Acasta vivicava, Kolbasov, Gregory A., Chan, Benny K. K., Molodtsova, Tina N. & Achituv, Yair, 2016
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4178.2.2 |
|
publication LSID |
lsid:zoobank.org:pub:D9ABE50D-4C9A-4086-BD65-F692AF81A50E |
|
DOI |
https://doi.org/10.5281/zenodo.6068431 |
|
persistent identifier |
https://treatment.plazi.org/id/12D0864A-1683-4932-80D5-044716D8BC43 |
|
taxon LSID |
lsid:zoobank.org:act:12D0864A-1683-4932-80D5-044716D8BC43 |
|
treatment provided by |
Plazi |
|
scientific name |
Acasta vivicava |
| status |
sp. nov. |
Acasta vivicava sp.nov.
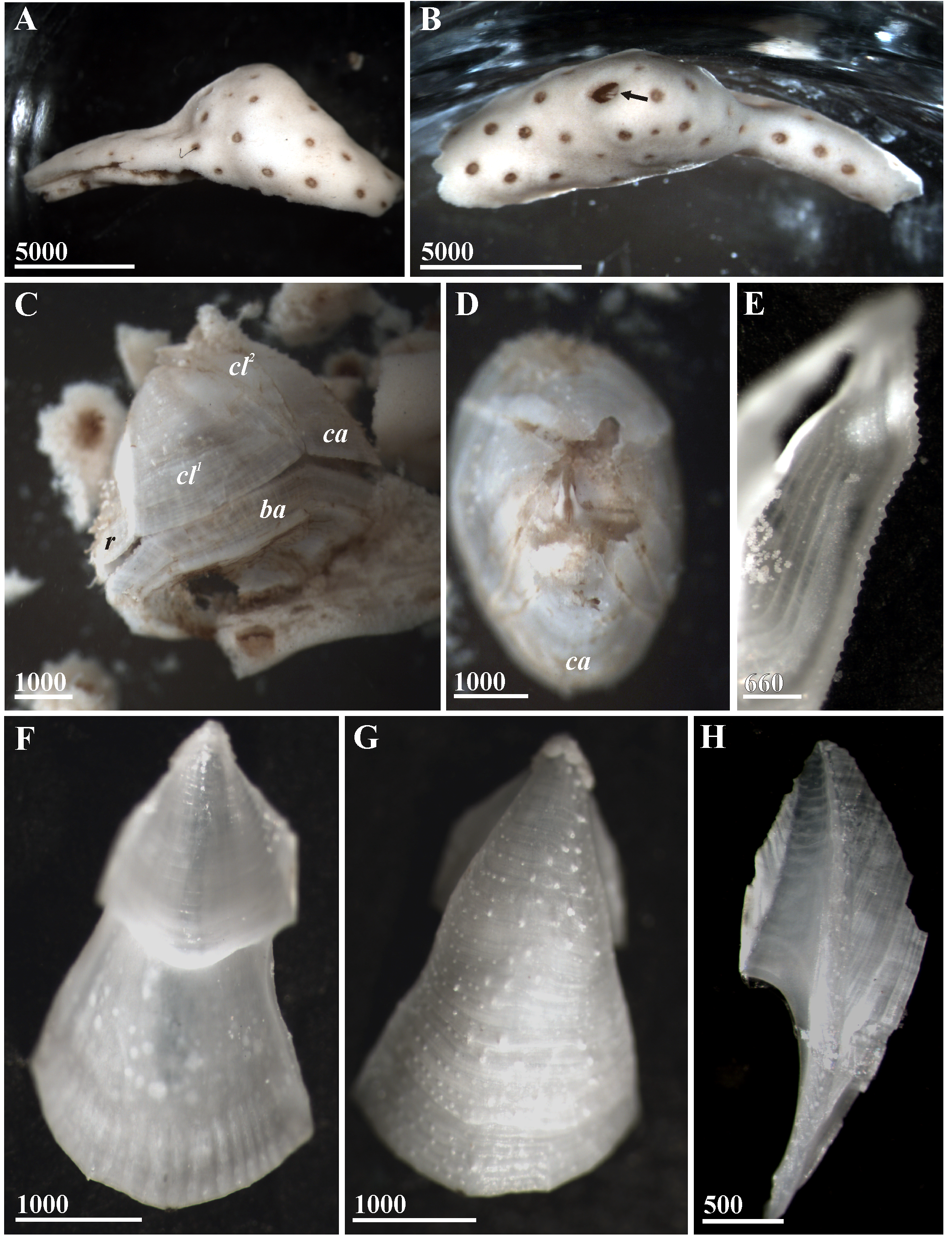
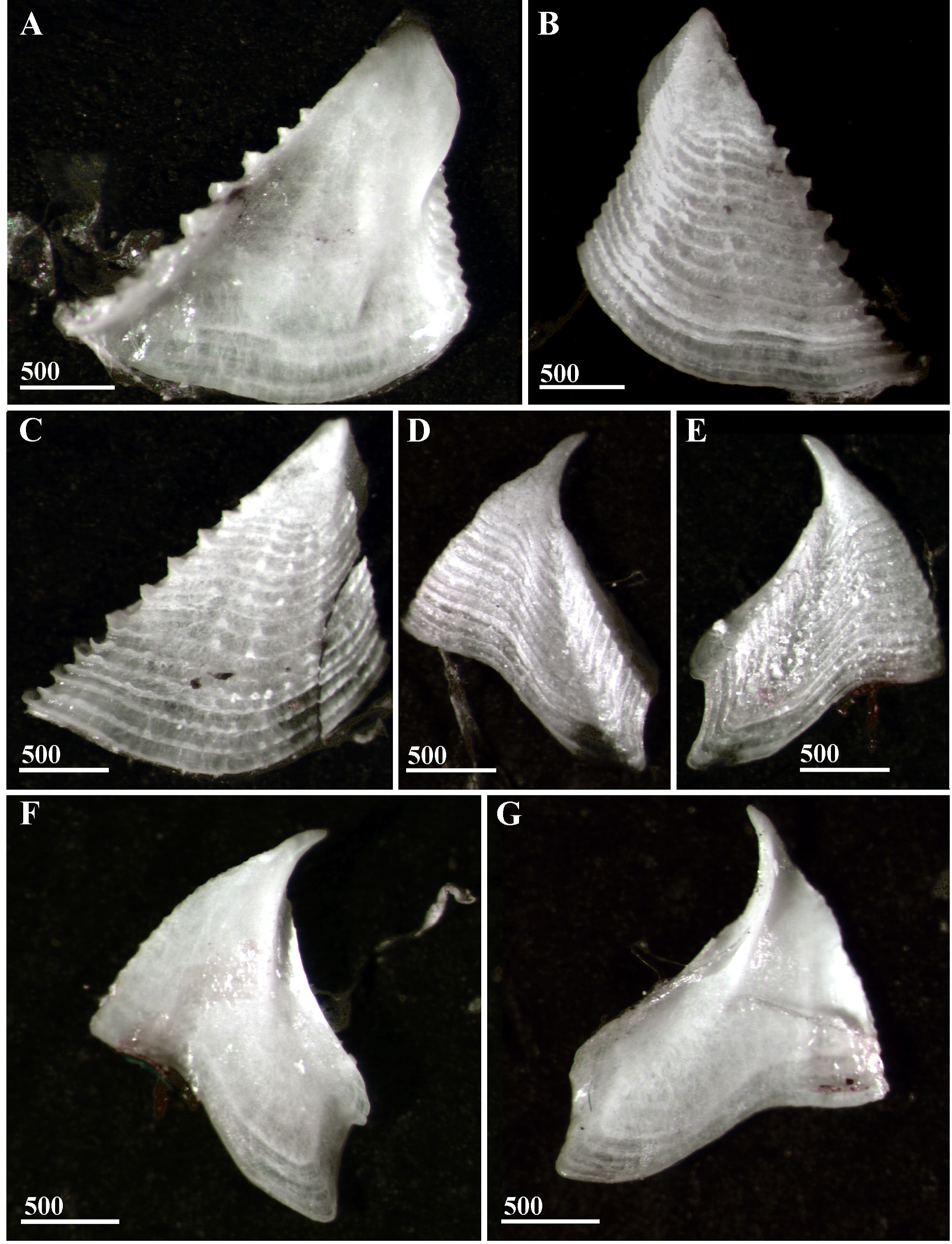
Figures 6–8 View FIGURE 6 View FIGURE 7 View FIGURE 8 .
Type material. National Museum of Natural History (Naturalis), Leiden, Holland. Locality : ‘ Oceanic Reef’s Expedition’ 1992–93, Seychelles, Amirante Islands, 6o12’S, 53o02’E, SCUBA diving, 0 2.01.1993, one specimen covered by a white scleraxonian alcyonacean Briareum sp. ( Briareidae ) GoogleMaps . Holotype RMNH. CRUS.C.10234 (shell compartments in ethanol, mouth parts and cirri mounted in glycerol on glass slide) is deposited in National Museum of Natural History (Naturalis), Leiden, The Netherlands .
Diagnosis. Shell white, with thin, feebly connected compartments, parietes with small dense external projections and developed basal longitudinal ribs on internal surface, carinolatus 2 narrow, basis saucer-shaped, with crenated margin, width of scutum equal to height, articular ridge prominent, tergum with long, sharp, beakshaped apex, basidorsal point of penis absent. Embedded in alcyonacean coenenchyme.
Description. Shell covered with host tissue forming gall ~ 8.8 mm long with small orifice ~ 0.9 mm long ( Fig. 6 View FIGURE 6 A, B). Shell white, slightly elongated in rostro-carinal axis, rostro-carinal basal length 4.5 mm, basal width 3 mm, height 4.0 mm, opercular orifice dentate, ~ 1.5 mm maximum length, compartments and basis feebly connected ( Fig. 6 View FIGURE 6 C, D). Basis saucer-shaped, with concentric growth lines, crenated margin, irregular depression in centre marking connection to axis of gorgonian, junction between basis and axis of host weak. Parietes ( Fig. 6 View FIGURE 6 F–H) with small, dense, external projections and fine growth lines, sheath ~1/2 total length, with horizontal striations, internal basal longitudinal ribs distinct. Radii and alae with oblique summits, radii reaching bases of parities and with fine oblique external striations, alae with horizontal striations. Carinolatus 2 narrow ( Fig. 6 View FIGURE 6 H), with parie only about 1/ 19 width of carinolatus 1.
Scutum ( Fig. 7 View FIGURE 7 A–C) thin, semitransparent, broad, width and height equal, external growth ridges crossed with few short, feeble longitudinal ridges, occludent margin dentate. Articular ridge ~1/2 length of tergal margin, prominent, with smooth, not truncated lower end, articular furrow developed, adductor ridge feeble, pits for adductor and depressor muscles not developed.
Tergum ( Fig. 7 View FIGURE 7 D–G) thin, semitransparent, with growth lines, apex long, sharp, beak-shaped, scutal margin concave, spur truncated, ~1/2 width of basal margin, distinctly separated from basiscutal angle, spur furrow shallow, wide. Articular ridge developed, ~1/2 total height, crests of depressor muscles absent.
Labrum ( Fig. 8 View FIGURE 8 A) with deep medial notch, 2 small teeth on each crest. Mandibular palps ( Fig. 8 View FIGURE 8 A) clubshaped, with long, dense setae distally. Mandible ( Fig. 8 View FIGURE 8 B–E) cutting edge with 5 teeth decreasing in size from upper to lower, teeth 1–3 well separated from each other, teeth 2–5 with bifid, inferior angle serrated, with 2 sharp denticles, inner margin with long, dense setae, outer margin with rare long, paired setae, lateral surface of blade with dense, small setae. Maxillules ( Fig. 8 View FIGURE 8 F, G) with 9–10 cuspidate setae of different lengths along straight cutting edge, notch absent, upper and lower pairs of cuspidate setae largest, long fine setae along outer and inner margins and on lateral surfaces of blade. Maxillae ( Fig. 8 View FIGURE 8 H) bilobed, with long setae along inner margins of lobes and on tip.
Most of the cirri of the specimen studied were dried and rami broken. Cirrus I with rami unequal, anterior ramus (12 segments) twice as long as posterior ramus (6 segments), both rami covered with moderately dense setae. Cirrus II with rami unequal, anterior ramus (7 segments) longer than posterior (5 segments), segments with dense setae. Cirri III ( Fig. 8 View FIGURE 8 I) with anterior ramus (8 segments) slightly longer than posterior (6 segments), several pairs of setae of different sizes along anterior edges of ramal segments, pair of stout setae at posterio-distal angle, segments of both rami without denticles or teeth. Cirri IV–VI with broken rami. Basal segments of cirrus IV without teeth and denticles. Penis ( Fig. 8 View FIGURE 8 J) long, annulated, basidorsal point absent.
Remarks. Nine species of the genus Acasta were found in alcyonaceans— A. alcyonica Rosell, 1991 ; A. alcyonicola Utinomi, 1953 ; A. echinata Hiro, 1937 ( Fig. 13 View FIGURE 13 H–K); and A. umitosaka Utinomi, 1962 from alcyoniinan alcyonaceans; A. antipathidis Broch, 1916 (this species was previously reported to have an antipatharian host; however, study of photographs of the syntypes revealed that the colony reported as a black coral in fact represents a denuded dead axis of an alcyonacean); A. gregaria Utinomi, 1959 ; A. hirsuta Broch, 1916 ; A. purpurata Darwin, 1854 ; and A. vipensis Van Syoc, Carrison-Stone, Madrona & Williams, 2014 from alcyonaceans (gorgonians). The new species, A. vivicava , differs from these species in having a narrower CL2, a scutum with a prominent articular ridge and a tergum with an elongated sharp apex. The new species can also be distinguished from A. alcyonica , A. purpurata and A. umitosaka by the absence of large slits or windows between the shell plates, and from A. alcionica , A. gregaria and A. hirsuta and A. vipensis by the absence of the longitudinal striation of scutum.
It should be noted that several archaeobalanid species that are epibionts of alcyonaceans and antipatharians also representing acastines were erroneously assigned to the genus Conopea , as demonstrated by the phylogenetic analyses (see below). These include C. canaliculata (Ren et Liu, 1978) ; C. dentifer ( Broch, 1922) ; C. folliculus ( Hiro, 1937) ; C. fragilis ( Broch, 1931) ; C. longibasis ( Hiro, 1937) ; C. navicula ( Darwin, 1854) ( Fig. 13 View FIGURE 13 A–G); C. pygmaea ( Broch, 1931) ; and C. squamosa ( Rosell, 1991) . Acasta vivicava also differs from C. canaliculata , C. dentifer , C. fragilis , C. longibasis , C. navicula and C. pygmaea in having the scutum with equal basal and tergal margins and with a prominent articular ridge, the tergum with an elongated sharp apex and a narrower carinolatus 2. The new species has opercular valves slightly similar to C. follisculus , but differs by the scutum having a more prominent articular ridge and by the absence of a depressor muscle pit. Acasta vivicava differs from C. squamosa by possessing a narrower carinolatus 2 and by the elongated, sharp apex of the tergum. Acasta vivicava also can be distinguished from C. canaliculata , C. dentifer , C. longibasis , C. fragilis and C. navicula by the absence of developed longitudinal striation of the scutum.
Etymology. The specific name represents the similarity to a bird living in the tree hollow, from the Latin ‘ vivet ’ meaning live and ‘ cavae ’ hollow.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |