Megaphyllum sensu stricto Verhoeff, 1894
|
publication ID |
https://doi.org/ 10.11646/zootaxa.3741.1.2 |
|
publication LSID |
lsid:zoobank.org:pub:BF5EA9B8-C6F4-448A-BEF9-1976AB4EC308 |
|
DOI |
https://doi.org/10.5281/zenodo.6151701 |
|
persistent identifier |
https://treatment.plazi.org/id/03C887D3-FFFB-FFB3-FF34-9FC5FC5DAEFA |
|
treatment provided by |
Plazi |
|
scientific name |
Megaphyllum sensu stricto Verhoeff, 1894 |
| status |
sensu stricto |
Subgenus Megaphyllum sensu stricto Verhoeff, 1894
Chromatoiulus (Chromatoilulus) : Lohmander 1936: 104; Attems 1940: 306. Brachyiulus (Chromatoiulus) : Verhoeff 1896a: 219; 1896b: 211; Attems 1899: 328. Brachyiulus (Brachyiulus) : 1930: 1668.
Megaphyllum (Chromatoiulus) : Verhoeff 1895:240
A rough definition of the subgenus Megaphyllum s.str. was given by Lazányi et al. (2012) on the basis of previous publications (Verhoeff 1894d, Attems 1927); prior to that it had never been defined under the subgenus name Megaphyllum . Verhoeff (1896a, 1896b) focused on gonopod structure, occasionally supplemented with other characters of the head, eye, repugnatorial glands, and form and setation of telson (Verhoeff 1930).
Type species: Megaphyllum projectum Verhoeff, 1894 .
Diagnosis. A subgenus of the genus Megaphyllum with considerably broader promere than opisthomere, tightly “embracing” the latter antero-laterally, with an oblique riffle on its caudal surface. Opisthomere always with a well-developed posterior process, joining the main opisthomere body at an acute angle; usually with an anterior process, this being vestigial or fully absent in some species; solenomere with two distinct processes—posterior and anterior, the latter positioned at the end of the flagellum-conducting lamella. Vulvae subcylindrical, with an oval, mostly apically positioned opening; operculum shorter than bursa.
Description: Body length and height: males: 19–53.9mm, 1.4–3.5mm; females: 20.2–54.6mm, 2.2–4.7mm; body ring number: 47–52 (+1–2 apodous+T). Females usually longer, higher and more brightly colored than males. Developmental stadia (identified after Enghoff et al. (1993)): adult males: VIII–XII (exceptionally XIII); adult females: IX–XIII (exceptionally XIV).
2 frontal, 4 supralabral and 19–28 labral setae. Ratio of 5th and 4th antennal articles: 5:4=1–1.1 (Fig. 6h). Form of gnathochilarium as on Fig. 6i. Metazonital setation considerably dense. Preanal process long and pointed, sometimes curved upwards (Fig. 6j). Anal valves densely setose. Subanal scale trapezoid, never pointed; apically mostly with 2–3 teeth (Fig. 6k). Preanal process blunter, subanal scale more rounded in females.
Males: Gnathal lobe well-developed. First leg-pair in the form of a simple hook (Fig. 6l). Penis flattened from antero-posteriad, apically with very short lobes and with two small, wide, (usually) rounded lamellae running parallel (ll for left and rl for right lamella) and pointing posteriad in some species—these lamellae sometimes not visible. Penis occasionally bent along its longitudinal axis (e.g. in M. montivagum (Verhoeff, 1901) , see Figs 11f and 11f View FIGURES 11 a – g *). Second pair of legs with two pads on postfemur and tibia (pf and t, respectively), coxa simple, without hump (Fig. 6n). All subsequent legs with three pads on femur (f), postfemur (pf) and tibia (t) (Fig. 6m). Pads on postfemur and tibia sometimes rippled (as if additional pads would exist on the same article) (see pf on Fig. 6m), or with vestigial proximal parts (i.e. only the distal part visible). Tarsus of midbody legs shorter than tibia: (tarsus/ tibia)=0.65–0.9, although an unusual individual with ratio 1.15 was also found. 7th pleurotergum protruding like a simple shovel on the border of pro- and metazonum.
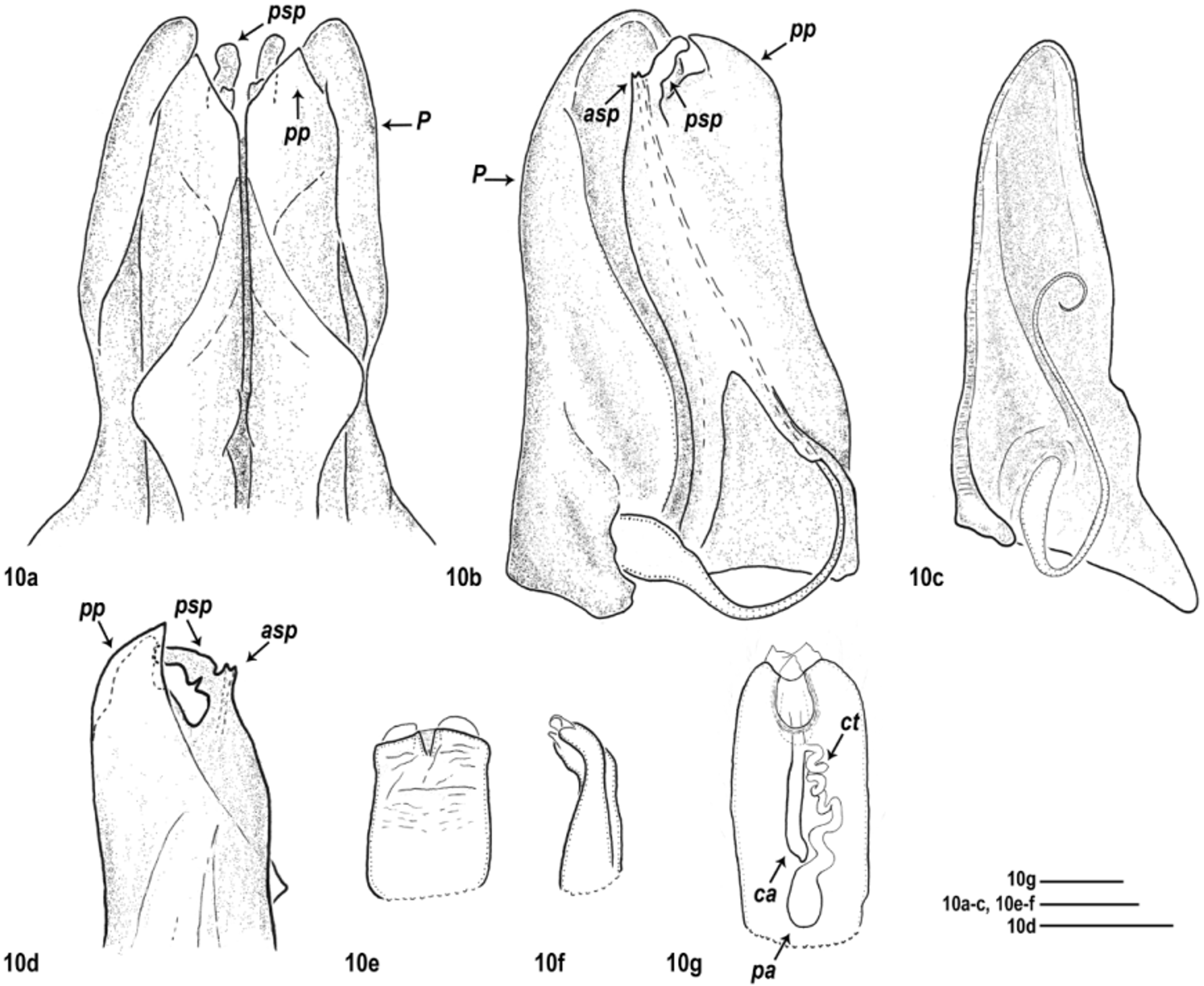
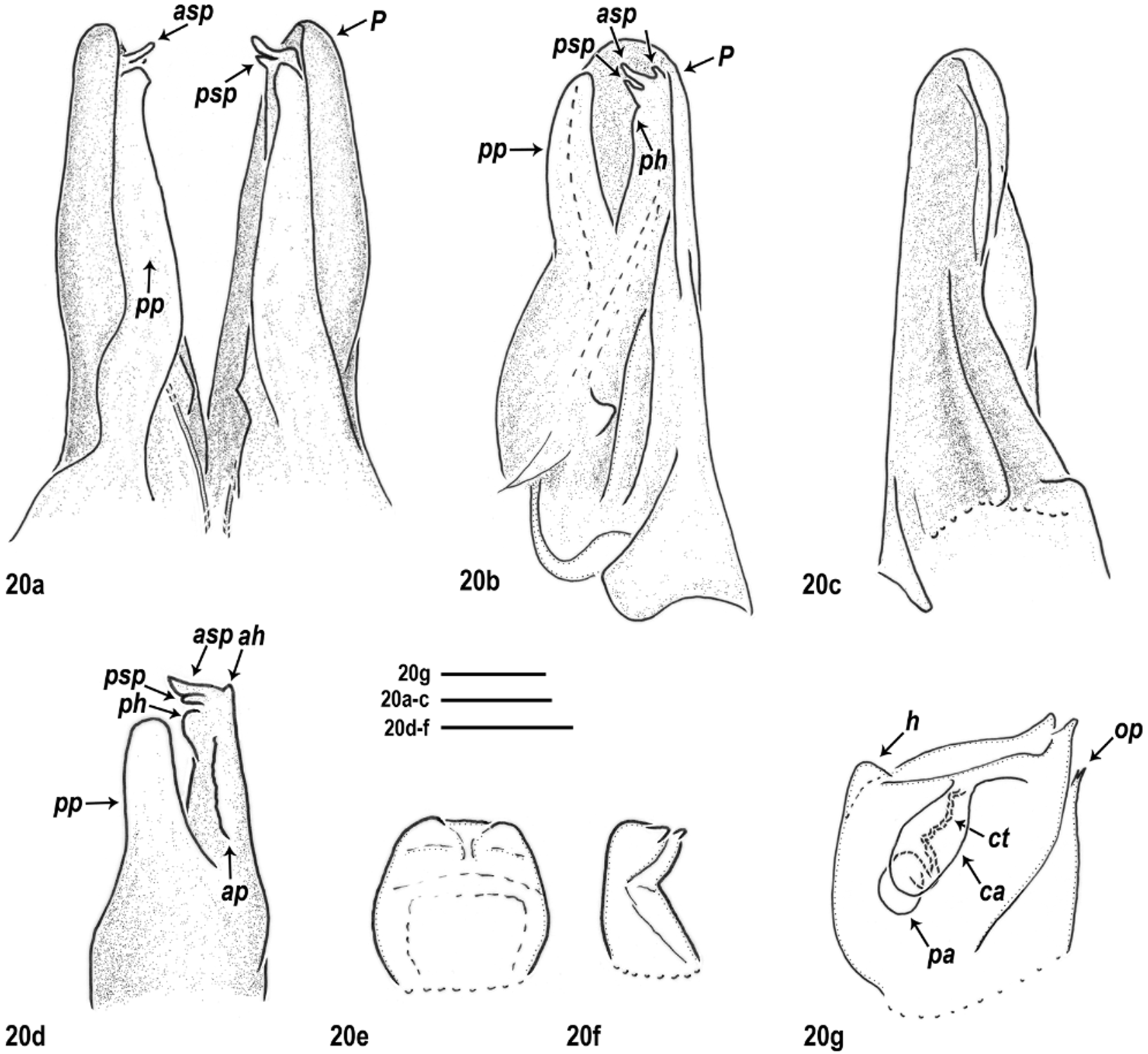
Male gonopods: Promere (P) very large, latero-anteriorly embracing the opisthomere. Connection between promeres due only to basal, transversal muscle fibres, and to a basal chitinous node between their mesal posterior margins, i.e. no additional connecting structure present. Promere bases relatively (compared to other congeners) well-developed, roundly protruding laterally. Posterior surface of the promere with an oblique ridge (r) forming a groove (g); and in some species an apical pit (“Fenestra”, i.e. window; see e.g. Verhoeff 1897b)—as an imprint of the opisthomere’s anterior process. Flagellum long and thin.
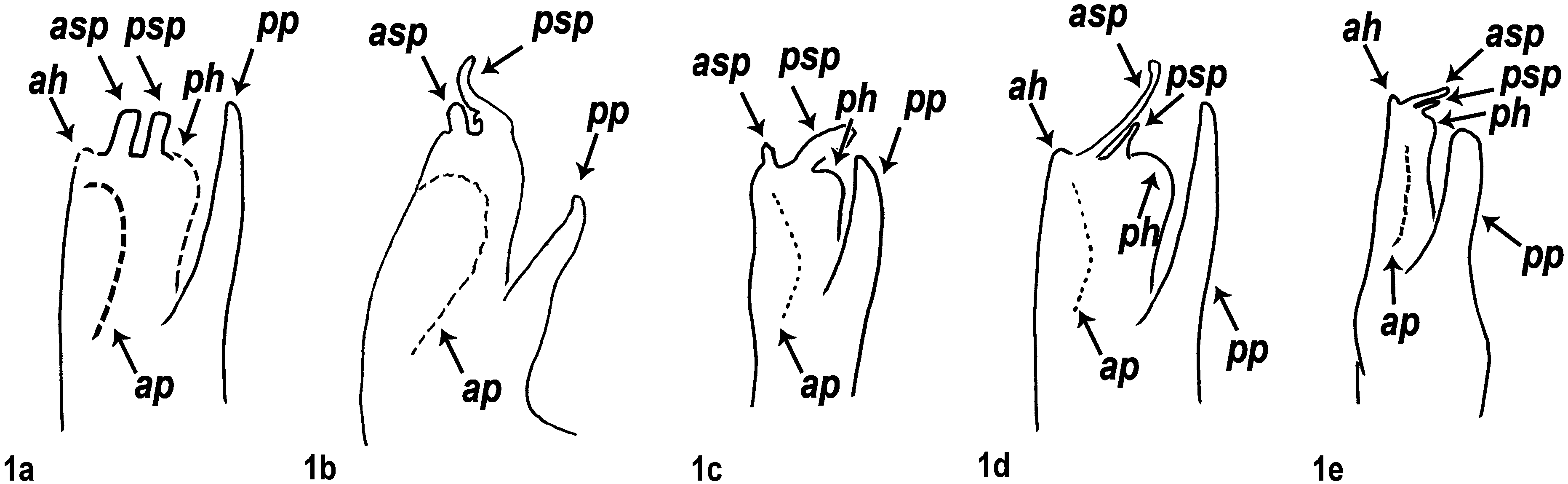
All species groups with a basic arrangement of opisthomere parts ( Figs 1b–1e View FIGURES 1 a – e ), while shape of opisthomere parts different within the actual species groups. Basically ( Fig. 1a View FIGURES 1 a – e ): opisthomere with a well-developed posterior process (pp) in every species group—most likely this was called “ Paracoxit ” or “ Coxalfortsatz ” (i.e. coxal process) by Attems (1927 and 1940, respectively) and all the other parts of the opisthomere were probably gathered under the name “Telopodit” (Attems 1927: fig. 287.). Opisthomere usually also with an anterior process or anterior ridge in same position (ap). Occasionally the apical part of the opisthomere is in the form of a hump around the solenomere, around the end of the flagellum-conducting lamella, posteriorly and/or anteriorly variably developed (ah for anterior hump and ph for posterior hump)—probably this hump was called “ Polster ” (i.e. pillow or pad, see e.g. Attems 1927, 1940). Solenomere always with an anterior and a posterior process (asp and psp) displaying several shapes and a great variation in length within species groups and species; sometimes even intraspecifically. Anterior solenomere process tubular (see Figs 9–11d View FIGURES 9 a – g View FIGURES 10 a – g View FIGURES 11 a – g , 15d View FIGURES 15 a – g )—this may be the sperm channel, i.e. “ Spermarinne ” by Lohmander (1936, p. 107) while the opening for the flagellum is basal to the anterior solenomere process (see Figs 6p, 8d).
Gonopod complex compact, in situ not informative. Thus, complete dissection of gonopods needed for species identification, e.g. at least separating right from left gonopods, sometimes also promere from opisthomere. Connection between promere–opisthomere stronger than between promere–promere or opisthomere–opisthomere. Opisthomere lying in the groove (g) of the promere. Solenomere sometimes exceeding the promere apically (e.g. M. rosenauense (Verhoeff, 1897)). The opisthomere’s posterior process (pp) never exceeding the promere apically.
Females: First two leg-pairs of adult females slightly swollen. Vulvae deeply hidden in vulval sacs, cylindrical and apically rounded, with distinct apical margin. Apical opening short; usually not, or only slightly exceeding the apical margin. Operculum (op) positioned latero-anteriorly, shorter than bursa (see e.g. Fig. 20g View FIGURES 20 a – g ). Bursa with a double receptaculum seminis consisting of two ampullae: central ampulla (ca) subequally wide along its whole length, opening to the surface in the apical opening; posterior ampulla (pa) drop-shaped, connected to the central one through a significantly more narrow spiral connecting tube (ct), (except for M. montivagum and M. carniolense (Verhoeff, 1897) , with gradually widening connecting tube into the posterior ampulla, without defined border).
Remarks. True sexual dimorphism in body coloration is detected in three species: M. projectum Verhoeff, 1894 , M. rosenauense (Verhoeff 1897a) and M. silvaticum . Females of these species have two bright longitudinal bands dorsally on a brownish ground, while males are dark grey with a black longitudinal line medially. Other species show three colour types: (1) two longitudinal bright bands on brownish or dark grey ground (e.g. M. dentatum (Verhoeff, 1898))—in these cases females are usually slightly more brightly colored; (2) one medial longitudinal yellowish or reddish band/dot-line (e.g. M. unilineatum (C. L. Koch, 1838)) on dark grey ground; (3) brownish body without any bands (e.g. M. rhodopinum (Verhoeff, 1928) , M. montivagum ).
1. The Megaphyllum projectum — species group Fig. 1b View FIGURES 1 a – e
Posterior opisthomere process (pp) occasionally rudimentary (in M. silvaticum ); most species with anterior process (ap) (except for M. erythronotum (Latzel, 1884)) . No apical hump, neither anteriorly nor posteriorly. Posterior solenomere process (psp) cockscomb-like, with a hook facing the anterior solenomere process (asp) (seen mostly from mesal view)—called “ Flagellumhäkchen ”, i.e. flagellal hook, by Verhoeff (1907). Anterior solenomere process (asp) usually shorter, mostly quite wide and easily discernible.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |