Within Domitius , D. culsu sp. nov. shows a closer affinity with the species from the same geographic area; particularly D. speluncarum , but also D. menozzi . Its position at the far end of the phyloge- netic tree of the genus suggests a more recent origin in comparison with the other congeneric species.
All the Domitius species distributed in the Northern Apennines share a close affinity with species from the Iberian Peninsula. Such close relations also reflected in genital morphology. For instance, all these species share a similar position of spermathecae, located in the lower-half of the vulva, and below the vulval pockets (see Figs. 1G View Figure 1 , 5B, D and Figs. 4 View Figure 4 A‒E in Ribera, 2018). On the other hand, D. sbordonii from the Central Apennines appears to be morphologically and genetically separated from all the other species of the genus, including those from Northern Apennines. The differ- ence is highlighted in the peculiar shape of the vulva, being the only Domitius species showing simple-coiled internal ducts and spermathecae located in the upper-half of the vulva, over the vulval pockets (see Fig. 5F View Figure 5 vs. Figs. 1G View Figure 1 , 5B, D and Figs. 4 View Figure 4 A‒E in Ribera, 2018). Upper-positioned spermathechae are also present in several other European nesticid genera such as Carpathonesticus ( sensu stricto), Krypthonesticus, and Nesticus ( sensu stricto). Based on these results it is possible to speculate that D. sbordonii represents a basal element within the genus Domitius , possibly still carrying the ancestral characters of the older forebear of the European nesticids.
Conclusions
All Italian species of the genus Domitius appear to be highly adapted to a permanent life in the subterranean environ- ment, showing eye reduction and lack of body pigmentation. They further present a localized distribution, with distinct genetic and morphological differences between the species living in the Northern and Central areas of the Apennines. At the same time a close affinity with the species distributed in the Iberian Peninsula is observed. Such features, to- gether with a high genetic p-distance among the species ( Table 2 View Table 2 ), suggests a potentially complex evolutionary history of the genus Domitius that still needs to be properly explored (see also Ribera, 2018).
Because of their apparent similarities in habitus and female genitalia, D. culsu sp. nov has previously been mistaken for D. speluncarum and ignored as a distinct species by previous arachnologists. A detailed molecular and morpho- logical analysis of both sexes of D. culsu sp. nov carried out in this work supports the validity of the new species and its close relationship with the other Domitius species from the same geographical area. Its potential susceptibility to external disturbance, and extremely limited distribution, makes D. culsu sp. nov of interest for conservation.
Finding a new nesticid species in Italy further suggests that our knowledge on the diversity of the family Nesticidae in Southern Europe is still far from complete. Further collections along the Italian peninsula will probably lead to the discovery of other highly-specialized nesticid species allowing a deeper and more precise understanding of the spider cave fauna in Italy and in the Mediterranean area.
| MSNV |
Italy, Verona, Museo Civico di Storia Naturale |
| MSNB |
MSNB |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Domitius culsu Ballarin
| Ballarin, Francesco 2020 |
Nesticus speluncarum
| Brignoli 1979: 214 |