Argulus chiropteroideus, Tavares, 2017
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4320.3.3 |
|
publication LSID |
lsid:zoobank.org:pub:D883949F-9Aae-4596-81Ae-445C40B07Bbe |
|
DOI |
https://doi.org/10.5281/zenodo.6009456 |
|
persistent identifier |
https://treatment.plazi.org/id/03CB87CA-FFC4-A004-FF59-9BEB957C3C63 |
|
treatment provided by |
Plazi |
|
scientific name |
Argulus chiropteroideus |
| status |
sp. nov. |
Argulus chiropteroideus sp. n.
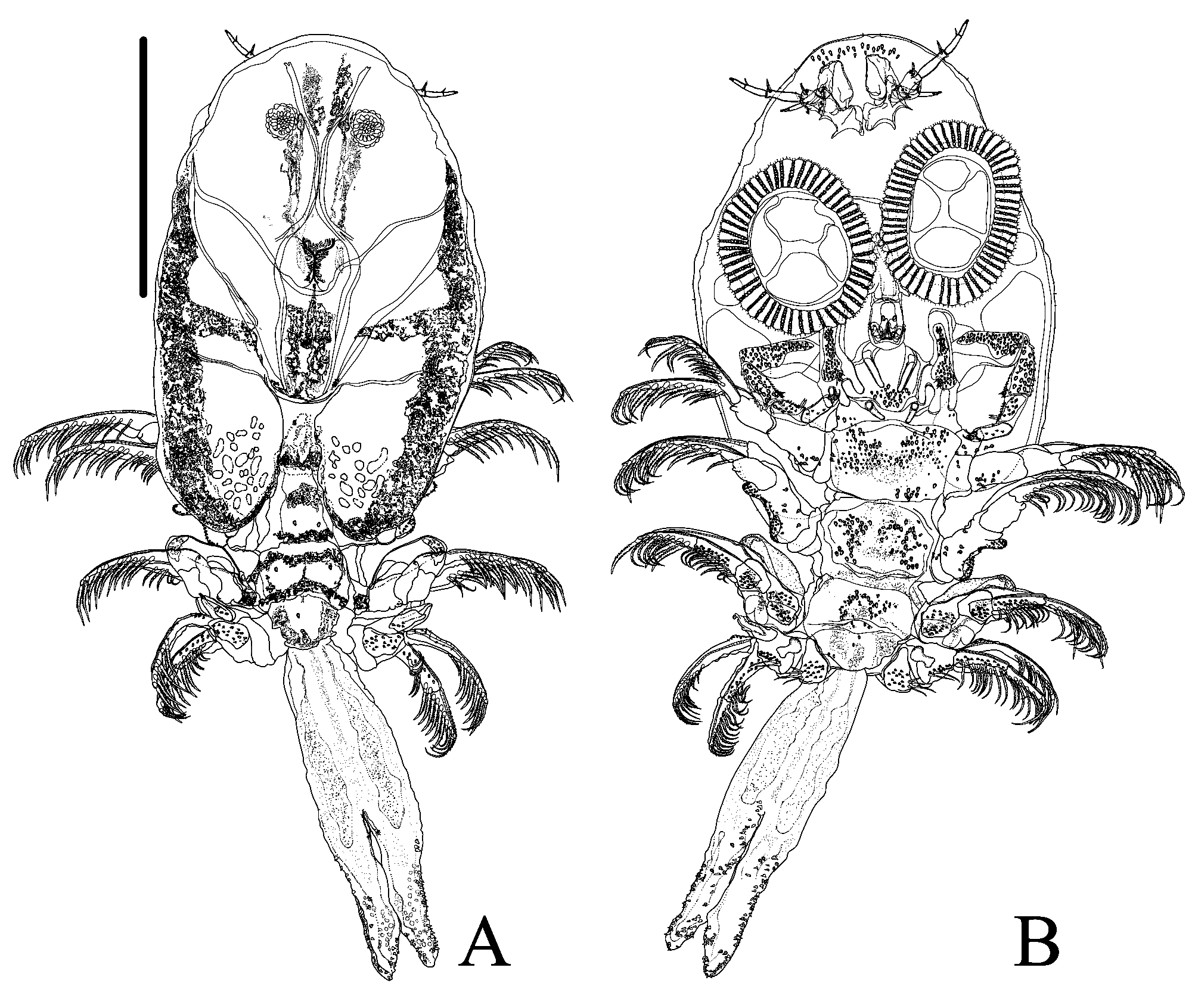
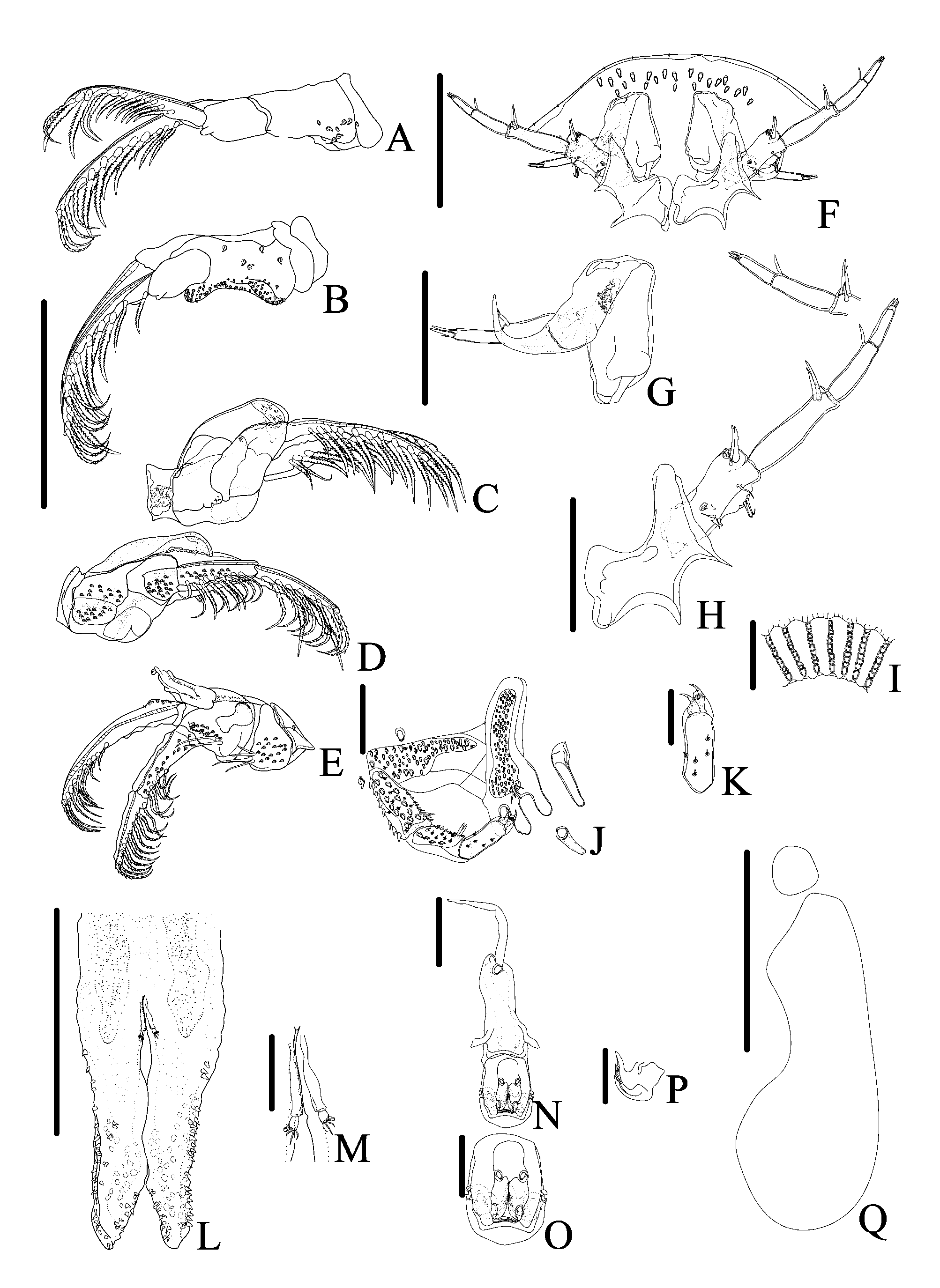
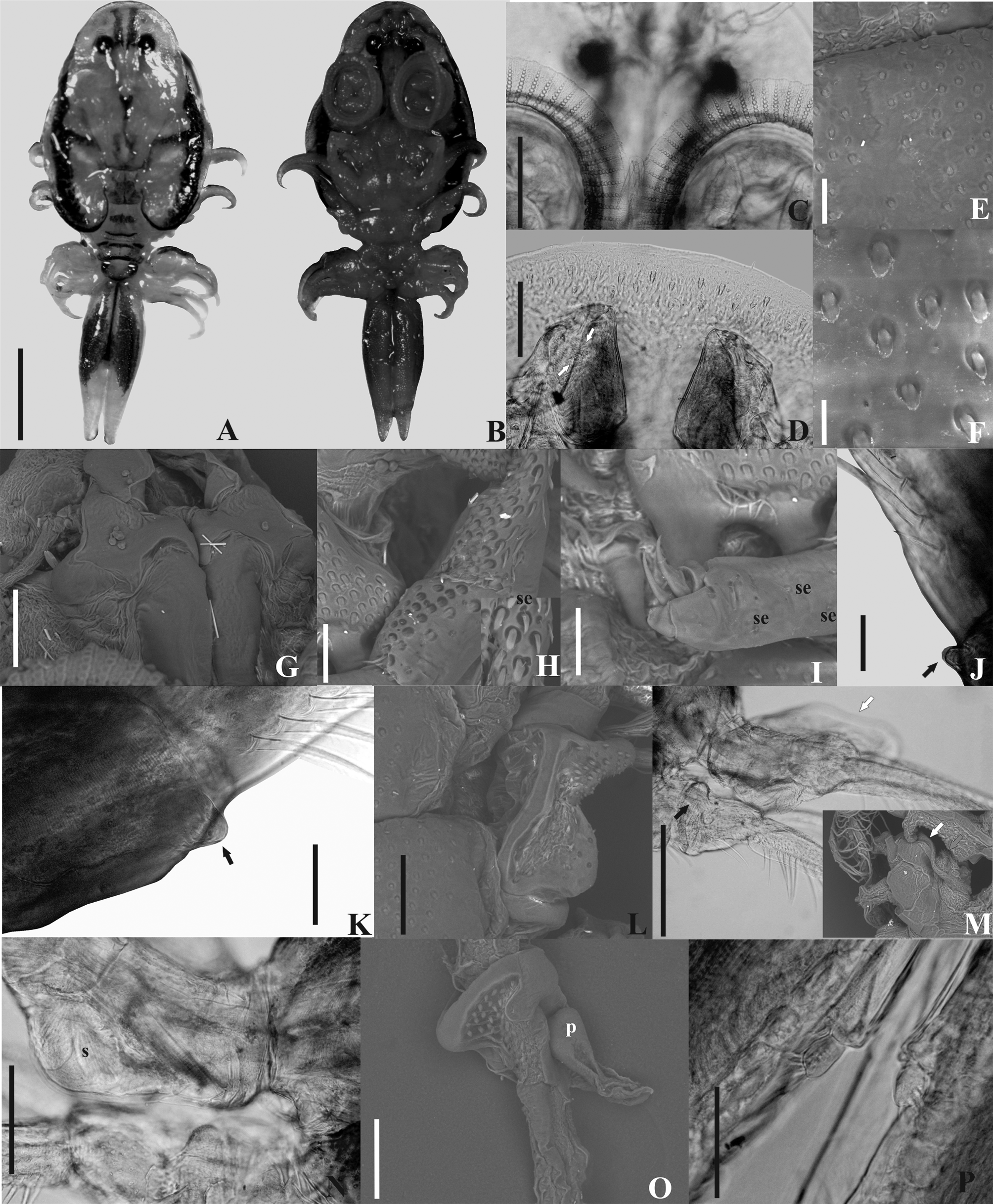
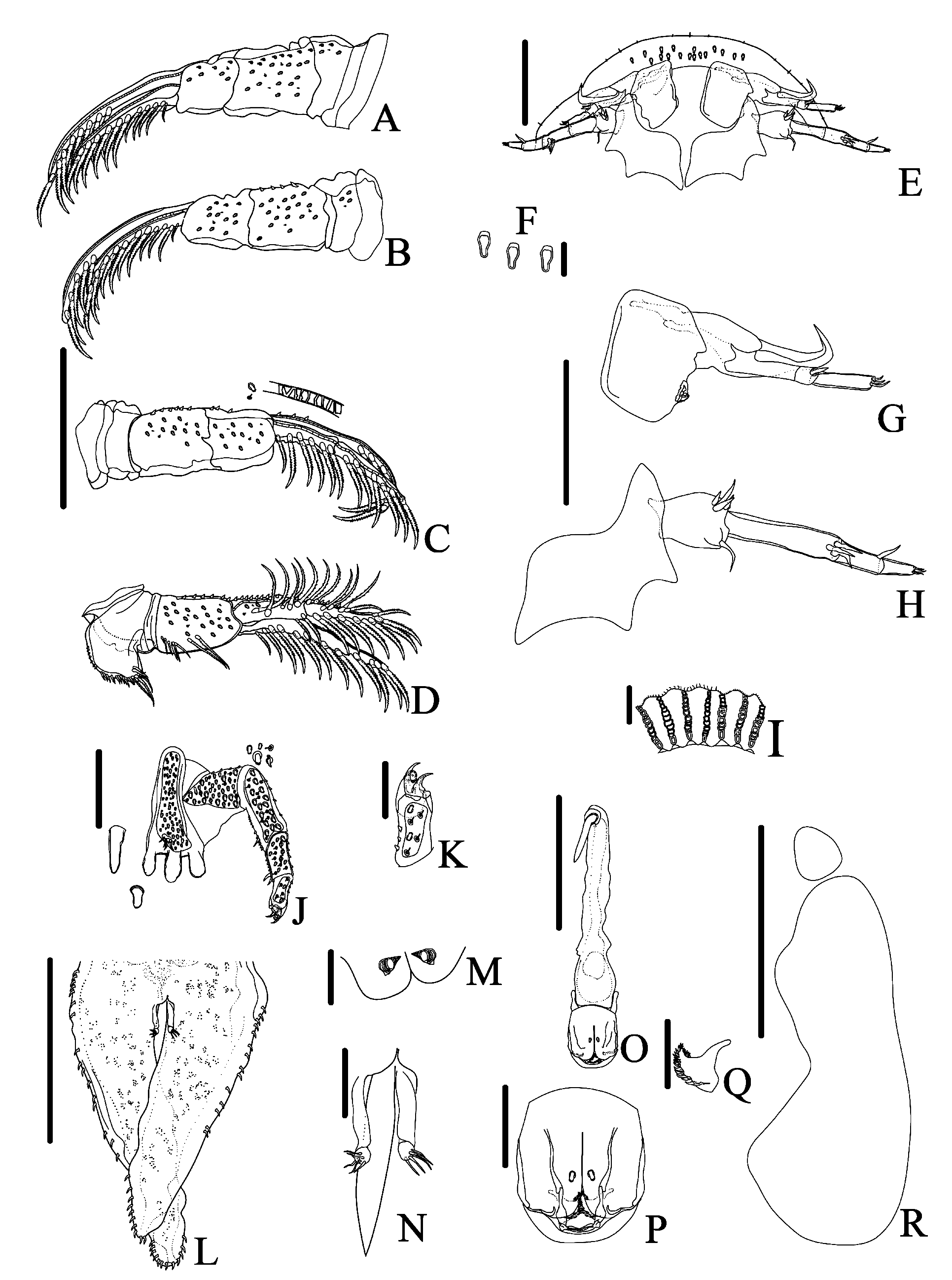
( Figures 1–6 View FIGURE 1 View FIGURE 2 View FIGURE 3 View FIGURE 4 View FIGURE 5 View FIGURE 6 )
Holotype, type host and type-locality. 1 adult male total length (mm) 4.7, in ethanol 70%, ZUEC-CRU 3521 , Pseudoplatystoma reticulatum , skin, Cuiabá River , Parque Nacional do Pantanal Matogrossense, Mato Grosso, Brazil (17°49′37.77″ S, 57°22′53.38”W), 8 July 2009. GoogleMaps
Allotype, type host and type-locality. 1 adult female total length (mm) 8.9, in ethanol 70%, ZUEC-CRU 3522 , Pseudoplatystoma reticulatum , skin, Cuiabá River , Parque Nacional do Pantanal Matogrossense, Mato Grosso, Brazil (17°49′37.77″ S, 57°22′53.38”W), 8 July 2009. GoogleMaps
Other hosts and localities. 6 Paratypes: 2 adult females INPA 2316, 2305, 2 adult females MZUSP 36229, in ethanol 70%, 1 adult male INPA 2306 View Materials , in etanol 70%, 1 imature male INPA 2304 View Materials , in Hoyer’s medium, 8 July 2009, Pseudoplatystoma reticulatum , skin, Cuiabá River Cuiabá River, Parque Nacional do Pantanal Matogrossense, Mato Grosso, Brazil (17°49′37.77″ S, 57°22′53.38”W) GoogleMaps . 5 vouchers: 1 adult female ZUEC-CRU 3520 , in ethanol 70%, 8 July 2008, Pseudoplatystoma corruscans , skin, Cuiabá River , Parque Nacional do Pantanal Matogrossense, Mato Grosso, Brazil (17°49′37.77″ S, 57°22′53.38”W) GoogleMaps ; 1 adult male INPA 2307 View Materials , 1 View Materials imature male ZUEC-CRU 3501 , 1 adult female INPA 2308 View Materials , in Hoyer’s medium, and 1 adult female ZUEC-CRU 3519 , in ethanol 70%, 3 June 2004, Pseudoplatystoma corruscans , gills, Paraguay River , Corumbá, Mato Grosso do Sul, Brazil (18°59'53.17"S, 57°36'44.43"O) GoogleMaps .
Material examined. Argulus chicomendesi . Holotype: 1 female (INPA 677). Paratypes: INPA 769, 1 female, 1 male; INPA 685, 4 females; INPA 678, 1 female; INPA 680, 2 males; INPA 681 3 females; INPA 768, 8 males, 8 females; INPA 764, 2 females, 1 male; INPA 765 14 females, 3 males; INPA 766, 2 females; INPA 769a–i, 6 females, 3 males. Voucher: INPA 1948, 1 female. INPA 1961 is not deposited in the collection.
Etymology. The name of the described species, Argulus chiropteroideus sp. n., is a latinised adjective derived from the mammalian order Chiroptera (containing the bats), which is composed by chiro (from Greek kheir), meaning hand + ptera (from Greek pteron) that means wing. It refers to the bat-wing shape of the postantennal spines of the described species.
Diagnosis. Females larger than males. Carapace longer than wide, pyriform, short, with naked setae evenly dispersed around perimeter; not reaching or reaching the anterior edge of the second pair of legs on females and on young males; on adult males it covers the second legs; about two series of inconspicuous spines with rounded tip on frontal region of the ventral surface of the carapace parallel to the anterior border of the cephalothorax. Respiratory area composed of an anterior area, spherical in males and pyriform in females, and a posterior oblong area, which has a constriction in the middle, with the posterior region larger than the anterior; caudal rami, pedunculated, nearly basal in anal sinus; postantennal spines single, with two lateral points, and one point anteriorly, resembling wings of bats. Males support 54–64 rods per suction cup; females support 56–64 rods per suction cup; 8–12 sclerites per rod in males and 7–13 in females, which decrease in number on medial-posterior portion, defined by the rim of suction cup. Basal plates of second maxilla with three claviform, digitate spines; scales arising from the pad and spam distally; 5 stout, naked setae on posterior margin of pad. Legs without flagella; coxa of second leg of male with posteroventrally bilobate structure, which bear several spines; third leg of male with paddle-like extension issuing anteroventrally from the dorsal side of the coxa, extending over bases; a fleshy lobe dorsally; and a bubble-like area ("socket") on posterior of base; fourth leg of male with a peg issuing anterodorsally from base; a ventrally bone-shaped fleshy lobe and 2 naked setae at base, natatory lobe with 4 naked setae; fourth leg of female with 2 plumose setae and 6 naked setae at the base and 2 plumose and about 20 stout setae at natatory lobe, which decrease in size medially.
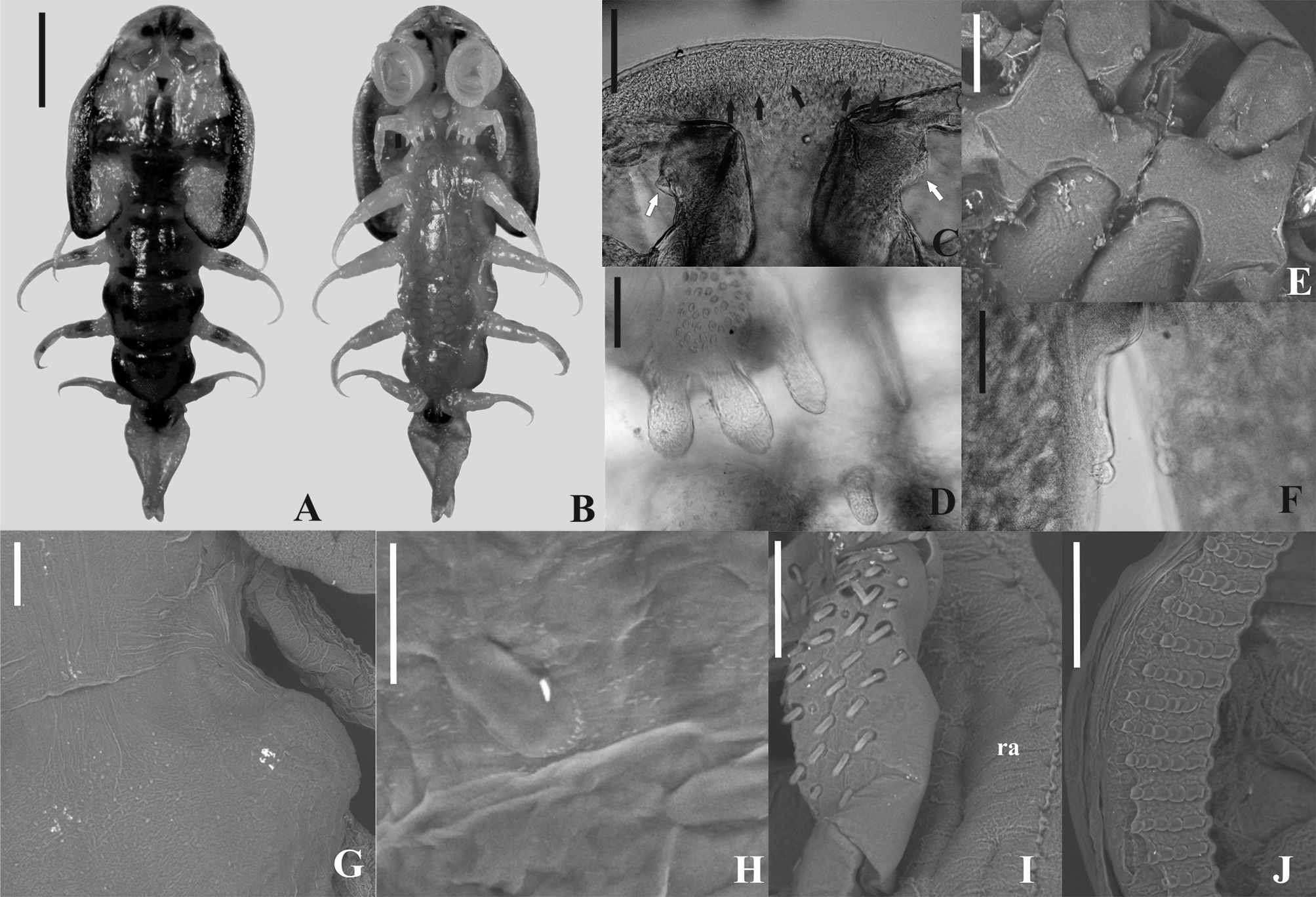
Description. Based on 5 males and 8 females. Females larger than males. Total length in males, 5.5 (4.6–5.9), in females, 8.4 (5–11). Carapace longer than wide, pyriform, short, with naked setae evenly dispersed around it; not reaching or reaching the anterior edge of the second pair of legs on females and in young males; on adult males it covers the second legs ( Figs. 1 View FIGURE 1 , 3A–B View FIGURE 3 , 4 View FIGURE 4 , 6 A–B View FIGURE 6 ). Carapace length in males, 2.3 (1.9–2.5), in females, 4.3 (2.5–53); maximum carapace width in males, 2 (1.6–2.5), and in females 3.8 (2.2–5.4). Carapace posterior sinus length in males, 0.7 (0.5–0.9, n = 3), in females, 1.6 (0.4–2.7); width in males, 0.6 (0.2–0.8), and in females, 1.4 (0.5–2.3). Cephalothorax length in males, 1.8 (1.7–2, n = 4), in females, 2.7 (2–3.6); anterior width in males, 1.2 (1–1.4, n = 4), and in females, 2.6 (1.5–3.7). Cephalothorax with ventral spines, rounded on tip on frontal region, area between the antenna lacking spines ( Figs. 2F View FIGURE 2 , 3D View FIGURE 3 , 5E–F View FIGURE 5 , 6C View FIGURE 6 ). Respiratory area composed of an anterior oval area situated close to the oblong posterior area, which has an anterior and a posterior portion largest ( Figs. 2Q View FIGURE 2 , 5R View FIGURE 5 ). Females without eggs in carapace alae.
Pair of compound eyes with diameters (µm) in males, 202 (165–235, n = 3) left and 215 (202–226, n = 3) right; and in females, 237 (183–275) left, 232 (183–275) right. Transverse distance between eyes (µm) in males, 339 (263–472, n = 3), and in females 424 (325–525). Nauplius eye with one anterior and two posterior ocelli, pigment cell between ocelli forming a dark "Y". Sclerotized dorsal ridges arise from the posterior groove that delimit the cephalothorax, move forward, and undergoes a lateral separation at the level of the nauplius eye, where a small lateral projection arises, it undergo a medial approximation, and it reaches the anterior region of the cephalothorax, opening laterally at level of the compound eyes. In the distal third of the antero-lateral grooves, there is in females, on each side, a medially directed groove, which bifurcates and ends in an elevation of the cephalothorax. In the male there is a posteromedial groove that unites below the nauplius eye.
Thorax dorsoventrally compressed, 4-segmented with two pairs of posteriorly-projecting spines ventrally. The anterior pair (accessory spines) larger than posterior pair (postmaxillary spines); posterior pair composed by digitate spines in males and pyriform in females ( Figs. 2J View FIGURE 2 , 5J View FIGURE 5 , 6D View FIGURE 6 ). Accessory spines situated between basal segments of second maxillae; postmaxillary spines positioned farther apart than accessory spines. Thorax covered with scattered sensilla and coarse and fine-pectinate scales on ventral and dorsal surface ( Figs. 3E–F View FIGURE 3 , 6G–H View FIGURE 6 ).
Four pairs of biramous swimming legs composed of a precoxa, coxa, basis, exopod, and endopod; with plumose setae on all exopods and endopods. Coxae of all legs of male and female with scattered sensilla and coarse-pectinate and spine-like scales. First two pairs of legs each without a recurved flagellum on dorsal surface. Only the endopod of third and fourth pair of swimming legs are two-segmented, all others endopods and exopods unsegmented ( Figs. 2D–E View FIGURE 2 , 3J–L View FIGURE 3 ). First and second legs of male with a ventral papillae at distal posterior region of bases; coxae of second legs of male with a bilobate structure posteroventrally, with scales on lobes distally ( Figs. 2A–B View FIGURE 2 , 3J–K View FIGURE 3 ). Third legs of male with paddle-like extension issuing anterodistally from the coxae, extending over base ( Figs. 2D View FIGURE 2 , 3M View FIGURE 3 ); a bubble-like area ("socket") ( Figs. 2D View FIGURE 2 , 3N View FIGURE 3 ) on posterior of bases with opening of socket ventrally; and a dorsal fleshy lobe tipped with spine–like scales from joint between coxa and basis ( Figs. 2C View FIGURE 2 , 3M View FIGURE 3 ). Fourth legs of males with a peg issuing anterodorsally from base; a bone-shaped lobe from joint between coxa and basis ventrally ( Figs. 2E View FIGURE 2 , 3M, 3O View FIGURE 3 ). Coxae of fourth legs of males and females with pyriform and a boot shaped posterior natatory lobes, respectively, fringed with plumose setae ( Figs. 2E View FIGURE 2 , 5D View FIGURE 5 ).
Abdomen bilobate, with pair of caudal rami, pedunculated, nearly basal in anal sinus ( Figs. 2L–M View FIGURE 2 , 3P View FIGURE 3 , 5L–M View FIGURE 5 , 6F View FIGURE 6 ). Abdomen length in males, 1.9 (1.7–2), in females, 2.3 (1.5–2.6); maximum width in males, 0.7 (0.5–0.9), and in females, 1 (0.7–1.5). Anal sinus length in males, 1.2 (1–2), and in females, 1.4 (0.9–1.6). Small, spine-like scales along most of edge and on ventral and dorsal of abdomen. Each caudal ramus with five stout, naked setae. Paired spermathecae of female brownish, round to oval, located anteriorly on abdomen; a pair of testis one lobed occupy half of abdomen of male.
First antennae 4-segmented: first segment (basal segment) sclerotized, large without stout posteriorlyprojecting posterior spine, but with a lateral sensorial structure ( Figs. 2G View FIGURE 2 , 3D View FIGURE 3 , 5G View FIGURE 5 , 6C View FIGURE 6 ). Second segment with a large recurved terminal spine. Third segment fleshy, cylindrical and smaller with stout seta distally that projects ventrally, one in males and two in females. Fourth segment fleshy, smaller than third segment, with four distal setae in males and three setae in females. Second antennae with 4 segments (3 fleshy segments) ( Figs. 1B View FIGURE 1 , 2H View FIGURE 2 , 4B View FIGURE 4 , 5H View FIGURE 5 ). Basal segment larger than others, in males, bearing four stout setae and one 2-setulate papillae proximally and, distally with 3 naked setae posteriorly and, 2 stout setae, one 3-setulate papillae and one 2-setulate papillae anteriorly; and in females, bearing one posteriorly directed naked setae and four anteriorly directed stout setae. Second segment in males and females, bearing 3 distal naked setae. Third segment in males, with 1 proximal and 1 distal naked setae; in females bearing 1 distal naked setae. Fourth segment with 3 distal naked setae in females and males. Postantennal spines single, with two lateral points, and one anterior point, resembling wings of bats ( Figs. 1B View FIGURE 1 , 2H View FIGURE 2 , 3C, G View FIGURE 3 , 4B View FIGURE 4 , 5H View FIGURE 5 and 6E View FIGURE 6 ).
First maxillae modified into suction cups in adults. In males, first maxilla outer diameter 0.9 (0.8–1) left, (0.8– 2.8) right 1.2, and inner diameter 0.6 (0.4–0.7) left, 0.6 (0.5–0.7) right. In females, outer diameter 1.2 (0.7–1.5) left, 1.2 (0.8–1.5) right, and inner diameter 0.8 (0.5–1) left, 0.8 (0.6–1) right. Number of support rods per suction cup 54–64 in males and 56–64 in females. Number of sclerites per support rod 8–12 (n = 587 rods from 5 specimens) in males and 7–13 (n = 968 rods from 8 specimens) in females. Shape of sclerites variable resembling a drop; proximal sclerite slender and longer, medial sclerites larger and, distal sclerites the smallest ( Figs. 2I View FIGURE 2 , 3 View FIGURE 3 CF, 5I, 6J). The shape of sclerites does not vary with the position on the rim, but the number decrease in the medial-posterior portion defined by the rim of suction cup. Second maxillae five-segmented with broad basal plate bearing three claviform, digitate spines ( Figs. 2J View FIGURE 2 , 5J View FIGURE 5 , 6D View FIGURE 6 ). The two outer spines closer than the median spine and the innermost one; median spine slightly larger than remaining two spines; basal plate with elevated pad bearing five stout, simple setae; scattered sensilla, coarse–pectinate, fine–pectinate and spine–like scales clustered in distinct patches on ventral surfaces of all five segments; one stout simple setae on third segment and two on fourth segment posterodistally ( Figs. 3H View FIGURE 3 , 6I View FIGURE 6 ). Fifth segment with two sharp claws and a rounded elongate lobe positioned behind the claws ( Figs. 2J–2K View FIGURE 2 , 3I View FIGURE 3 , 5J–K View FIGURE 5 ).
Mouth tube short, lacking armature, reaching the base of thoracic accessory spines in males. Labium with two embedded scales ( Figs. 2N–O View FIGURE 2 , 5N–O View FIGURE 5 ). Pair of serrated mandibles inside mouth tube; in males, mandibles with one median process anteriorly directed and, only laterally serrated ( Fig. 2P View FIGURE 2 ); females lack the median process in mandibles, which is median, anterior, lateral and posteriorly margins serrated ( Fig. 5Q View FIGURE 5 ). Preoral stylet present ( Figs. 2N View FIGURE 2 , 3C View FIGURE 3 , 5O View FIGURE 5 ).
Remarks. In Argulus chiropteroideus sp. n. specimens, the carapace alae does not reach the third legs. In freshwater environments in South America, this characteristic is shared by adults of Argulus elongatus Heller, 1857 ; Argulus ichesi Bouvier, 1910 ; Argulus violaceus Thomsen, 1925 ; A. multicolor ; Argulus patagonicus Ringuelet, 1943 and A. chicomendesi ( Bouvier 1910; Heller 1857; Malta & Varella 2000; Ringuelet 1943; Stekhoven Jr. 1937; Thomsen 1925). The argulid described here is two times longer than it is wide and the carapace does not exceed the second legs; the carapace is ornamented with anteroventral spines, there are no spines on the lateroventral region; the second maxillae has 3 digitate spines at the basal plate; and legs 3 and 4 have 2-segmented endopods. All these characteristics are shared with A. chicomendesi ( Malta & Varella 2000), but A. multicolor also shares several of these character states (Lemos de Castro 1949, 1951; Stekhoven Jr. 1937).
A. chiropteroideus sp. n. differs from A. chicomendesi , but not from A. multicolor , due to the shape and distribution of spines in the frontal region of the ventral surface of the carapace. There are about two series of inconspicuous spines, which are parallel to the anterior border of cephalothorax in A. chiropteroideus sp. n. In A. chicomendesi , however, these spines are conspicuous and distributed in around five series that undergo a decrease in the number of spines posteriorly ( Malta & Varella 2000). There are multi-digit scales on the thorax, second maxillae and legs of A. chicomendesi and in second maxillae of A. multicolor , but this type of scale is absent in A. chiropteroideus sp. n.
The anterior and posterior spine are absent on the second segment of the first antenna of A. chiropteroideus sp. n. whereas they are present in A. chicomendesi ( Malta & Varella 2000), and there is one anterior and one posterior spine on the first segment and two posterior spines on the second segment of first antenna of A. multicolor . The single postantennal spines of A. chiropteroideus sp. n. is medially convex with two lateral tips and one anterior tip, resembling the wings of a bat, distinguished from A. chicomendesi , which has single, triangular postantennal spines ( Malta & Varella 2000), and from A. multicolor , where the spine has one lateral and one posterior tip.
The sclerites of the support rod in A. chiropteroideus sp. n. differ in shape and number from sclerites of A. chicomendesi and A. multicolor . Although in A. chiropteroideus sp. n. and A. chicomendesi the distal sclerites are cup-shaped, and the basal one, more elongated, there are 8–12 (males) and 7–13 (females) sclerites per support rod in the first maxillae of A. chiropteroideus sp. n., opposed to 6 sclerites per support rod in A. chicomendesi ( Malta & Varella 2000). Moreover, the basal sclerites of A. chiropteroideus sp. n. are shorter, and the remaining sclerites less robust, than those observed for A. chicomendesi . In A. multicolor , the morphology of the sclerites are quite different from the aforementioned species, where the sclerites are small, curved transverse lines (Lemos de Castro 1949, 1951; Stekhoven Jr. 1937).
The second maxilla, which has three claviform, digitate spines in A. chiropteroideus sp. n. also distinguishes it from A. chicomendesi and from A. multicolor . A. chicomendesi has three conical, short spines ( Malta & Varella 2000), whereas A. multicolor have no spines at the base of the second maxilla (Lemos de Castro 1949, 1951; Stekhoven Jr. 1937)
Legs 1 and 2 are devoid of a dorsal flagellum in A. chiropteroideus sp. n., while this structure is present in A. chicomendesi ( Malta & Varella 2000) and in A. multicolor (Lemos de Castro 1949, 1951; Stekhoven Jr. 1937). Additionally, the male of A. chiropteroideus sp. n. has a posteroventral bilobed structure at the coxae of the second legs, a structure that is absent in A. chicomendesi ( Malta & Varella 2000) and in A. multicolor (Lemos de Castro 1951) . Instead, A. chicomendesi has three set of conspicuous spines, one in each of the pre-coxae, coxae and base. Males of A. chiropteroideus sp. n. has a paddle-like extension issuing anterodistally from the coxae of the third leg and dorsally, a fleshy lobe tipped with spine–like scales, which are also absent in A. chicomendesi and A. multicolor . The fleshy bone-shaped lobe, placed ventrally between the coxa and the base of the fourth leg of A. chiropteroideus sp. n. is also absent in A. chicomendesi and A. multicolor . Finally, the morphology of the pegs of the three species is quite different. In A. chiropteroideus sp. n. it is elongated, whereas it is rounded in A. chicomendesi and A. multicolor .
On average, the biometrical values of A. chiropteroideus sp. n., A. chicomendesi and A. multicolor are different. The females (8.4mm) and males (5.5mm) of A. chiropteroideus sp. n. are larger than females (4.7 mm) and males (3.1mm) of A. chicomendesi , though it be smaller than a female of A. multicolor examined here (11.3mm) and those females (9.4) examined by Lemos de Castro (1949). The same was observed for the length and width of the carapace and for length and width of abdomen.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
SubClass |
Branchiura |
|
Order |
|
|
Family |
|
|
Genus |