Alevonota hepatica
|
publication ID |
https://doi.org/10.5281/zenodo.3762479 |
|
DOI |
https://doi.org/10.5281/zenodo.3804420 |
|
persistent identifier |
https://treatment.plazi.org/id/03E087B0-C601-FFA6-BFBE-FBF1FED0FED3 |
|
treatment provided by |
Valdenar |
|
scientific name |
Alevonota hepatica |
| status |
|
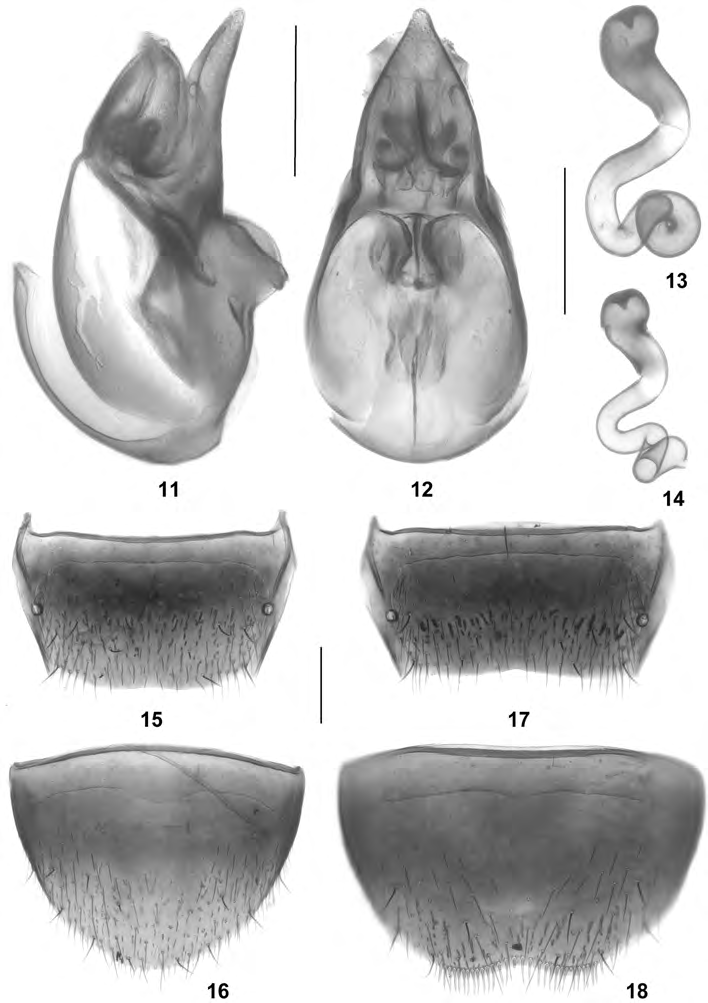
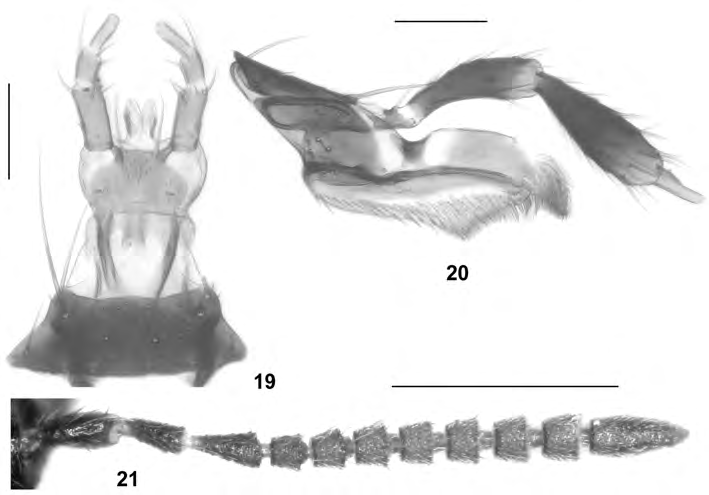
Alevonota hepatica View in CoL (ERICHSON, 1839), nov.comb. ( Figs 11-21 View Figs 11-18 View Figs 19-21 )
Homalota hepatica View in CoL ERICHSON, 1839: 102. Homalota major AUBE, 1850: 306. Calodera castaneipennis FAIRMAIRE & LABOULBENE, 1856: 380. Enalodroma fucicola THOMSON, 1859: 39. Homalota exarata SHARP, 1869: 186. Atheta ( Ptychandra) hepatica : GANGLBAUER (1895)). Atheta ( Enalodroma) hepatica : BERNHAUER & SCHEERPELTZ (1926). Atheta ( Enalodroma) hepatica : BENICK & LOHSE (1974). Aloconota hepatica : SAWADA (1984). Enalodroma hepatica: LOHSE (1989), LEE & AHN (2012). M a t e r i a l e x a m i n e d: Portugal: 1♀, Tronco, E Chjaves, 41°46'N, 7°18'W, 840 m,
meadow, near stone wall, sifted, 22.III.2002, leg. Meybohm (cAss). Spain: 2♀♀, Castilla-León, P.
N. de Guadarrama, Real Sitio d. S. Ildefonso (SG), Puerto de la Fuenfria, flight interception trap,
28.V.-14.VII.2017, leg. Lencina & González (cAss); 1♀, Castilla-León, Burgos, Sierra de Neila ,
Campino, 1500-1900 m, 22.V.1994, leg. Schülke & Grünberg (cAss) Switzerland: 1♂, Ticino,
Alpe di Neggia (Vira), 1350-1450 m, 29.V.1987, leg. Feller (cAss). Germany: 1♀, Niedersachsen,
25 km SW Hannover, Deister, Nienstedt, under bark, 23.V.1986, leg. Assing (cAss); 1♀,
Niedersachsen, Stadthagen, forest, pitfall trap, 29.VI.1991, leg. Sprick (cAss); 1♀, Niedersachsen, E Schladen, Hedeper, Westerberg, pitfall trap, V.2001, leg. Schmidt (cAss); 1♀, Niedersachsen, Wilhelmshafen env., Neuenburger Urwald, wood eclector (oak), 8.IV.-10.V.1993, leg. Menke (cAss); 1♂, Nordrhein-Westfalen, Bonn env., 10.IV.1991 (cAss); 1♂, Brandenburg, Karstadt, Perleberg, pitfall trap, 20.VII.1994, leg. Sprick (cAss); 1♂, Sachsen Leipzig, LSG Lössnig-Dölitz, pitfall trap near poplar trunk, 17.V.-8.VII.1995, leg. Sprick (cAss). Italy: 1♂, 2♀♀, Piemonte, Valle Stura (CN), Vallone Riofreddo, 1050 m, 5.V.1997, leg. Assing (cAss); 1♀, Basilicata, Rifreddo (PZ), 1100 m, 21-22.V.1984, leg. Angelini (cAss). Czech Republic: 3♂♂, České středohoři, Lipská hora, 24.IV.-18.V.1993, leg. Moravec (cAss). Slovenia: 1♂, Negova, Negovsko jez., 24.IV.1995, leg. Drovenik (cAss). Serbia: 1♂, Gramada planina, S Sastav Reka, 29.VII.2007, leg. Stévanović (cAss). Macedonia: 1♂, Cerovo vill., 41°43'N, 20°50'E, 880 m, near stream, 19.VI.2011, leg. Hlaváč (cAss). Greece: 1♂, 1♀, Makhedonía, NW Kavála, Pangéo, ski resort env., 1700 m, beech forest, litter sifted, 24.V.1999, leg. Assing (cAss); 1♀, same data, but 1650 m (cAss); 1♀, same data, but 1650 m, 28.V.1999 (cAss); 1♂, same data, but road to ski resort, 1300 m, 24.V.1999 (cAss); 1♂, Pangéo, peak region above ski resort env., 1900 m, debris near snowfield sifted, 24.V.1999, leg. Assing (cAss); 1♀, Oros Lepetimnos, track to Prof. Ilias, 39°20'N, 26°15'E, 730 m, large rocks with Quercus ilex, litter sifted, 25.III.2016, leg. Assing & Hetzel (cAss); 1♀, Ikaría, NE Pezi, 37°34'N, 26°04'E, 860 m, plateau, N-slope with Crataegus monogyna and ferns, litter and roots near stream sifted, 12.IV.2017, leg. Assing (cAss); 1♀, Ikaría, NE Pezi, 37°34'N, 26°05'E, 860 m, grazed plateau, fern litter and roots sifted, 12.IV.2017, leg. Assing (cAss). Turkey: see ASSING (2009, 2013). Russia: 1♂, Krasnodarskiy Kray, Temnolesskaya, Mezmay env., 850 m, 9.VI.1999, leg. Smetana (cAss). Armenia: see ASSING & SCHÜLKE (2019). Iran: 1♀, Mazandaran, 6 km W Part Kola, 36.14°N; 53.41°E, 2050 m, flight interception trap, VI.2015, leg. Barimani (cAss).
C o m m e n t: Intrageneric variation of external morphology in Alevonota is enormous, not only regarding body size and coloration, but also regarding proportions of body parts, punctation, male secondary sexual characters, and particularly habitat adaptations such as eye size, pigmentation, and the lengths of the elytra and the hind wings. On the other hand, Alevonota species share a more or less slender body with a parallel abdomen, often rather slender antennae with weakly coniform antennomeres IV-X, a median lobe of the aedeagus of relatively uniform morphology, and a relatively stout spermatheca with a relatively short proximal portion (often S-shaped). Moreover, except for the species exclusively confined to subterranean habitats (all the Canarian and some Continental species), West Palaearctic Alevonota share a similar phenology and natural history, i.e., short epigeic or aerial dispersal periods in spring and a subterranean reproduction period. Consequently, they are mostly collected - more or less accidentally - with pitfall traps and/or on the wing (flight interception traps, car-nets) during spring, only exceptionally in other seasons ( ASSING 2002, ASSING & WUNDERLE 2008).
A morphological examination of material of Enalodroma hepatica yielded no significant differences suggesting that this species should not belong to Alevonota. True, its habitus in more robust and the antennae are less strongly incrassate than those of other species from the European continent, but these difference are insignificant when compared to those between Canarian hypogean, Canarian endogean, and Continental European, and East Palaearctic species. On the other hand, the mouthparts, the general morphology of the antennae (antennomeres weakly conical and close) ( Fig. 21 View Figs 19-21 ), the shape of the head (subquadrate), the pronotal pubescence pattern, the parallel abdomen, the modifications of the male tergites VII and VIII (present only in some Alevonota species), the morphology of the median lobe and the parameres of the aedeagus, the shape and chaetotaxy of the female sternite VIII, and the general shape of the spermatheca are in agreement with the current concept of Alevonota. Moreover, a sexual dimorphism of the elytra ( E. hepatica: male elytra on either side with a longitudinal fold near posterior portion of suture) is shared with species such as Alevonota ocaloides (BRISOUT DE BARNEVILLE, 1863) (male elytra with rugose sculpture, female elytra with weak elevation along suture). For additional similarities and arguments see ÁDÁM (2010). The male and female sexual characters and the mouthparts are illustrated in Figs 11-20 View Figs 11-18 View Figs 19-21 . In fact, external and especially aedeagal characters suggest that E. hepatica may be more closely allied to A. rufotestacea (KRAATZ, 1856), the type species of the genus, than A. gracilenta .
The hypothesis that E. hepatica belongs to the Alevonota lineage is also strongly supported by its ecology. Since E. hepatica is usually found as singletons or in small numbers and mostly with pitfall and flight interception traps during the dispersal period in spring, BENICK & LOHSE (1974) inferred that the species is probably associated with subterranean nests and burrows of small mammals. However, it has never been recorded in greater quantities in systematic studies of such habitats (see, e.g., ISRAELSON 1971a, b), suggesting that the reproduction habitat is of a cryptic subterranean nature, like that of many Alevonota species (ASSING & WUNDERLE 2008). The material listed above was collected from a variety of very different habitats such as the leaf litter of various types of forest, in a meadow, under bark, from dead wood, near streams, and in alpine habitats above the tree-line. The altitudes range from sea level up to 2050 m. Additionally, the species has been reported from flood debris ( BARANOWSKI 1979), river banks and the shore of a reservoir (KUNZE & KACHE 1998, WENZEL 1997), sedge reed ( RENNER 1980), seashore deposits ( BARANOWSKI 1979), gardens ( HOLZER 1998, WAGNER 1997), xerothermous grassland ( BRENNER 1993, FELDMANN & LÜCKMANN 1998), bogs ( VOGEL 1998), from a nest in a hollow oak (BENICK & LOHSE 1959), plant debris (SCHOLZE & JUNG 1994), decaying plant matter (VOGEL & KAUFMANN 1982), flowering Crataegus ( LINKE 1962), and tree sap exudations (birch) ( LINKE 1962). Specimens were primarily collected with pitfall traps ( BRENNER 1993, FELDMANN & LÜCKMANN 1998, KÖHLER & KINKLER 2010, KUNZE & KACHE 1998, RENNER 2011, VOGEL 1982, 2013, ZANETTI et al. 2016), flight interception traps ( OWEN 1993, ZANETTI et al. 2016), wine traps ( ZANETTI et al. 2016), car-nets ( SCHÜLKE 2012, VOGEL 2013), and sweeping vegetation ( ERMISCH 1940). Of the 58 specimens reported from South Korea, as many as 56 were collected with flight interception traps and only two by sifting leaf litter (LEE & AHN 2012). ERMISCH (1940) observed a gradation in 1938, but failed to collect a single specimen in the same localities in the preceding and succeeding years. Practically all the revised and the literature records are based on single or few specimens collected during the period from March to June, mostly in April and May. Based on these observations, there is little doubt that all these records were accidental and that the habitats where the material was found do not represent the actual reproduction habitat. The latter is evidently cryptic and essentially unknown, as is the case for the majority of continental West Palaearctic Alevonota species.
Therefore, based on the above observations and argumentation, it is concluded that the species previously named Enalodroma hepatica in fact belongs to Alevonota, resulting in the following synonymies and combination: Alevonota THOMSON, 1858 = Enalodroma THOMSON, 1859, nov.syn., = Ptychandra GANGLBAUER, 1895 (previously a junior synonym of Enalodroma); Alevonota hepatica (ERICHSON, 1839), nov.comb.
Alevonota hepatica is not only widespread in the West Palaearctic region, from the Iberi- an Peninsula eastwards to Russia, Ukraine, Turkey, Armenia, and Iran, but has also been reported from South Korea (LEE & AHN 2012). The above material includes the first
records from Portugal, Slovenia, Serbia, Macedonia, the Russian South European Territory, and Iran.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
SubFamily |
Aleocharinae |
|
Genus |
Alevonota hepatica
| Assing, Volker 2019 |
Homalota hepatica
| Homalota hepatica ERICHSON, 1839: 102 |
| Homalota major AUBE, 1850: 306 |
| Calodera castaneipennis FAIRMAIRE & LABOULBENE, 1856: 380 |
| Enalodroma fucicola THOMSON, 1859: 39 |
| Homalota exarata SHARP, 1869: 186 |
| Atheta ( Ptychandra ) hepatica : GANGLBAUER (1895)) |
| BERNHAUER & SCHEERPELTZ (1926 |
| BENICK & LOHSE (1974). |
| SAWADA (1984) |
| LOHSE (1989) |
| LEE & AHN (2012). |