Argulus nattereri Heller, 1857
|
publication ID |
https://doi.org/10.11646/zootaxa.4320.3.3 |
|
publication LSID |
lsid:zoobank.org:pub:D883949F-9Aae-4596-81Ae-445C40B07Bbe |
|
DOI |
https://doi.org/10.5281/zenodo.6009464 |
|
persistent identifier |
https://treatment.plazi.org/id/03CB87CA-FFCD-A003-FF59-98E0920C3BAE |
|
treatment provided by |
Plazi |
|
scientific name |
Argulus nattereri Heller, 1857 |
| status |
|
Argulus nattereri Heller, 1857
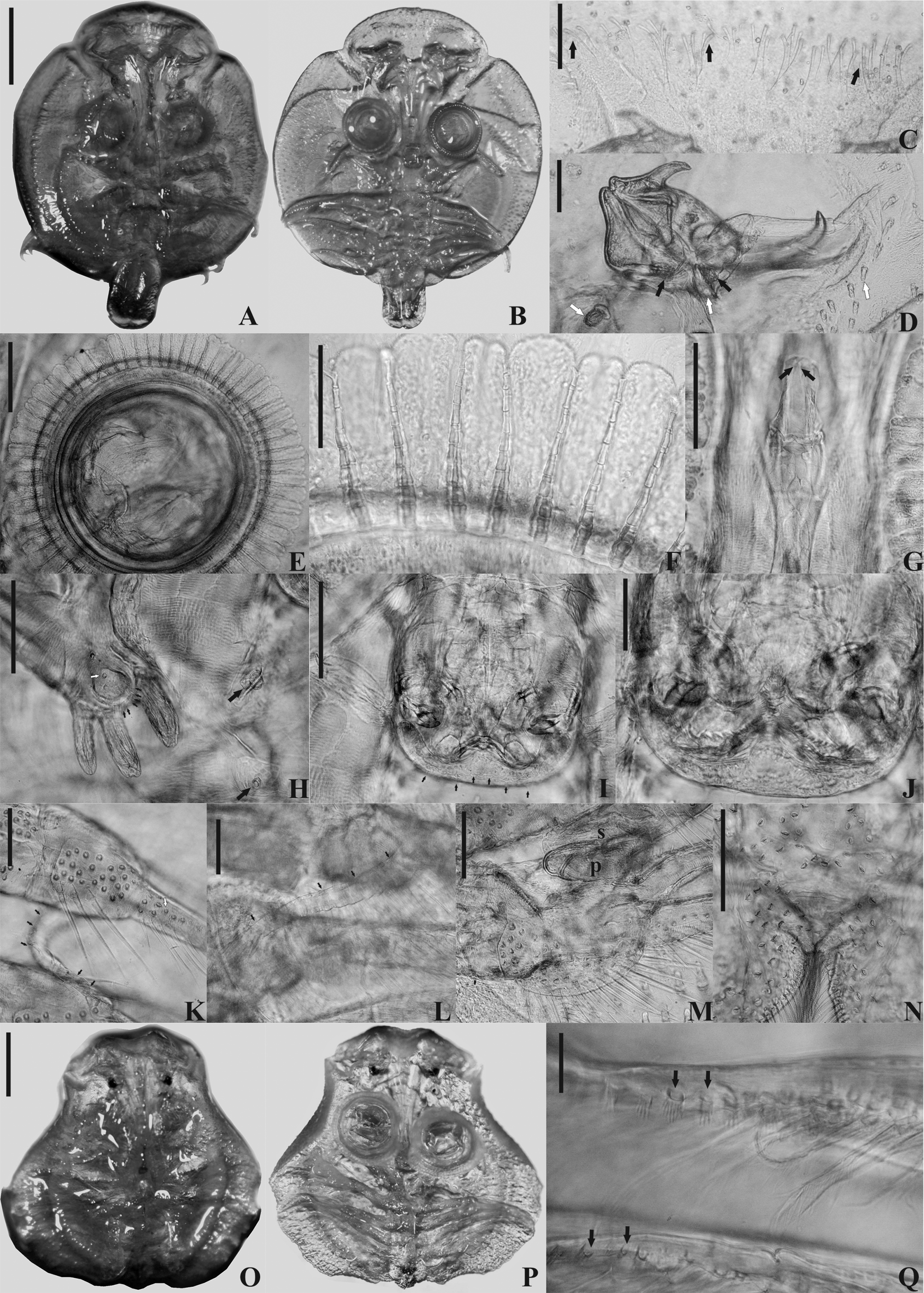
( Figure 8 View FIGURE 8 )
A. nattereri : HELLER (1857): 103 –106, PL. II, fIGS. 4–12; KRøyER (1863): 23 –25, 29, PL. I, fIGS. 3A–D; THORELL (1864): 60; WILSON (1902): 720, PL. XXII, fIG. 63; THIELE (1904): 23 –25, fIGS. 43–52; MOREIRA (1913): 8, PL. II, fIG. 1; Argulus silvestrii LAHILLE, 1926: LAHILLE (1926): 10 –11, 16, fIG. 3; Argulus salminei KRøyER, 1863: PART MEEHEAN (1940): 502; A. nattereri : RINGUELET (1943): 57 –62, PL. III, fIGS. 1–6; WILSON (1944): 552, 573–574, PL. 24, fIGS. 74–78; LEMOS DE CASTRO (1985): 10, fIGS, 11– 13; THATcHER (2006): 393, 395, 398, fIG. 9.5
Type host. Hydrocyon brevidens (= Salminus brasiliensis [Cuvier 1816]) ( Heller 1857)
Type locality. Undefined location in Brazil ( Heller 1857).
Other hosts and localities. Salminus brevidens (= S. brasiliensis ), state of Mato Grosso, Brazil ( Moreira 1912, 1913); P. corruscans , S. brevidens (= S. brasiliensis ), Parana River in Zona de San Pedro, Argentina ( Ringuelet 1943); Pimelodus albicans (Valenciennes, 1840) , P. corruscans, Ysoró Stream , municipality of Cuatro Bocas, province of Corrientes, and in the Parana River, province of Entre Ríos, both in Argentina ( Barzanti 1976; Chemes & Takemoto 2011); P. tigrinum and Pseudoplatystoma fasciatum (Linnaeus, 1766) , Beni and Ichilo Rivers Bolívia, ( Mamani et al. 2004).
Material deposited. 3 vouchers: 1 adult female ZUEC-CRU 3516 , in ethanol 70%, 3 June 2004, Salminus brasiliensis , skin, Paraguay River, Corumbá-MS, Brazil ( 18°59'53.17"S, 57°36'44.43"O) GoogleMaps ; 1 adult male ZUEC- CRU 3517, in ethanol 70%, and 1 adult male INPA 2307 View Materials , in Hoyer’s medium, 4 July 2008, skin, Pseudoplatystoma corruscans, Cuiabá River, Parque Nacional do Pantanal Matogrossense ( 17°49′37.77″ S, 57°22′53.38″ W) GoogleMaps .
Measurements. Based on 2 males and 1 female. Total length (mm) 4.54 (3.7–5.3) in males, 4.5 in females. Carapace length 4.55 (2.6–4.5) in males, 4.6 in female; maximum carapace width 3.5 (2.6––4.4) in males, 4.6 in female. Carapace posterior sinus length 1.3 (1.2–1.35) in males, 1.1 in female; width 1 (0.9–1.1) in males, 1.2 in female. Cephalothorax length 2.2 (1.8–2.6) in males, 2.5 in female; anterior width 2.1 (1.7–2.5) in males, 2.5 in female. Abdomen length 0.9 (0.8–1) in males, 0.5 in female; width 0.7 (0.6–0.75) in males, 0.5 in female. Abdomen posterior sinus length 0.2 (0.15–0.3) in males, 0.13 in female; width 0.13 (0.11–0.15) in males, 0.04 in female. Eyes diameter (µm) 175 left and 225 right in one male, 150 left and 182 right in female; transverse distance between eyes (µm) 800 in one male, 800 in female. First maxillae outer diameter (µm) 837 left (675–1000) and 899.4 right (724–1075) in males, 1300 left and 1250 right in female; inner diameter 650 left (525–775) and 616 right (508–725) in males, 1075 left and 975 right in females. Number of support rods in suction cups 64–70 in males, 66–69 in female; number of sclerites per support rod 6–9 in males, 7–9 in female.
Remarks. Among the species of Argulus from freshwater environments of South America, only A. nattereri ( Figs. 8A–B View FIGURE 8 ) and Argulus juparanaensis Lemos de Castro, 1950 have the carapace alae covering all four legs, although males of A. pestifer also display this characteristic ( Heller 1857; Lemos de Castro 1950; Ringuelet 1943), as do females of A. salminei according to Meehean (1940) but not according to Ringuelet (1943).
The morphology of the sclerites of rods of the suction cups of A. nattereri and A. pestifer , is similar, although as well as being smaller in A. nattereri , there is variation in numbers, with 8–10 in specimens of A. nattereri ( Figs. 8E–F View FIGURE 8 ) and just two in A. pestifer ( Fig. 9E View FIGURE 9 ). Specimens of A. nattereri exhibit a strong chitin plate from the dorsal to the second antennae, which narrows and runs laterally and forwards ( Thiele 1904) ( Fig. 8D View FIGURE 8 ). This structure is also present in A. pestifer , although in this species these plates are also in the frontal and median region of the cephalothorax ( Ringuelet 1948). The preoral stylet, which was observed in its inverted state ( Fig. 8G View FIGURE 8 ), and the presence of flagellum on the first two pairs of legs ( Figs. 8K–L View FIGURE 8 ) in both males and females, differentiates A. nattereri from A. pestifer . The presence of ventral spines along the length of the carapace alae ( Figs. 8M, 8D View FIGURE 8 ) is also an important diagnostic characteristic of A. nattereri , as these spines are absent in A. pestifer ( Ringuelet 1948) and are limited to the frontal region, more specifically to the anterolateral sinuses, in A. juparanaensis (Lemos de Castro 1950) . Additionally, the morphology of the antenna, antennules, postantennal spines, and the robust and acute spine, which emerges from the carapace towards the basal segment of the antennae ( Fig. 8D View FIGURE 8 ), the socket on the 3rd leg and the peg on the 4th legs of males ( Fig. 8M View FIGURE 8 ) allow identification of A. nattereri . Nevertheless, A. nattereri is distinguished from all species above by the presence of naked setae regularly scattered at the dorsal side of cephalothorax and carapace alae ( Figs. 8A, 8C View FIGURE 8 ), structures which are absent in A. salminei , A. pestifer and A. juparanaensis ( Heller 1857; Lemos de Castro 1950; Ringuelet 1943, 1948; Thiele 1904).
We observed some morphological differences in our specimens of A. nattereri comparing with the description of other authors. The morphology of the carapace alae of the female was in accordance with Heller (1857) ( Figs. 8O–P View FIGURE 8 ) but differed from Wilson (1944), because the posterior lobes of the carapace did not overlap. The number of sclerites of the suction cup was lower, with 6–9 sclerites per support rod in males and 7–9 in females, whereas 9–10 ( Ringuelet 1943) and 14–16 ( Wilson 1944), have been previously recorded. Coarse-pectinate scales were observed by Thiele (1904), but the sensilla on the mouth tube ( Figs. 8I, J View FIGURE 8 ), similar to those previously observed in Argulus yucatanus Poly, 2005 from Mexico ( Poly 2005), were also observed in the A. nattereri of the present study. We observed, as indicated by Ringuelet (1943), two pairs of small, thick and blunt accessory spines ( Fig. 8H View FIGURE 8 ), although Wilson (1944) has stated that this structure was missing for this species. We also observed that the endopods and exopods of the third and fourth legs were of similar size, rather than the longer exopods viewed by Ringuelet (1943). We observed smaller endopods only on the first and second legs, in accordance with Thiele (1904), although we did not see the 3-segmented extension of the first endopod, as per Ringuelet (1943). A finger-like papillae, resembling the tactile papillae of some females such as those of Argulus americanus Wilson, 1902 ( Wilson 1904) , was observed rooted at the base of the natatory lobes of the fourth leg of the male of A. nattereri ( Fig. 8N View FIGURE 8 ). While tactile papillae are situated on either side of the opening of the oviduct between the bases of the posterior legs of females and are not found in males, we observed a differently located structure. Ventrally, erect coarse pectinate scales cover the thorax, abdomen and legs of A. nattereri ( Fig. 8Q View FIGURE 8 ).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
SubClass |
Branchiura |
|
Order |
|
|
Family |
|
|
Genus |
Argulus nattereri Heller, 1857
| Tavares, Luiz E. R. 2017 |
A. nattereri
| THATcHER 2006: 393 |
| WILSON 1944: 552 |
| RINGUELET 1943: 57 |
| MEEHEAN 1940: 502 |
| LAHILLE 1926: 10 |
| MOREIRA 1913: 8 |
| THIELE 1904: 23 |
| WILSON 1902: 720 |
| THORELL 1864: 60 |
| KRoyER 1863: 23 |
| HELLER 1857: 103 |