Prosorhynchus tonkinensis, Truong, Thuong Van, Palm, Harry W., Bui, Te Quang, Ngo, Huong Thi Thuy & Bray, Rodney A., 2016
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4170.1.3 |
|
publication LSID |
lsid:zoobank.org:pub:19D90117-0D96-4A96-9AD5-4EFD01A15269 |
|
DOI |
https://doi.org/10.5281/zenodo.5695070 |
|
persistent identifier |
https://treatment.plazi.org/id/03DC8339-9065-FFE8-FF3C-FE31FE2A78A0 |
|
treatment provided by |
Plazi |
|
scientific name |
Prosorhynchus tonkinensis |
| status |
sp. nov. |
Prosorhynchus tonkinensis View in CoL n. sp.
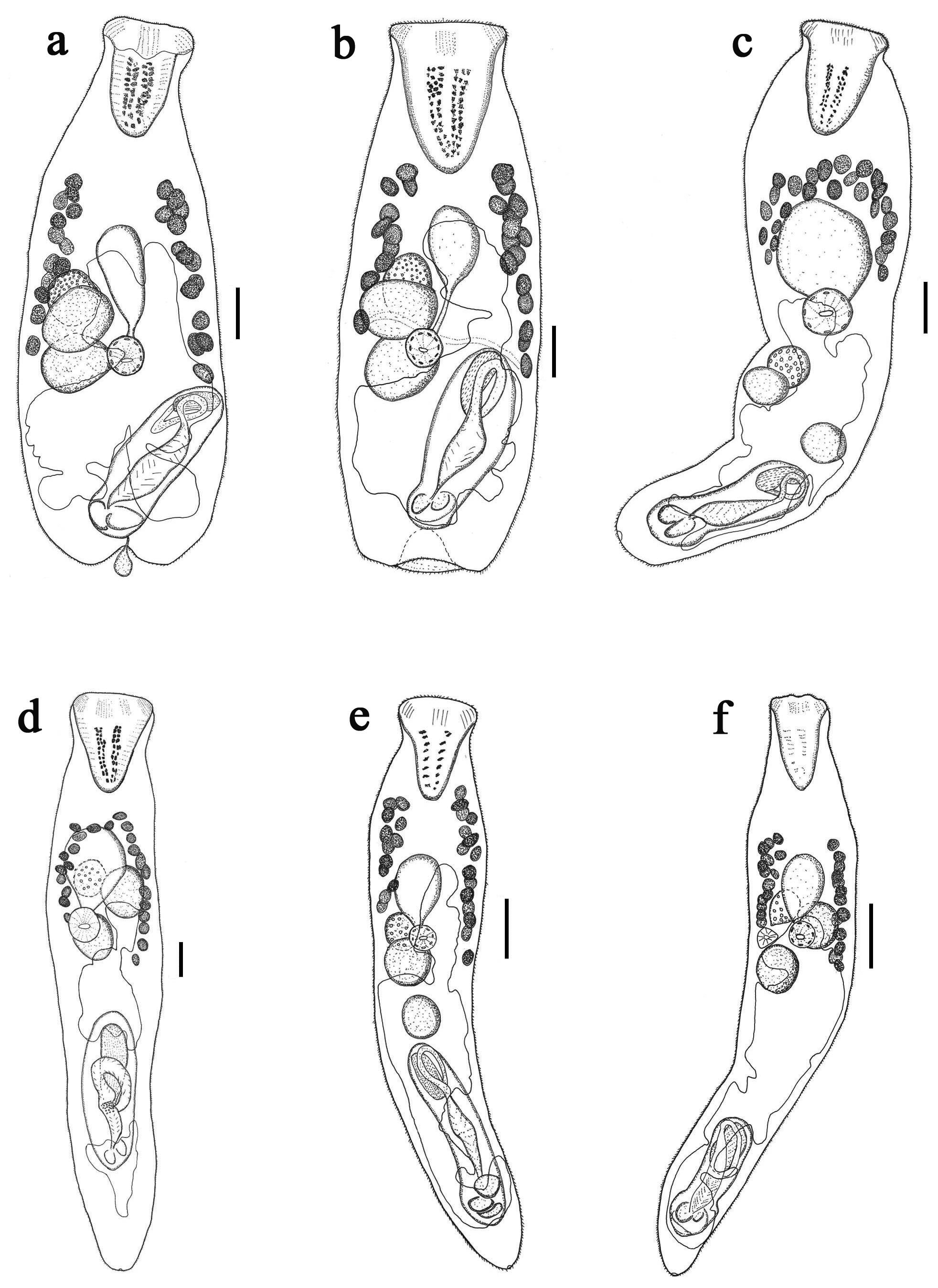
( Fig. 1 View FIGURE 1 a)
Type host. Epinephelus coioides (Hamilton, 1822) .
Site. Pyloric ceca, intestine.
Type-locality. Nghia Hung , Nam Dinh, Vietnam (19°59'18.5"N, 106°10'44.7"E, September 2013). GoogleMaps
Etymology. The species name refers to the location in the Gulf of Tonkin.
Description. Measurements and ratios based on 13 specimens are given in Table 1. Body pyriform, widest at two thirds of body, broadly rounded posteriorly, indented posterior extremity. Tegument spinous; spines squamous, tiny, reach to posterior extremity. Rhynchus large, muscular, conical, longer than wide. Mouth located at level of posterior testis or somewhat posterior to posterior testis, always inside posterior half of body. Pharynx spherical, in posterior half of body. Cecum tubular or oval sac like, extends anteriorly from pharynx.
Testes 2, subspherical, close together, normally contiguous or slightly separated, slightly diagonal, dextral in about midbody. Cirrus-sac elongate, muscular, not thick-walled, more or less parallel sided, reaching or overlapping posterior testis. Seminal vesicle elongate-oval, dextral in proximal portion of cirrus-sac. Pars prostatica long, in two distinct parts; proximal part narrow, curved proximally to form convoluted seminal duct, joins seminal vesicle at postero-dorsal aspect; distal part wider, straighter, surrounded by dense layer of gland-cells, lining of filaments in chevron arrangement. Ejaculatory duct narrow, complex genital lobe inside genital atrium. Genital atrium large. Genital pore distinctly separated from posterior end.
Ovary oval, anterior and/or overlapping anterior part of anterior testis. Uterus reaches anteriorly into vitelline region, extends posteriorly to genital pore. Eggs tanned, numerous. Vitellarium consists of two lateral fields of 12– 16 follicles, usually symmetrical, long vitelline field is dextral, anterior extremity closer to posterior margin of rhynchus than cecum, ovary or uterus; posterior extremity at about level of anterior testis. Excretory pore terminal; extent of excretory vesicle not seen.
Remarks. The character of the indented posterior extremity of Prosorhynchus tonkinensis n. sp. differs from all other species described so far in the genus Prosorhynchus . We also used the visual key to Prosorhynchus developed by Bray & Palm (2009) to setup a comparison diagram ( Fig. 2). This demonstrates that only two species have no black squares against their names, namely P. atlanticus and P. crucibulum (Rudolphi, 1819) .
Prosorhynchus atlanticus View in CoL was recorded from the Black Grouper Mycteroperca bonaci Poey View in CoL in the Atlantic Ocean by Manter (1940b). It was described with a uterus that is ‘almost never extending anterior to ovary (slightly so in 1 of 29 specimens). In P. tonkinensis View in CoL , the uterus always extends to the level of the ovary, sometimes anteriorly to it. Furthermore, as can be seen from table 2, P. atlanticus View in CoL can be distinguished from P. tonkinensis View in CoL in the body width as a percentage of the body length (about 27–33% compared to 34–62%) and in the premouth percentage (44–49% vs 50–80%). Prosorhynchus crucibulum View in CoL was re-described from Conger conger View in CoL from the north Atlantic Ocean by Matthews (1973). It can be distinguished from P. tonkinensis View in CoL by its arched vitellarium and smaller egg size (24–26 × 16–18 vs 29–35 × 20–22). They have been recorded from widely different hosts and geographical regions.
Figure 2 shows that seven further species have only a single black square against the new species, namely P. australis View in CoL , P. congeri Yamaguti, 1970 View in CoL , P. djedaba Reimer, 1985 View in CoL , P. longisaccatus View in CoL , P. mcintoshi View in CoL , P. paracrucibulus Velasquez, 1959 View in CoL and P. rotundus Manter, 1940 View in CoL . Prosorhynchus australi s from the Brazilian Codling Urophycis brasiliensis (Kaup) ( Szidat 1961) View in CoL and the Argentine Conger View in CoL Conger orbignianus (Valenciennes) ( Etchegoin et al. 2005) in Argentinian View in CoL waters differs in an arched vitellarium and a long and pointed rhynchus. Prosorhynchus congeri View in CoL was recorded from Conger View in CoL sp. in Hawaii ( Yamaguti 1970). It differs from P. tonkinensis View in CoL in an arched vitellarium, smaller rhynchus percentage (5–8% vs 25–39%) and smaller egg size (25–28 × 14–19 compared to 29– 35 × 20–22). Prosorhynchus djedaba View in CoL from the Shrimp Scad Alepes djedaba (Forsskål) View in CoL off Mozambique ( Reimer 1985) differs from P. tonkinensis View in CoL in the arched vitellarium, the ovary overlapping the cecum and smaller egg length (15–16 × 10–11 compared to 29–35 × 20–22). Prosorhynchus longisaccatus View in CoL from ‘a Serranidae View in CoL : commonly called “leche”’ off New Caledonia ( Durio & Manter 1968) differs from P. tonkinensis View in CoL in the distinct arched vitellarium. Prosorhynchus mcintoshi View in CoL was first described from the Buru Glass Perchlet Ambassis buruensis Bleeker in Philippine View in CoL waters by Velasquez (1959). It differs from P. tonkinensis View in CoL in its arched vitellarium, slightly smaller width, rhynchus and premouth percentages and a greater preUR percentage (see Table 2). Prosorhynchus paracrucibulus View in CoL from A. buruensis in Philippine View in CoL waters ( Velasquez 1959) differs from P. tonkinensis View in CoL in its arched vitellarium, smaller rhynchus percentage and cirrus-sac reach percentage (see Table 2). Prosorhynchus rotundus View in CoL was reported from Mottled Soapfish Rypticus bicolar (Valenciennes) in the Galapagos ( Manter 1940a). It can be distinguished by the testes being wider than long, a smaller rhynchus percentage (19–21% compared with 25– 39%), the uterus filling most of the body (smaller PreUR% - see Table 2) and a larger egg size (39–44 × 20–23 vs 29–35 × 19–22). In addition, P. tonkinensis View in CoL can be distinguished from these Prosorhynchus View in CoL species in its indented posterior extremity.
sp. B ( VN). % refer to % of body length.
Species Prosorhynchus Prosorhynchus sp. A Prosorhynchus sp.
tonkinensis n. sp. ( VN) B ( VN) Host E. coioides E. coioides E. coioides Location Nghia Hung , Nam Dinh Cat Ba, Hai Phong Cat Ba, Hai Phong Number of specimens 7 3 1
Min Max Min Max
Length 830 1247 1020 1082 1597 Width 367 519 308 389 313 Pre-vitelline distance 219 349 232 308 320 Pre-cecal distance 121 505 312 420 375 Pre-uterine distance 333 553 530 618 584 Pre-mouth distance 616 801 554 651 644 Pre-testicular distance 452 667 682 710 493 Pre-ovarian distance 405 645 643 652 473 Rhynchus length 240 363 198 277 280 Rhynchus width 180 279 144 177 230 Rhynchus to vitellarium 0 58 31 33 49 Rhynchus to uterus 47 192 316 429 308 Rhynchus to cecum 47 170 116 152 92 Long vitelline field 371 505 237 281 447 Short vitelline field 270 386 182 237 291 Number of follicles in above 26 27 23 28 24 Vitelline fields 438 442 279 281 447 Cecum length 159 245 176 212 220 Cecum width 78 151 182 761 162 Pharynx length 57 100 82 97 87 Pharynx width 46 102 70 96 75 Ovary length 96 137 76 97 105 Ovary width 84 131 66 74 98 Ovary to anterior testis 0 0 0 0 0
......continued on the next page Length Width % Rhynchus % PreVR % PreUR % Prosorhynchus tonkinensis n. sp. 830–1247 34–51 23–34 23–31 36– 44 P. atlanticus 705–1677 27–33 24–26 23–25 36– 49 P. australis 1620–2900 30–43 19–23 16 25
P. congeri 1100 –1900 42– 51 5–8 22 34
P. crucibulum 1500 –2600 36–58 27–37 14–22 28– 36 P. djedaba 556–761 29–30 26–28 31 40
P. longisaccatus 1096 –1201 29–30 23–24 21 33
P. mcintoshi 820–1000 26–30 14–21 23 56
P. paracrucibulus 1096 –1900 36 13–16 23 -
P. rotundus 712–825 44–51 19–21 23 17
continued.
Site. Intestine.
Locality. Cat Ba, Hai Phong, Vietnam (20°46'30"N, 107°04'38"E, August 2013, July 2014). GoogleMaps
Description. Based on two specimens ( Table 1). Body fusiform, widest at vitelline region. Tegument spinous; spines squamous, tiny reach to posterior extremity. Rhynchus conical, muscular, longer than wide, blunt posterior end. Mouth always in midbody, anterior to ovary and anterior testis. Pharynx spherical, muscular. Cecum oval, saclike, extends anteriorly from pharynx.
Testes 2, subspherical, oblique or symmetrical, anterior testis immediately posterior to ovary; posterior testis located near cirrus-sac. Cirrus-sac elongate, muscular, not thick-walled, reaching posterior testis, located at posterior extremity of body. Seminal vesicle elongate, dextral in proximal portion of cirrus-sac. Pars-prostatica long, in two distinct parts; proximal part narrow, curved proximally to form convoluted seminal duct, joins seminal vesicle at posterior-dorsal aspect; distal part wider, straighter, surrounded by dense layer of gland-cells, lining of filaments in chevron arrangement. Ejaculatory duct narrow, complex genital lobe inside genital atrium. Genital atrium large. Genital pore distinctly separated from posterior end.
Ovary oval, anterior to or overlapping anterior testis. Uterus passes posteriorly from middle of anterior testis, extends to level of genital pore. Eggs tanned, numerous. Vitellarium forming an arch of 23–28 follicles, located in anterior half of body, beginning at level of mid-cecum, reach between cecum and rhynchus far anterior to testes. Excretory pore terminal. Excretory vesicle not clear in this specimen.
Remarks. Figure 3 shows 4 species without a black square against their name, namely, P. pacificus , P. atlanticus , P. australis and P. mcintoshi . Both P. pacificus from the Sailfin Grouper Mycteroperca olfax Jenyns at the Galapagos Island ( Manter 1940a) and P. atlanticus from M. bonaci from Florida waters ( Manter 1940b) can be distinguished from Prosorhynchus sp. A ( VN) by two lateral vitelline fields. Prosorhynchus australis ( Szidat 1961) differs from Prosorhynchus sp. A ( VN) in its long tapering rhynchus and slightly different PreUR% and Premouth% (see table 3 - comparison of Prosorhynchus sp. A ( VN) with similar body shape species). Prosorhynchus mcintoshi from the Duskytail Grouper Epinephelus bleekeri (Vaillant) in Philippine waters ( Velasquez 1959) is distinguished from Prosorhynchus sp. A ( VN) by its tandem testes, which are dextral at the level of the ovary. It also slightly differs in Premouth% and CSR% (see Table 3).
VN: Vietnam; NC: New Caledonia.
As illustrated in Figure 3, seven species have only a single black square and are similar to Prosorhynchus sp. A ( VN), namely P. bulbosus Kohn, 1961 , P. crucibulum , P. epinepheli Yamaguti, 1939 , P. longisaccatus , P. luzonicus Velasquez, 1959 , P. mizellei Kruse, 1977 and P. paracrucibulus . However, both P. bulbosus from Garrupa sp. in Brazilian waters ( Kohn 1967) and P. luzonicus from the Barrramundi Lates calcarifer (Bloch) in Philippine waters ( Velasquez 1959) can be easily distinguished from Prosorhynchus sp. A ( VN) in the separated vitelline fields, whereas in Prosorhynchus sp. A. ( VN), the vitellarium has an arched form. Prosorhynchus crucibulum from C. conger in the Northern Atlantic Ocean ( Matthews 1973) differs in the uterus reaching anterior to the caecum and a tapering rhynchus. Minor differences occur in length, Rhynchus%, PreUR% and Premouth% (Table 3). Prosorhynchus epinepheli was described from the Hong Kong Grouper Epinephelus akaara (Temminck & Schlegel) in Japan by Yamaguti (Yamaguti 1939). Its original illustration and description differs from Prosorhynchus sp. A ( VN) in the uterus extending anteriorly beyond the cecum and the mouth located between the testes. It also differs in other minor features such as Length, Width %, PreVR %, PreUR % and PTR % (Table 3). Prosorhynchus longisaccatus differs in the uterus which extends anteriorly along the left side of the cecum and the cirrus-sac is long and reaches to the pharynx. Prosorhynchus mizellei from the Smooth Lumpsucker Aptocyclus ventricosus (Pallas) in Amchitka waters, Alaska ( Kruse 1977) has a sub-pyriform body whereas Prosorhynchus sp. A ( VN) is fusiform. These species can also be distinguished in PreVR % (8–10 % vs 19–26 %) and minor differences in length, Width %, PreUR % and egg size (Table 3). Prosorhynchus paracrucibulus was detected from the scales of the Buru Glass Perchlet Ambassis buruensis Bleeker in Luzon , the Philippines, by Velasquez (1959). Its original description with some missing parameters seems to be of an immature worm, but Table 3 still demonstrates the distinction between P. paracrucibulus and Prosorhynchus sp. A ( VN) in Rhynchus % (13–16 % compared to 19–26 %), including minor differences in CSR % (27 % vs 34–43 %).
Figure 3 illustrates 9 species that have two black squares, namely P. congeri Yamaguti, 1970 , P. djedaba , P. longus Velasquez, 1959 , P. milleri , P. ozakii , P. polydactyli Yamaguti, 1970 , P. promicropsi , P. scalpellus McFarlane, 1936 and P. squamatus Odhner, 1905 . However, almost all of them can be distinguished from Prosorhynchus sp. A ( VN) in further features. For example, P. congeri from Conger sp. off Hawaii has its testes always located at the level of the cecum. Prosorhynchus djedaba can be distinguished in its extensive uterus, the cirrus-sac reaching the pharynx, and the mouth located at testes level. In contrast, in Prosorhynchus sp. A ( VN) the uterus never reaches to the vitelline field, the mouth is often located anterior to the testes and there is a gap between the pharynx and the cirrus-sac. Furthermore, it differs in a smaller egg size (15–16 × 10–11 vs 27–30 × 18), and slightly in length, Rhynchus %, PreVR % and CSR % (Table 3). Prosorhynchus longus from the Indian Halibut Psettodes erumei (Bloch & Schneider) off the Philippines ( Velasquez 1959) is distinguished its tandem testes, the vitellarium arranged in 2 lateral rows and located posterior to the ovary. Prosorhynchus milleri from the Yellowedged Lyretail Variola louti (Forsskål) off Australia ( Bott & Cribb 2009) differs from Prosorhynchus sp. A ( VN) in its tandem testes, compared with oblique testes. Furthermore, Table 3 shows two major distinguishing features (Width % and Rhynchus %) and five minor distinguishing features (length, PreVR %, PreUR %, CSR % and egg size). Prosorhynchus ozakii from the Snowy Grouper Hyporthodus niveatus (Valenciennes) off Florida ( Manter 1934) is distinguished by the vitellarium separated in 2 lateral fields. Prosorhynchus polydactyli from the Sixfinger Threadfin Polydactylus sexfilis (Valenciennes) off Hawaii ( Yamaguti 1970) clearly differs from Prosorhynchus sp. A ( VN) in its tandem testes, sinistral ovary, the uterus filling the bulk of the body, and a smaller rhynchus. Prosorhynchus promicropsi from the Atlantic Goliath Grouper Epinephelus itajara (Lichtenstein) off Florida ( Manter 1940b) differs in its vitellarium in two follicle fields, and the cecum extended anteriorly beyond the vitellarium. Prosorhynchus scalpellus from the Cabezon Scorpaenichthys marmoratus (Ayres) from Departure Bay, British Columbia ( McFarlane 1936) is clearly distinguished in that the uterus reaches anteriorly to the vitelline field, cecum, ovary and testes, and the seminal vesicle reaches beyond the thick walled part of the cirrus-sac. In contrast, in Prosorhynchus sp. A ( VN), the uterus never reaches the middle of vitelline field, the cirrus-sac contains the seminal vesicle and the cirrus-sac wall is thin. Table 3 also shows clearly the two major features separating P. scalpellus and Prosorhynchus sp. A ( VN) (PreUR % and SCR %) and seven minor distinguishing characters (Length, Width %, Rhynchus %, PreVR %, PTR % and Egg size). Prosorhynchus squamatus from a wide variety of fishes in the northern Atlantic Ocean is clearly distinguished from Prosorhynchus sp. A ( VN) in its tandem testes and the uterus reaching anterior to the cecum ( Odhner 1905). As can be seen from Table 3, major differences are the Rhynchus % and PreUR %, and minor differences are PreVR % and Premouth %. In Vietnam, P. epinepheli has been reported from E. coioides , E. bleekeri and the Malabar Grouper E. malabaricus (Bloch & Schneider) in Khanh Hoa province and was described with the mouth located between the sub-globular testes (Vo, 2010; Vo et al. 2011; Vo et al. 2012) but in our specimens, the mouth is located anteriorly to the testes and ovary. Prosorhynchus sp. B from E. coioides of New Caledonia has a description and illustration very similar with Prosorhynchus sp. A ( VN) in the present study. Although it differs from our specimens in table 3 (Width % and Rhynchus %), almost all features are very similar. Therefore, we suggest that Prosorhynchus sp. A ( VN) is synonymous with Prosorhynchus sp. B. ( Bray & Justine, 2013).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Prosorhynchus tonkinensis
| Truong, Thuong Van, Palm, Harry W., Bui, Te Quang, Ngo, Huong Thi Thuy & Bray, Rodney A. 2016 |
P. djedaba
| Reimer 1985 |
P. congeri
| Yamaguti 1970 |
Urophycis brasiliensis (Kaup) ( Szidat 1961 )
| (Kaup) (Szidat 1961 |
P. paracrucibulus
| Velasquez 1959 |
P. rotundus
| Manter 1940 |