Cyrtodactylus erythrops, Bauer, Aaron M., Kunya, Kirati, Sumontha, Montri, Niyomwan, Piyawan, Panitvong, Nonn, Pauwels, Olivier S. G., Chanhome, Lawan & Kunya, Tunyakorn, 2009
|
publication ID |
https://doi.org/10.5281/zenodo.188206 |
|
DOI |
https://doi.org/10.5281/zenodo.6226865 |
|
persistent identifier |
https://treatment.plazi.org/id/03BFC161-FFB3-FFB0-D8FC-21FEFCA902EA |
|
treatment provided by |
Plazi |
|
scientific name |
Cyrtodactylus erythrops |
| status |
sp. nov. |
Cyrtodactylus erythrops View in CoL sp. nov.
Figures 1–3 View FIGURE 1 View FIGURE 2 View FIGURE 3 .
Holotype. Thai Natural History Museum ( THNHM) 15377 (field number MS 150), adult male; Thailand, Mae Hong Son Province, Pangmapha district, Tham Lod, 19°37’05”N 98°11’01”E, 640 m a.s.l., collected by Eakarit Punamphon, April 2005.
Additional Specimen. Juvenile (photographed but not collected); same data as holotype (see Coloration below).
Etymology. The specific epithet is derived from the Greek erythros [= red] and ops [= eye] and refers to the reddish life coloration of the iris of this species. Tuk kai ta daeng ( Thai), Red-eyed bent toed gecko (English), Cyrtodactyle aux yeux rouges (French), Rotaugen-Bogenfingergecko ( German), Rood ogige kromvingergekko (Dutch).
Diagnosis. Cyrtodactylus erythrops can be distinguished from all congeners on the basis of the following combination of characters: body slender, limbs and digits long, dorsal scalation with 18–20 rows of large, flattened, smooth to weakly-keeled or weakly conical tubercles, some in contact with one another, 28 ventral scales across belly between weakly developed ventrolateral folds, no precloacal groove, precloacal pores in a continuous series of 9, separated from a broken series of 9–10 femoral pores on each thigh by 2–3 poreless scales, femoral scales enlarged, dorsal pattern consisting of roughly transversely-oriented rows of palerimmed, dark brown blotches, posteriorly forming irregular crossbands, and a series of blotches forming a broken nuchal loop.
Description. Adult male ( Figure 1 View FIGURE 1 ). Measurements (in mm): SVL 78.4, TailL (tail broken, see below) 7.3 TrunkL 31.3, ForeaL 13.8, CrusL 16.6, HeadL 22.4, HeadW 14.91, HeadD 9.8, SnEye 8.9, OrbD 6.4, EyeEar 6.8, EarL 2.3, Internar 2.6, Interorb 3.4). Specimen dehydrated and somewhat brittle. Head relatively long (HeadL/SVL = 0.29), wide (HeadW/HeadL = 0.67), not markedly depressed (HeadH/HeadL = 0.44), markedly distinct from very slender neck. Loreal region concave anteriorly, weakly inflated posteriorly, canthus rostralis relatively prominent. Snout elongate (SnEye/HeadL = 0.40), rounded; longer than eye diameter (OrbD/SnEye = 0.72); scales on snout and forehead small, rounded, granular, homogeneous; scales on snout smooth, flat, pentagonal to hexagonal, 4–8 times larger than minute, smooth, flattened to very weakly domed, juxtaposed scales on occipital region. Eye large (OrbD/HeadL = 0.29); pupil vertical with crenelated margins; superciliaries short, with rounded outer margins except for a few scales at posterodorsal border of orbit, which bear tiny conical spines. Ear opening vertically elliptical, large (EarL/HeadL = 0.10); eye to ear distance subequal to diameter of eyes (EyeEar/OrbD = 1.07). Rostral much wider ( 3.4 mm) than deep ( 2.2 mm), incompletely divided (~40%) dorsally. Two enlarged supranasals separated by a single smaller internasal. Rostral in contact with supralabials I, nostrils, supranasals and internasal. Nostrils rounded, each surrounded by supranasal, rostral, first supralabial and four postnasals. A concave narial valve occupies the posterior 75% of the nostril. At least 3–4 rows of small scales separate orbit from supralabials. Mental triangular, wider ( 3.5 mm) than deep (2.0 mm). A single pair of enlarged median postmentals, each postmental bordered anteromedially by mental, anterolaterally by first infralabial, and posterolaterally by an enlarged lateral chinshield (= second postmental) approximately 50% of its size. Together the median postmentals border a row of five chin scales, each approximately twice the size of all more posterior chin and gular scales. Supralabials to midorbital position 7 (left)/6 (right); enlarged supralabials to angle of jaws 8 (left)/7 (right). Enlarged infralabials 6 (left)/7 (right) to inflection of jaw, 9 (bilateral) to corner of rictus. Interorbital scale rows across narrowest point of frontal bone 18.
Body slender, relatively short (TrunkL/SVL ratio 0.40) with extremely weakly developed, non-denticulate ventrolateral folds. Dorsum highly heterogeneous, with small, irregular, juxtaposed, domed to weakly conical, oblong scales intermixed with flattened, smooth to very weakly conical or weakly keeled, oval to rounded tubercles. Keels, where present, restricted to posterior 30–50% of tubercles. Tubercles extending from posterior margins of orbits and of frontal bone onto entire dorsum; 18–20 irregular rows at midbody, lateralmost rows reaching ventrolateral folds. Tubercles increasing in size from head (2–3 times size of adjacent granules) to midbody, largest in paravertebral rows (5–8 times size of adjacent scales), narrowly separated from one another or in direct contact, especially on posterior flanks ( Figure 2 View FIGURE 2 ). Ventral scales much larger than dorsals (exclusive of tubercles), smooth, with rounded or pointed free margins, imbricate, becoming larger posteriorly, midventrally, and in precloacal region. Midbody scale rows across belly to base of ventrolateral folds 28. Gular region with relatively homogeneous, smooth, domed, granular scales.
Precloacal region with four sequential transverse rows of enlarged scales, each 3 to 5 times size of adjacent ventral scales. A single series of nine precloacal pores. A diamond-shaped patch of eight very large scales between precloacal pore-bearing scales and granular scales on anterior cloacal lip. No precloacal groove. A continuous row of enlarged femoral scales, each about 1.5–2 times size of those in more anterior scale rows and> 4 times that of those on posteroventral surface of thigh, extending along length of femur almost to knee and continuous with the enlarged precloacal scales. Femoral pores separated from precloacal pores by a diastema of 2 (left) to 3 (right) poreless scales; 9 (right) to 10 (left) femoral pores, with medialmost pore on each thigh separated by one poreless scale from the more distal pores, which form a continuous series ( Figure 3 View FIGURE 3 ).
Scales on palm and sole smooth, flat, oval to pentagonal or hexagonal. Scalation on dorsal surfaces of hind limbs similar to body dorsum with enlarged, flattened, weakly-keeled tubercles interspersed among smaller scales. Scales of dorsal surface of forearms less strongly tuberculate, tubercles oval, feebly keeled. Upper arms without tubercles, scales somewhat variable in size and arranged in diagonal rows. Fore and hindlimbs moderately long, slender (ForeaL/SVL ratio 0.18; CrusL/SVL ratio 0.21). Digits long, slender, strongly inflected at interphalangeal joints, all bearing robust, slightly recurved claws. Basal subdigital lamellae broad, rectangular, without scansorial surfaces (3-5-5-5-6 left manus; 5-5-5-7-6 left pes); narrow lamellae distal to digital inflection and not including ventral claw sheath: 9-10-10-11-11 (left manus), 8-11- 11-13-13 (left pes); interdigital webbing apparently absent (slight webbing could be obscured by skin shrinkage due to dehydration). Relative length of digits: III> IV> II> V> I (manus; digits II-V very similar in length); IV> V> III II> I (pes); distal phalanges of left pes partly separated from foot. Original tail broken near base in preserved specimen, but when captured the specimen had a TailL of 83 mm, with enlarged subcaudal plates. Series of 3 enlarged, raised, keeled cloacal spurs on each side of vent.
Coloration (in preservative). Dorsal ground color mottled straw to light brown, with roughly transverse rows of large, oval, dark brown spots or blotches with somewhat lighter centers; each spot surrounded by a thin whitish rim one scale wide or less. Spots over shoulders and limb insertions not in distinct rows, those between limb insertions in 4 distinct transverse rows, last three largely coalescent, forming irregular transverse bands. These bands narrowing on flanks, interband regions of flanks with irregular dark blotches. A pair of dark blotches over sacrum and another over tail base. Confluence of white margins of blotches on posterior portion of trunk forming a very thin vertebral stripe from level of adpressed elbows on to tail.
Dorsum of head light brown, with distinct dark markings like those of body dorsum, forming a broken nuchal loop extending to the posterior margin of each eye and along the ventral margin of each orbit. A less well defined series of dark markings from posterodorsal margin of orbit across posterior parietal plate. Dark markings with indistinct margins on snout, loreal region, around margin of orbit and on frontal area. Labial scales with speckles of brown pigment, with less heavily pigmented borders. Anterior supralabials and ventral portion of mental and postmentals whitish. Supraorbital skin translucent with underlying scleral ossicles clearly visible. Limbs with irregular, ill-defined, alternating mid- to dark brown and straw bands. Venter cream to beige with darker pigmentation on anterior cloacal lip and enlarged patch of precloacal scales.
In life this species has red eyes and the dark dorsal markings of the head and body are edged by very pale yellowish borders ( Fig. 4 View FIGURE 4 ). A juvenile specimen (not collected) had a pinkish-gray dorsum with purplishbrown markings. Dorsal blotches in this specimen exhibit a greater degree of fusion and irregularity than in the holotype and the original tail exhibits bold alternating banding of very dark brown and opalescent white ( Fig. 5 View FIGURE 5 ).
Comparisons with Other Species. Although a number of species groups within Cyrtodactylus have been proposed on the basis of phenetic similarity (e.g., the louisiadensis complex, Kraus 2008) or geographic proximity (e.g., the Sundaland group, Hayden et al. 2008), there exists no explicit phylogenetic hypothesis of relationships within the genus. Ongoing molecular phylogenetic work (Bauer et al., unpublished) suggests that certain geographic clusters of taxa and morphologically similar species do correspond to monophyletic groups within Cyrtodactylus . However, fewer than 60% of the species have been sampled thusfar and it is not yet possible to unambiguously assign all species to particular clades. Thus, we here provide comparisons of C. erythrops sp. nov. with all of the Cyrtodactylus species described as of 31 December 2008 (exclusive of the distinctive South Asian Geckoella , sometimes considered as a subgenus of Cyrtodactylus ). Although highly artificial, we have employed commonly used and easily scorable single differential characters for this purpose. Recent summaries of some of these, and other, features for large numbers of Cyrtodactylus species have been provided by Youmans & Grismer (2006), Grismer et al. (2007), Grismer & Ahmad (2008), Linkem et al. (2008), and Rösler & Glaw (2008).
The condition of precloacal and femoral pores in males has been widely employed to distinguish Cyrtodactylus species from one another (e.g., Smith 1935; Darevsky & Szczerbak 1997; Das & Lim 2000; Bauer 2002, 2003; Youmans & Grismer 2006). Although females of some species lack pores, these are present in other taxa (Bauer 2003). These features appear, however, to vary little between adult males within a species and we consider them to be reliable diagnostic characters. However, for those species known only from females or juveniles, we have used other features for diagnostic purposes. The presence of precloacal pores in males distinguishes C. erythrops from C. eisenmani Ngo ; C. gordongekkoi (Das) (recently transferred from Cnemaspis by Biswas 2007), C. jellesmae (Boulenger) , C. laevigatus Darevsky , C. semenanjungensis Grismer & Leong , C. sermowaiensis (de Rooij), C. thirakhupti , and C. wallacei Hayden et al. It may be distinguished from the following species by the presence of femoral pores: C. adleri Das , C. angularis , C. annulatus (Taylor) , C. ayeyarwadyensis Bauer ; C. badenensis Nguyen et al. ; C. brevidactylus Bauer , C. chauquangensis Hoang et al. , C. condorensis (Smith) , C. consobrinoides (Annandale) , C. cracens Batuwita & Bahir ; C. cryptus Heidrich et al. , C. edwardtaylori Batuwita & Bahir ; C. elok Dring , C. fraenatus (Günther) , C. gansi Bauer ; C. ingeri Hikida , C. intermedius , C. irianjayaensis Rösler , C. irregularis (Smith) , C. khasiensis (Jerdon) , C. lateralis (Werner) , C. malayanus (de Rooij), C. matsuii Hikida ; C. nigriocularis Nguyen et al. ; C. oldhami (Theobald) , C. pantiensis Grismer et al. , C. papuensis (Brongersma) , C. paradoxus (Darevsky & Szczerbak) , C. peguensis (Boulenger) , C. pseudoquadrivirgatus Rösler et al. , C. quadrivirgatus Taylor , C. ramboda Batuwita & Bahir ; C. soba Batuwita & Bahir ; C. subsolanus Batuwita & Bahir ; C. sumonthai , C. sworderi (Smith) , and C. yoshii Hikida , and from the following species by the presence of a diastema between the series of femoral pores and the precloacal pores: C. batucolus Grismer et al. , C. epiroticus Kraus , C. feae (Boulenger) , C. jarujini Ulber , C. klugei Kraus , C. louisiadensis (de Vis), C. malcolmsmithi (Constable) , C. phongnhakebangensis Ziegler et al. , C. robustus Kraus , C. seribuatensis Youmans & Grismer ; C. serratus Kraus , C. tiomanensis Das & Lim , and C. variegatus (Blyth) .
C. erythrops may be distinguished from the following species by the absence of a longitudinal or steeply angled precloacal sulcus or groove from C. aurensis Grismer ,, C. cavernicolus Inger & King, C. macrotuberculatus Grismer & Ahmad , C. philippinicus (Steindachner) , C. pubisulcus Inger , C. pulchellus Gray , C. rubidus (Blyth) , C. spinosus Linkem et al. , and C. stresemanni Rösler & Glaw , and by the absence of a recessed transverse precloacal groove in: C. fumosus (Müller) , C. marmoratus (Kuhl) , C. sadleiri Wells & Wellington , from C. biordinis Brown & McCoy by the presence of a single, versus double row of femoral pores, and from C. chrysopylos Bauer by the absence of an enlarged pore-bearing scale posterior to the precloacal pore row. An enlarged series of femoral scales separates the new species from C. annandalei Bauer , C. buchardi David et al. , C. grismeri Ngo , and C. wakeorum Bauer.
Cyrtodactylus erythrops may be distinguished from C. aequalis Bauer , C. murua Kraus & Allison , and C. russelli Bauer by its flattened ( versus prominently conical or mucronate) dorsal tubercles, from C. aaroni Günther & Rösler , C. agamensis (Bleeker) , C. baluensis (Mocquard) , C. caovansungi Orlov et al. , C. capreoloides Rösler et al. , C. consobrinus (Peters) , C. darmandvillei (Weber) , C. hontreensis Ngo et al. , C. huynhi Ngo & Bauer , C. interdigitalis Ulber , C. jarakensis Grismer et al. , C. takouensis Ngo & Bauer , C. tripartitus Kraus , and C. zugi Oliver et al. by its much lower number of ventral scales (28 versus 36 or more), from C. agusanensis (Taylor) , C. brevipalmatus (Smith) , C. derongo Brown & Parker , C. gubernatoris (Annandale) , C. loriae (Boulenger) , C. novaeguineae (Schlegel) , C. papilionoides Ulber & Grossmann , C. wetariensis (Dunn) , and C. ziegleri Nazarov et al. by the presence of enlarged subcaudal plates in the original tail, and from C. mimikanus (Boulenger) , C. redimiculus King , C. salomonensis Rösler et al. , and C. tuberculatus (Lucas & Frost) by its spotted/blotched ( versus banded) dorsal pattern. This pattern difference also distinguishes C. erythrops from its Thai congeners C. chanhomeae Bauer et al. and C. tigroides Bauer et al. (note that the condition of the femoral scales in these taxa was incorrectly listed in the comparative table of Youmans & Grismer 2006; they do, in fact, share with C. erythrops enlarged femoral scales).
The new species is phenetically quite similar to C. slowinskii Bauer from Myanmar, but is considerably smaller ( 78 mm versus 108 mm maximum SVL) and has relatively larger and more closely-spaced dorsal tubercles, and a dorsal pattern consisting of more well-defined but irregularly distributed, oval markings ( versus regularly distributed blotches of irregular shape). It also shares many similarities with members of the C. irregularis complex (sensu Nazarov et al. 2008), but can be differentiated from C. irregularis , C. pseudoquadrivirgatus , and C. ziegleri on the basis of the characters noted in the comparisons above, as well as its red eyes and specific dorsal patterning.
Cyrtodactylus abrae , described by Wells (2002), supposedly from the Iron Range of Queensland, Australia, has been demonstrated to be a nomen nudum ( Couper et al. 2004).
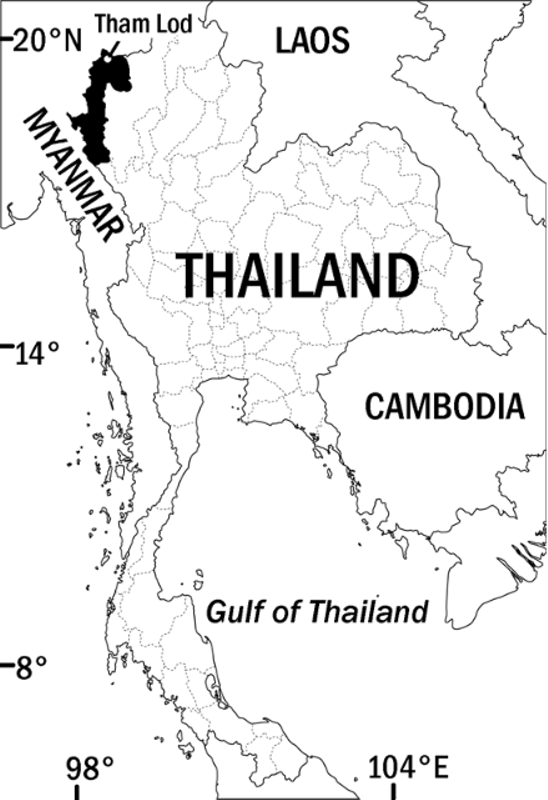
Distribution and Natural History. Cyrtodactylus erythrops sp. nov. has been collected only from Mae Hong Son Province in the extreme northwest of Thailand ( Figure 6 View FIGURE 6 ). The type locality is less than 10 km from the border of Myanmar and and lies in the West Thanon Thongchai Mountains,which are continuous with the Shan Plateau. It is thus likely that this species may occur in Shan State. Neither this vast region of Myanmar nor the adjoining areas of northern Thailand have been adequately surveyed for Cyrtodactylus and we suspect that many new species will eventually be discovered in the region.
Cyrtodactylus erythrops sp. nov. feeds by both day and night. Its natural diet appears to be composed principally of cave crickets, termites, and various insect larvae, whereas in captivity it takes both meal worms and termites. Breeding takes place in the winter season, at which time males emit advertisement vocalizations to females. The eggs, which are non-adhesive are oviposited in rock crevices within caves. The incubation period is approximately two months. THNHM 15377 was collected, along with two juveniles that were kept alive, just outside the entrance to Tham Lod cave.
We take this opportunity to make a correction to the type locality of another Thai cave gecko, C. sumonthai , fromTham Khang Khao, Khao Wong, Rayong. The incorrect latitude and longitude were inadvertantly provided in the description of that species (Bauer et al. 2002). The corrected coordinates are 12°53’N, 101°49’E.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |