Eumenes smithii de Saussure, 1852
|
publication ID |
https://doi.org/10.11646/zootaxa.4459.1.1 |
|
publication LSID |
lsid:zoobank.org:pub:633A9498-FCC6-4439-BE33-A45B9E51B550 |
|
DOI |
https://doi.org/10.5281/zenodo.5986386 |
|
persistent identifier |
https://treatment.plazi.org/id/03C5878F-FFE8-F31D-FF30-9312D035FDC7 |
|
treatment provided by |
Plazi |
|
scientific name |
Eumenes smithii de Saussure, 1852 |
| status |
|
Eumenes smithii de Saussure, 1852 View in CoL
Eumenes smithii de Saussure, 1852 View in CoL , 1: 43.
Type data: Syntypes at NHM and MNHN.
Type locality: “ La presqu’île de Florida ” ( USA).
de Saussure, 1875, 254: 104.
Cresson, 1887: 287 (cat.).
Dalla Torre, 1894, 9: 32 (cat.);
Dalla Torre, 1904, 19: 25 (cat.).
Bequaert, 1938, 33: 70 (note on types).
Bequaert, 1944, 71: 79.
MacLachlan, 1980, 53: 617, 619 (key).
Buck et al. 2008, 5: 44 (key), 154.
Eumenes belfragei Cresson, 1872 , 4: 232.
Type data: Lectotype female and two paratypes male at ANSP (examined).
Type locality: “ Texas ” ( Lectotype and paralectotypes).
Cresson, 1887: 287 (cat).
Dalla Torre, 1894, 9: 19 ( belfragii [!]; cat.).
Dalla Torre, 1904, 19: 21 ( belfragi [!]; cat).
Cresson, 1916, 1: 103 (designation of lectotype).
Bequaert, 1938, 33: 68 (note on type).
Bohart & Menke, 1974, 47: 459, 461.
Krombein, 1979: 1508.
Eumenes smithii smithii ; Bohart, 1951: 885 (cat.).
Krombein, 1958: 163.
Krombein, 1979: 1508 (cat.).
Ahlstrom, 1995, 1: 102 (list).
Eumenes smithii belfragei ; Bohart, 1951: 885 (cat.).
Krombein, 1958: 163: 1508.
Giordani Soika, 1978, 29: 29.
Rodríguez-Palafox, 1996: 479 (list).
Ecology: Prey: Geometridae larvae.
Ethology: Hartman, 1913, 3: 353–360 (nest building, prey, life history).
Isely, 1917, 10: 350 (nest).
Distribution: U.S.A. (AL, FL, GA, KS, MS, NC, OK, TN, TX).
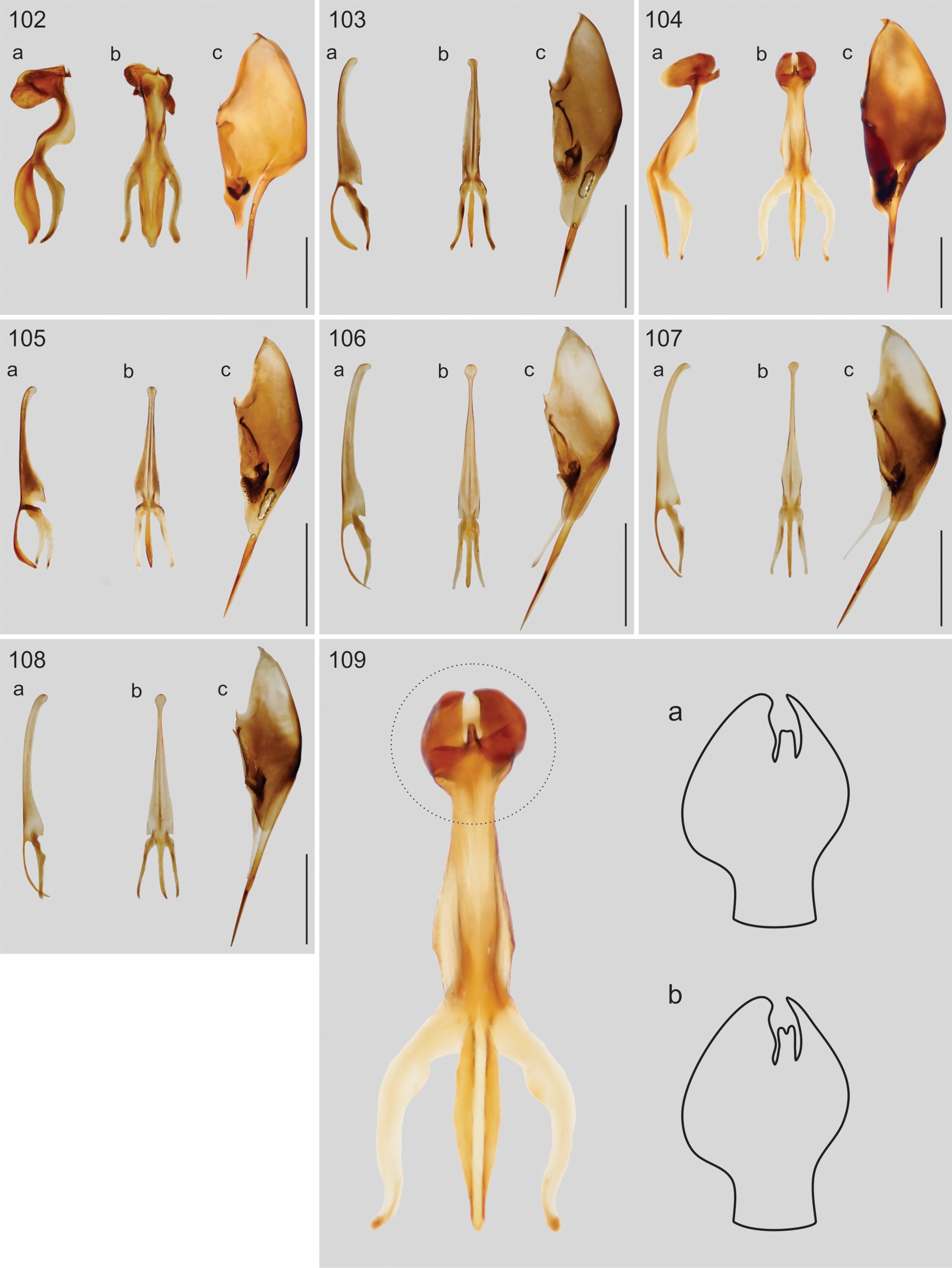
Comments and diagnosis. We did not examine the type specimens of Eumenes smithii but some specimens (females and males) were compared to the original description and later redescriptions (de Saussure, 1852, 1875; Bequaert, 1944); these were used to produce the diagnosis of the species ( Fig. 18 View FIGURES 13–26 ).
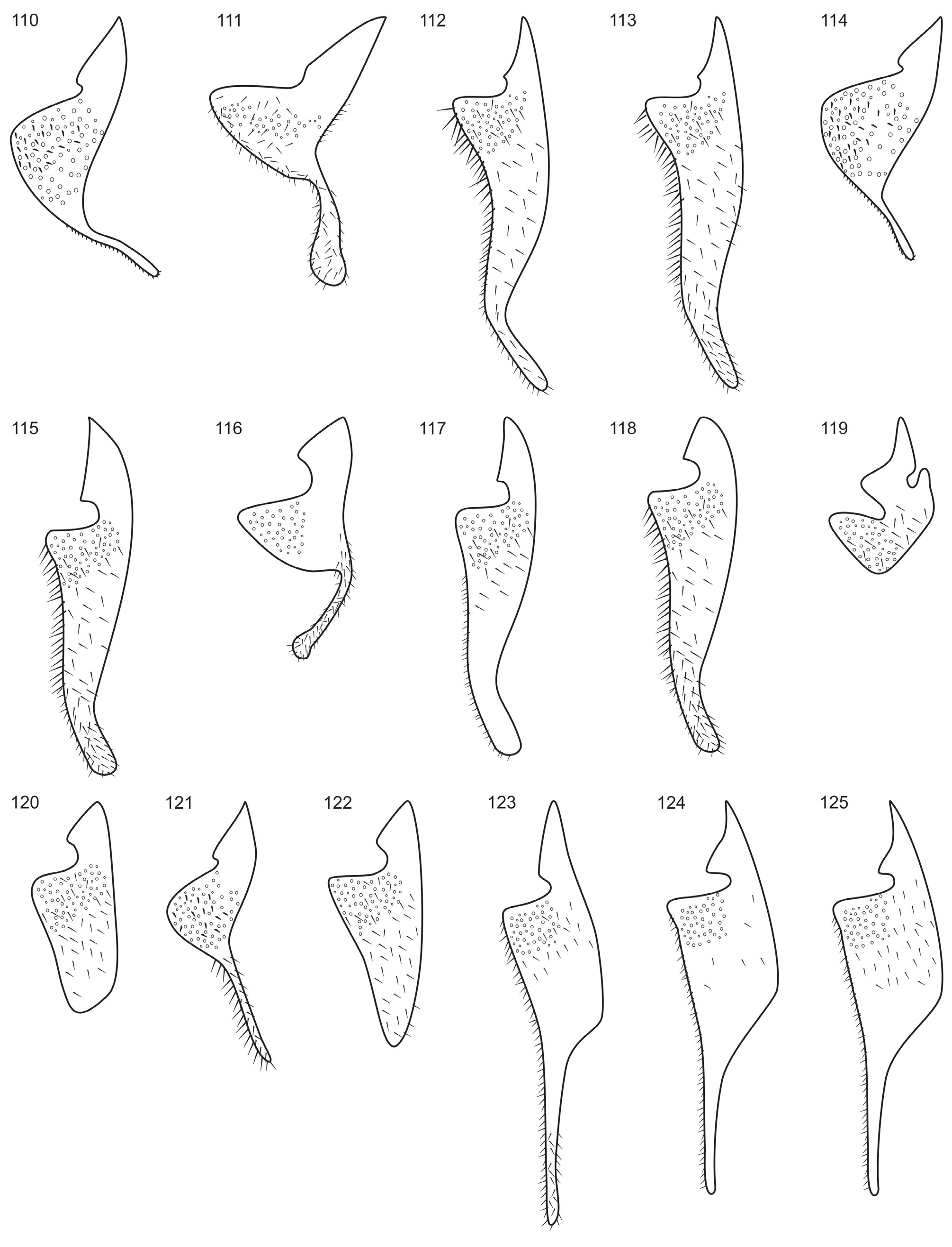
Eumenes smithii has its distribution in the central-east of the USA and presents darker background coloration (mainly of the scutellum and metasoma) than E. americanus . The clypeus is always black with coarse punctation, evenly distributed, up to the apex ( Fig. 67 View FIGURES 66–76 ). T1 has coarse punctation from the pre-basal region, becoming denser near the apex ( Fig. 38 View FIGURES 27–42 ). The punctation is not as deep and dense on the dorsal surface of T2 ( Fig. 54 View FIGURES 43–58 ). The apex of aedeagus has a rounded protuberance (in lateral view) ( Fig. 104a View FIGURES 102–109 ) and the apical ventral projection varies from truncate to weakly concave, ( Fig 109a, b View FIGURES 102–109 ); the ventral lobe is weakly rounded ( Fig. 104a View FIGURES 102–109 ); and the digitus has short, but more evident bristles than E. americanus ( Fig. 121 View FIGURES 110–125 ). We examined specimens originally identified as E. smithii and E. belfragei and there were no evident morphological differences to separate both taxa, but a range of variation can be observed for the species: most specimens from the central USA ( Kansas, Oklahoma and Texas) have the yellow marks more developed, mostly on T2, the apical projection of the aedeagus strongly concave, and the bristles of the digitus more evident. Specimens from the southeastern USA (records from Alabama, Florida, Georgia, Mississipi and North Carolina), however, showed no yellow marks on T2, although those on the mesosoma are present as in the central region. The apical projections of the aedeagus of this population were scarecely concave or truncate ( Fig. 109a, b View FIGURES 102–109 ) and the bristles of the digitus were less evident. Males examined from Tennessee and Mississipi showed intermediate features in a less truncate apical projection and bristles of the digitus, and besides that, specimens from both regions showed no consistent pattern of the yellow marks, including the type specimen of E. belfragei which is from Texas but has no yellow marks on metasoma, as in the typical E. smithii . This evidence leads us to believe that both taxa are indeed synonyms with intra-specific variation across the range of distribution of the species.
Color, head: Black, with yellow marks as follows: mandibles and labrum. Brownish (sometimes with yellowish marks) as follows: around the edges of the clypeus; inter-antennal region; a band on inner margin of compound eyes, which goes from the base of the clypeus to the ocular sinus; a narrow band on upper half of gena, adjacent to the compound eye; antenna, with some distal flagellomeres with blackish marks. Mesosoma: Black, with brownish and yellowish marks as follows: entirely pronotum, tegula and parategula (also with yellow marks); triangular marks on each side of mesosucutum; scutellum (with a narrow yellow band on apex); metanotum (with a yellow band); broad mark on upper half of mesepisternum, adjacent to the mesepimeron; all lateral region of the posterior face of propodeum. Marks on coxae; almost entirely femora and entirely tibiae and tarsi, although the distal tarsi are darker. Metasoma: T1 and S2 black on basal half and brownish on apical half; T2 with black marks on basal region and a transverse black band on middle area. Other regions are covered with brownish marks. T3– T6 and S3–S6 brownish with some yellow marks.
Pubescence, head: frons, vertex and occipital region with moderately long and golden pubescence. Clypeus and gena with shorter pubescence, whitish, better observed in oblique view. Some longer and erect bristles scattered on clypeus. Mesosoma: Mesoscutum, scutellum and metanotum with moderately long golden pubescence. Lateral surface of mesosoma whitish pubescence (better observed in oblique view) as long as on mesoscutum. Posterior face of propodeum with long (longer than frons and mesoscutum) and whitish pubescence. Apical half and basal half of fore coxa and femur, respectively, and trochanter with erect golden bristles.
Metasoma: Dorsal surface of T1 and T2 with short golden pubescence. Ventral surface of T1 with scattered moderately long golden pubescence. T3–T6 and S2–S6 with short golden pubescence, except long bristles on apex.
Surface of integument: Clypeus with coarse punctation homogeneously distributed. Frons and vertex with coarse and dense punctation, which becomes very sparse behind the ocelli. Gena with reduced punctation.
Mesosoma: Covered with coarse and dense punctation, except on anterior and posterior portion of mesepisternum, where the punctation is sparser (almost absent on anterior portion). Compared to E. americanus , the punctation from mesoscutum, scutellum, metanotum and posterior face of propodeum are coarser, forming carinae between the punctures. Metasoma: T1 with coarse and dense punctation from the base; dorsal face of T2 covered with slightly coarse (less than T1 and mesosoma), dense, becoming weaker towards apex and lateral margins. S2 with evident punctation mostly on lateral margins. T3–T6 and S3–S6 with reduced punctation
Structure: Clypeus convex, with the apex concave, producing two rounded apical projections with weak carinae on the apex of each one ( Fig. 67 View FIGURES 66–76 ). Inter-antennal region slightly wider than the antennal socket, longitudinally cariniform. Lateral ocelli closer to the compound eyes than to each other. Occipital carina well developed and angled on middle region of gena, not so close to the compound eye near mandibles. Mesosoma: Pronotal fovea present; pronotal carina weakly developed on dorsal face of pronotum and more developed on lateral face of it. Mesepimeron slightly elevated regarding the mesepisternum. Posterior projection of tegula weak developed, rounded; parategula lamelliform; posterior face of propodeum strongly concave on basal half. Basal half of the external surface of fore coxae with a well developed carina. Metasoma: T1 long, about 2.5 times longer than wide, moderately swollen in dorsal view ( Fig. 38 View FIGURES 27–42 ). Lateral margins on the apical half subparallel. T2 longer than wide, with an apical lamella that becomes reduced towards lateral margins ( Fig. 54 View FIGURES 43–58 ). Angle of T2, in lateral view, rising up abruptly.
Males: Clypeus narrower than females; F11 short with the apex pointed, surpassing the apical edge of F8 ( Fig. 88 View FIGURES 77–92 ). Dorsal surface without longitudinal carina. Ventral surface without microscopic erect bristles. S7 flattened apically, with bristles scattered on apex (also on T7). Color, pubescence and punctation as on females.
Male genitalia: Aedeagus as in Fig. 104a, b View FIGURES 102–109 . Apical projection varying from truncate to strongly concave ( Fig. 103a, b View FIGURES 102–109 ). Paramere ( Fig. 104c View FIGURES 102–109 ) with long bristles on middle area of gonostyle; digitus strongly slender on apex, with short and evident bristles ( Fig. 121 View FIGURES 110–125 ); cuspis with thin, long and very dense pubescence; volsella with short (shorter than on cuspis) but evident scattered bristles; distal lobe truncate with a few erect bristles on the edge.
Variation: As discussed above, specimens from the central USA ( Texas, Oklahoma and Kansas) usually have more developed yellow marks on the mesosoma. Besides that, the apical projection of the aedeagus is usually more concave ( Fig. 103b View FIGURES 102–109 ) and the bristles of the digitus are more evident.
Type material. Lectotype female of Eumenes belfragei is in good condition and bears the labels: ‘Tex’ ‘[Red Label] TYPE No. \ 20671’ ‘ Eumenes \ belfragei \ Cres.’.
| ANSP |
Academy of Natural Sciences of Philadelphia |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
|
|
SubGenus |
Eumenes |
Eumenes smithii de Saussure, 1852
| Grandinete, Yuri Campanholo, Noll, Fernando Barbosa & Carpenter, James 2018 |
Eumenes smithii
| de Saussure 1852 |