Thieleella bathypacifica, Geiger, Daniel L. & Mclean, James H., 2010
|
publication ID |
https://doi.org/ 10.5281/zenodo.275645 |
|
DOI |
https://doi.org/10.5281/zenodo.6210887 |
|
persistent identifier |
https://treatment.plazi.org/id/03DCBC11-C247-FF9F-0ED2-1EA5FDA564A6 |
|
treatment provided by |
Plazi |
|
scientific name |
Thieleella bathypacifica |
| status |
sp. nov. |
Thieleella bathypacifica View in CoL n. sp.
Figures 23–24 View FIGURE 23 View FIGURE 24
Type material. Holotype ( FMNH 307884 ) , shell, operculum and radula mounted on SEM stubs. RV Atlantis, submersible grab, leg. J. Voight.
Type locality. 2572 m, ALVIN Dive 3938, Genesis, East Pacific Rise, 12.811˚N, 103.940˚W [approximately midway between Acapulco, Mexico, and Clipperton Island]. Sample with anemone and serpulid rocks.
Etymology. Bathy- referring to the deep-sea (bathyal) habitat of the species; -pacifica referring the Pacific Ocean; adjective.
Description. Shell trochiform globular to 1.85 mm. Protoconch of 0.75 whorls, reticulate sculpture, no apertural varix, apertural margin straight. TI of 0.875 whorls, 15 indistinct axial cords, prominent spiral cord forming ridge in position of selenizone, at intersection forming thickenings. TII of at least 1.5 whorls. Shoulder convex, 26 distinct raised axial cords, first spiral line after 0.33 TII whorls, 7 spiral lines after 1.5 whorls on outer two thirds of shoulder, spiral lines running over axial cords. Base convex, barely constricted below selenizone, evenly curving into umbilicus, same density of axial cords as on shoulder, crossed by spiral lines, 23 after 1.25 TII whorls, spiral lines running over axial cords. Selenizone at periphery, keels of moderate strength, high (higher than width of selenizone), regular growth lunules; slit open, with parallel margins.
Operculum round, thin, multispiral, central nucleus, covering aperture.
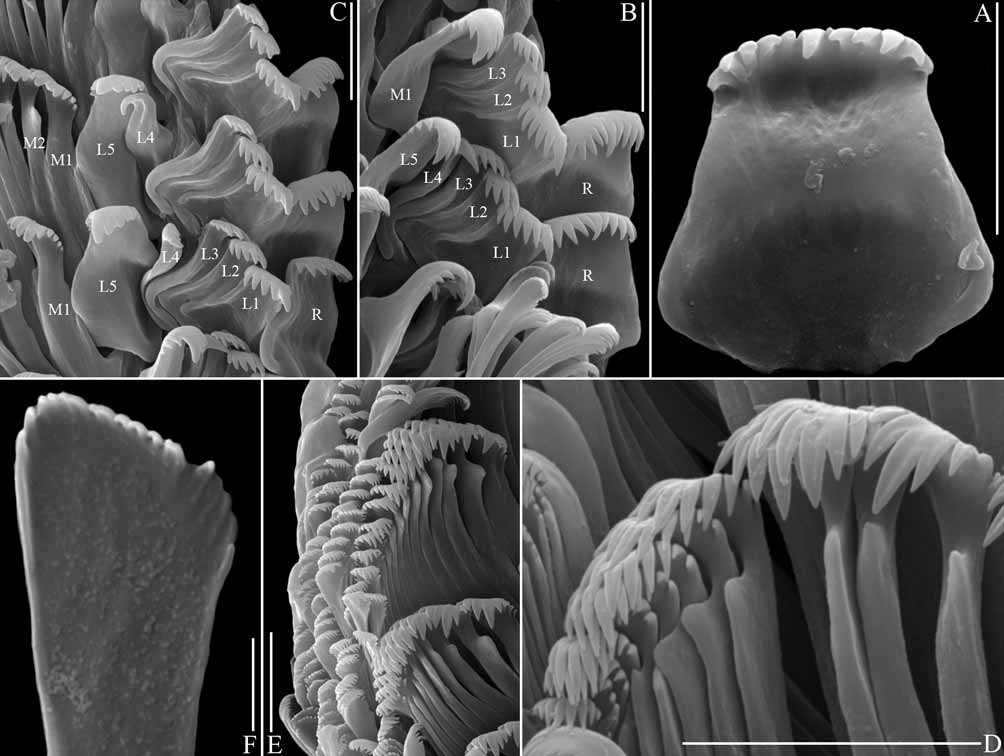
Radula ( Fig. 20 View FIGURE 20 ). Rachidian trapezoid, central denticle often largest, approximately five denticles on each side ( Fig. 20 View FIGURE 20 A–C: R). Lateral teeth 1–3 similar, 6,4,4 cusps respectively ( Fig. 20 View FIGURE 20 B–C: L1–3). Lateral tooth 4 reduced, hook-shaped, apical denticle largest, two small denticles on inner cutting edge ( Fig. 20 View FIGURE 20 C: L4).
Lateral tooth 5 enlarged by broadening, inner edge with 4–5 denticles, outer edge with three denticles ( Fig. 20 View FIGURE 20 B–C: L5). Inner marginal teeth triangular with approximately 3–4 denticles on each side ( Fig. 20 View FIGURE 20 D–E); with posterior projection on upper shaft (except marginal tooth 1: Fig. 20 View FIGURE 20 C: M1); outer marginal teeth spoon or paddle shaped with many fine denticles along apical edge ( Fig. 20 View FIGURE 20 E–F).
Differential diagnosis. Thieleella baxteri from the northeastern Pacific has elevated lamellar axial sculpture. Anatoma janetae Geiger, 2006 , from deep water of the eastern Pacific has a protoconch with flocculent sculpture in spiral orientation, the spiral cord on TI does not form a marked angulation, and shows a marked change of sculpture from predominant axial elements to exclusively spiral elements after 1.25 TII whorls.
Thieleella peruviana from deep water off Peru, on TI has only a week spiral cord not forming an angulation on the shell’s profile, and has more (42 vs. 15) and weaker axial cords on TI.
Remarks. The only specimen at hand is most likely not fully mature, because the final quarter whorl does not markedly descend along the coiling axis. The specimen is sufficiently distinct to warrant description as new, and material from that depth is unlikely to be collected in the foreseeable future. The specimen was preserved with the body in the shell, hence, was collected live in its natural deep-sea habitat.
The radula of the species shows a thus far unknown detail, namely the posterior projection on the apical portion of the shaft of the marginal teeth from marginal tooth 2 onwards. It is comparable to the food groove known from Tegula funebralis (A. Adams, 1855) and Liotiidae (see Morris & Hickman 1981, Hickman & McLean 1990). The occurrence of such a food groove in thus far a single species of Anatomidae demonstrates further the by now fairly well-documented variability of the radula within Anatomidae ( Geiger 2006a, Geiger & Sasaki 2008, Sasaki et al. in press).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
SubClass |
Vetigastropoda |
|
Order |
|
|
Family |
|
|
Genus |