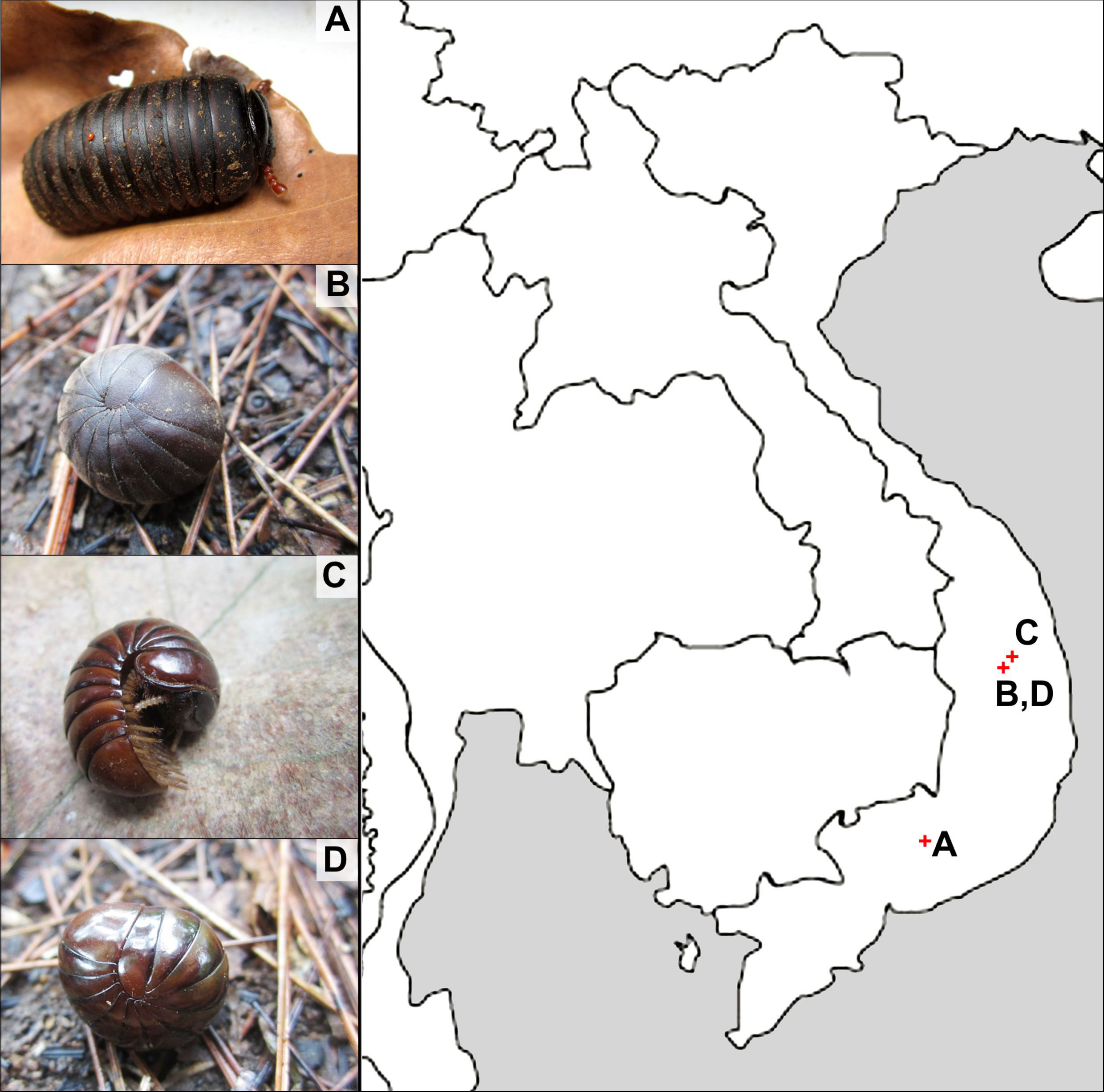
Sphaerobelum cattiense Semenyuk, Golovatch and Wesener, 2018
|
publication ID |
https://doi.org/10.11646/zootaxa.4459.3.7 |
|
publication LSID |
lsid:zoobank.org:pub:8CA301B7-A24A-4CB1-8231-FF0BAFF175DA |
|
DOI |
https://doi.org/10.5281/zenodo.6495776 |
|
persistent identifier |
https://treatment.plazi.org/id/03D7EC02-FFDA-2424-CBBD-E057DC7AFBC1 |
|
treatment provided by |
Plazi |
|
scientific name |
Sphaerobelum cattiense Semenyuk, Golovatch and Wesener |
| status |
sp. nov. |
Sphaerobelum cattiense Semenyuk, Golovatch and Wesener View in CoL , sp. nov.
Figs 1A View FIGURE 1 , 2 View FIGURE 2 , 3 View FIGURE 3 , 4A View FIGURE 4
Material examined: Type specimens. Holotype male (ZMUM), Vietnam, Dong Nai Province, Cat Tien National Park, monsoon lowland tropical forest (N 11°26' E 107°21', 100–200 m. a.s.l.), litter, floor and under logs, 22– 24.VI.2011, leg. I.I. Semenyuk. Paratypes: 1 males, 2 females (ZMUM), same data, together with holotype; 1 male, 1 female, 1 ex. of undetermined sex (ZFMK MYR 6516, 6519, 6565, same data as holotype, but collected in VI.2017). Other material. 2 males, 2 females, 1 immature (topotypes), ZFMK MYR 6566, Vietnam, Dong Nai Province, Cat Tien National Park, Lagerstroemia calyculata and Afzelia xylocarpa forest, VIII.2011, leg. E. Galoyan.
Diagnosis. Medium-sized, 25–30 mm long and 15–17 mm wide, dark olive-brown Sphaerobelum with a microsetose surface and orange antennae and appendages. Modified femur with a dentate mesal margin ( Fig. 2D View FIGURE 2 ), a character shared only with S. bicorne and S. konkakinhense sp. nov. Differing from both other congeners in the vulval operculum which lacks a recessed apical margin and projects in a single, well-rounded, mesal tip ( Fig. 2I View FIGURE 2 ). Immovable finger of posterior telopod swollen apically, but extending only mesally ( Figs 2E, F View FIGURE 2 ). Also differing from S. bicorne and S. konkakinhense sp. nov. in the absence of a lateral projection on the prefemur of the walking legs.
Description. Measurements: Holotype male. Body length ca 30 mm, width of thoracic shield = 14.6 mm, of tergite 9 = 15.2 mm (= broadest), height of thoracic shield = 8.5 mm, of tergite 9 = 9.3 mm (= highest). Male paratypes ca 25 or 29 mm long, maximal width ca 17 or 15 mm, respectively. Females: length ca 27.5–30 mm, width of thoracic shield = 14.3–16 mm, of tergite 9 = 15.3–17 mm (=broadest), height of thoracic shield = 8.6–9.0 mm, of tergite 9 = 9.3–10 mm (= highest). Coloration: in live specimens, ventral side including head and all tergites blackish brown; collum, tergites and posterior part of anal shield brown. Thoracic shield brown at margins. Legs and antennae dark orange. In preserved specimens, dark olive to olive brown with contrasting, narrow, darkish, orange bands at caudal margins of all tergites ( Fig. 1A View FIGURE 1 ). Head dark brown to dark olive-brown. Antennae and legs contrasting bright orange. Head: wide and short, partly micropunctate. Clypeolabral region very densely setose. Occipital region finely and confusedly striolate and with a row of longer hairs near caudal edge. Eyes with>65 ocelli. Aberrant ocellus located inside antennal groove. Antennae short, with rounded joints, extending posteriorly to leg-pair 2. First antennomere with cuticular scales ( Fig. 3A View FIGURE 3 ). Lengths of antennomeres: 1>2=3=4=5<<6 ( Fig. 3A View FIGURE 3 ). Antennomere 6 densely pubescent, sensilla basiconica surrounding apical disc. Shape of antennae sexually dimorphic: cylindrical in female; thickened, almost axe-shaped in male ( Fig. 3B View FIGURE 3 ). Apical disc with ca 105/109 (male) ( Figs 2H View FIGURE 2 , 3B View FIGURE 3 ) or 56–61 (female) apical cones. Apical cones typical of Diplopoda ( Fig. 3C View FIGURE 3 ). Organ of Tömösváry located inside antennal groove. Gnathochilarium: structure typical of the order. Stipites and paramentum bearing several long setae. Mandibles (not examined). Stigmatic plates: first stigmatic plate triangular, apex well-rounded, weakly curved towards coxa 1. Laterotergites: laterotergite 1 slightly projecting, well-rounded and slender at tip. Collum with 2–3 rows of sparse setae near caudal margin and a few short, isolated setae on surface. Thoracic shield: surface like that of tergites. Shallow grooves beset with numerous long setae, sloping towards groove with 5 keels. Tergites: surface very densely micropunctate, many of the punctures carrying a tiny seta. Tips of paratergites of midbody tergites projecting posteriorly. Endotergum: inner section lacking any large spines or setae. Middle area with a single row of large, sparse, elliptical, cuticular impressions. Apically, 2–3 dense rows of short marginal bristles, a few with their tips slightly protruding beyond tergal margin ( Fig. 4A View FIGURE 4 ). Bristles not smooth, but with numerous small spicules ( Fig. 4A View FIGURE 4 ). Anal shield: large and regularly rounded. Surface similar to that of tergites. Underside with a single, short, black, locking carina, the latter located close to last laterotergite. Legs: leg-pair 1 with 2 or 3, leg-pair 2 with 3 or 4, leg-pair 3 with 4–6 ventral spines. First two legpairs without an apical spine. Leg-pairs 4–21 with 7–10 ventral spines and one apical spine. On leg-pair 9, femur 1.5X, tarsus 3.4X longer than wide ( Fig. 2D View FIGURE 2 ). All podomeres densely setose. Coxa with a large and well-rounded process. Prefemur of usual shape, but at apical margin with a mesal projection. Femur extended mesally into a dentate margin featuring 6–8 teeth.
Female sexual characters: vulva large, covering 2/3 of coxa, extending mesally to half of prefemur ( Fig. 2I View FIGURE 2 ). Operculum rounded, very slightly invaginated medially, mesal margin strongly projecting into a well-rounded lobe twice as high as remaining operculum. Subanal plate: large, shaped like a wide, well-rounded triangle, in some females with a slight invagination at the highest point of apical margin, creating two wide, well-rounded lobes.
Male sexual characters: gonopore covered with a single, undivided, triangular, sclerotized plate. Anterior telopods ( Figs 2A–C View FIGURE 2 ): consisting of a syncoxite plus 4 telopoditomeres. Telopoditomere 1 rectangular, as long as
wide. Telopoditomere 2 large, as long as telopoditomeres 3 and 4 combined. Process of telopoditomere 2 located posteriorly, not visible in anterior view. Process slender, projecting as far as base of telopoditomere 4, apical part well-rounded. Telopoditomere 3 shortest, as long as wide, anterior side with a few small, triangular spines, posterior side smooth. Telopoditomere 4 well-rounded, twice as long as wide, slightly tapering apically. Posterior aspect with 5 or 6 black spines, apical-most being the largest. All podomeres covered with long setae. Posterior telopods ( Figs 2E–G View FIGURE 2 ): telopoditomere 1 elongate, twice as long as wide. Immovable finger (process of telopoditomere 2) shorter than movable finger, consisting of telopoditomeres 3 and 4. Immovable finger with a characteristic, distally swollen apex, extending only mesally, apex therefore only slightly broader than base. Telopoditomere 3 elongate, towards immovable finger in apical half with a large extension carrying at least two large spines. Telopoditomere 4, 2.8X longer than wide, tapering apically, slightly curved towards immovable finger. At inner margin, telopoditomere 4 with a large spine at midpoint and 3 or 4 smaller spines apically. Entire telopodite on anterior side covered with long setae, except for apical part of telopoditomere 3 and telopoditomere 4. In posterior view, telopoditomere 1 clothed with numerous long setae, telopoditomere 2 only basally and at lateral margin clothed with setae, telopoditomere 3 with setae only at lateral margin.
Distribution and Ecology. This species is widespread exclusively in patches of mature, lowland, monsoon forest at elevations of about 120 m. a.s.l. Active animals prefer patches with less leaf litter and usually walk on the forest floor, sometimes digging into the leaf litter layer. Resting individuals were found in leaf litter, and very rarely in open places. Millipedes roll up at rest, being active both in daylight and in the dark. When disturbed, and having rolled up, animals unroll a few minutes later and typically do not roll up again from subsequent slight disturbances. The peak of abundance of adult animals is in June-July, at the onset of the rainy season, forming local swarming sites in some forest areas. Mating was noted during swarming time, and was observed infrequently. During the remaining parts of the year, individuals are more scarce, and show low population densities. Juveniles or oviposition places were not observed.
Name. Adjective, to emphasize Cat Tien National Park, its type locality ( Fig. 1 View FIGURE 1 ).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |