Taphozous longimanus, Hardwicke, 1825
|
publication ID |
https://doi.org/10.5281/zenodo.3740269 |
|
DOI |
https://doi.org/10.5281/zenodo.3810753 |
|
persistent identifier |
https://treatment.plazi.org/id/03D587F2-FFCB-4C00-FF1C-3147FAC6EFA6 |
|
treatment provided by |
Plazi |
|
scientific name |
Taphozous longimanus |
| status |
|
6 View On . Long-winged Sheath-tailed Bat
Taphozous longimanus View in CoL
French: Taphien à longs bras / German: Langflügel-Grabfledermaus / Spanish: Tafozo alilargo
Other common names: Indian Sheath-tailed Bat, Long-winged Tomb Bat
Taxonomy. Taphosous [sic] longimanus Hardwicke, 1825 ,
“ Calcutta,” Bengal , India.
Taphozous longimanus is in the subgenus Taphozous . Four subspecies recognized.
Subspecies and Distribution.
. l. longimanus Hardwicke, 1825 - Indian subcontinent (Nepal, India, Bangladesh, and Sri Lanka).
. l albipinnis Thomas, 1898 - Borneo and Labuan I.
. l. kampenii Jentink, 1907 —Java, but distribution is not clearly delimited.
. l. leucopleurus Dobson, 1878 - Flores I, Lesser Sundas, but distribution is not clearly delimited.
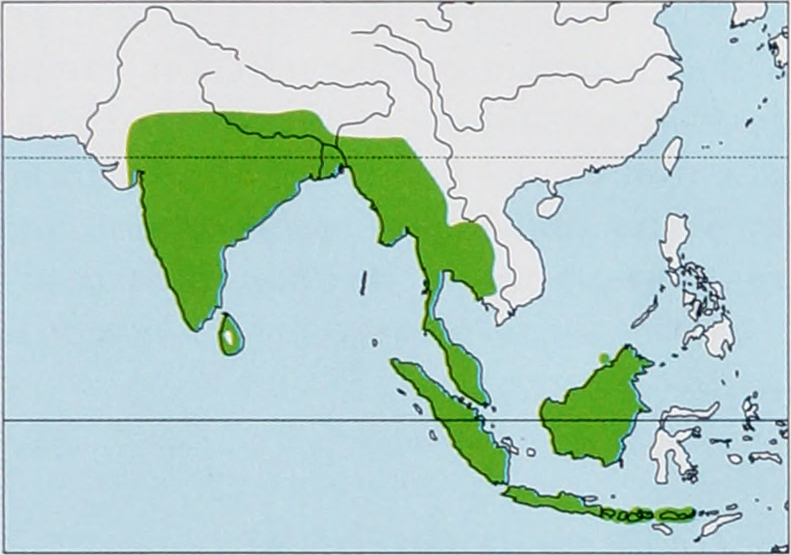
Wide distribution from South Asia and Indochina to Greater and Lesser Sundas, but subspecies to which many of these populations belong are unknown. View Figure
Descriptive notes. Head-body 73-86 mm, tail 20-30 mm, ear c.20 mm, forearm 54— 63 mm. No specific data are available for hindfoot length or body weight. Dorsum of the Long-winged Sheath-tailed Bat varies from reddish brown to black, sometimes speckled with white patches. In Thailand, females tend to be gray, and males tend to be brown. Chin is naked. Legs are covered with short fur. Upper surface of wing and uropatagium membranes are furred to about one-half the length of humerus and femur. A well-developed gular pouch and radio-metacarpal pouches are present in males; females have only rudimentary fold on throat. Wings are long, narrow, and whitish and attached at ankles. Tail tapers to narrow tip. Third finger is longer than forearm.
Habitat. Wide variety of habitats from arid areas to humid zones from sea level to elevations of C.1200 m.
Food and Feeding. Diet includes beetles and cockroaches.
Breeding. Long-winged Sheath-tailed Bats are bom in midJanuary and mid-May. At Varanasi, India, spermatogenesis occurs in September, with appearance of spermatozoa. Spermatogenic arrest occurs in winter dormancy in December and is followed by a second peak of spermatogenesis inJanuary that coincides with ovulation in females. A third peak of spermatogenesis begins in late April, and tubules are found completely regressed after May. Cycle of accessory sex glands generally parallels testicular cycle, reaching maximum hypertrophy in September-January and again in late April. Thereafter, accessory sex glands are involuted. Spermatozoa are present in cauda epididymis throughout the year.
Activity patterns. The Long-winged Sheath-tailed Bat roosts in caves, tunnels, abandoned human structures (e.g. forts, dungeons, and wells), and hollows and crowns of trees including palms and banyan figs { Ficus benghalensis , Moraceae ) and under large rocks and eaves of houses. It emerges from roosts very early in the evening and flies fast as it forages.
Movements, Home range and Social organization. The Long-winged Sheath-tailed Bat roosts alone or in colonies that can number hundreds of individuals.
Status and Conservation. Classified as Least Concern on The IUCN Red List. The Longwinged Sheath-tailed Bat has a large distribution and presumably large and stable overall population, occurs in protected reserves, and is tolerant of habitat modification. Although populations generally are thought to be stable, some scattered local populations might be decreasing. In South Asia, the Long-winged Sheath-tailed Bat occurs in Hazaribagh Wildlife Sanctuary inJharkhand and Kanha National Park in Madhya Pradesh, and it likely is present in most protected areas in peninsular India and many in South-east Asia. Additional studies are needed on taxonomy, distribution, abundance and population trends, reproduction, and ecology. Some populations might be threatened by roost disturbance, especially from guano mining in South-east Asia.
Bibliography. Bates & Harrison (1997), Bates, Harrison & Muni (1994a, 1994b, 1994c), Brasset (1963), Khan (2001), Molur et al. (2002), Singh (1997), Srinivasulu & Srinivasulu (2005, 2012).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
SubFamily |
Taphozoninae |
|
Genus |