Platalina genovensium Thomas, 1928
|
publication ID |
https://doi.org/ 10.1093/mspecies/seaa008 |
|
publication LSID |
lsid:zoobank.org:pub:9401F41A-6F49-4609-8699-73FCA5D5DA4A |
|
persistent identifier |
https://treatment.plazi.org/id/03ECA20A-FFF0-875A-EFAB-1D62FCF4FA24 |
|
treatment provided by |
Felipe |
|
scientific name |
Platalina genovensium Thomas, 1928 |
| status |
|
Platalina genovensium Thomas, 1928 View in CoL
Peruvian Long-tongued Bat
Platalina genovensium Thomas, 1928:121 View in CoL . Type locality “neighborhood of Lima,” Lima, Peru.
CONTEXT AND CONTENT. Context as for genus, no subspecies are recognized (Griffiths and Gardner 2008).
NOMENCLATURAL NOTES. Thomas’s (1928) original description of Platalina genovensium was based on the holotype (BMNH [British Museum of Natural History] 27.11.19.38) initially preserved as a fluid specimen subsequently prepared as a skin and skull, collected in Lima, Peru, by Nicolo Esposta on 23 April 1909 (Carter and Dolan 1978). Thomas (1928) misspelled twice the last name of Nicolo Esposta as “Esposto” (Carter and Dolan 1978). The generic name, Platalina , comes from two Italian words: “piatta,” meaning flat, and “linea,” meaning line; in reference to the “straight line” of the skull in lateral view. The species name, genovensium , is in honor of Thomas’s Genovese colleagues at the Museo Civico, Genova, where the holotype was stored before being moved to the British Museum in exchange for other specimens (Carter and Dolan 1978). Vernacular names for P. genovensium are long-snouted bat, Peruvian long-nosed bat, Peruvian long-tongued bat, murciélago hocico largo, murcie peruano ( Galaz et al. 1999; Aragón and Aguirre 2007; Griffiths and Gardner 2008; Pacheco et al. 2009; Malo de Molina et al. 2011; Zamora et al. 2013; Maguiña and Amanzo 2016).
DIAGNOSIS columns, the medial column with three or more vibrissae and the lateral column with two vibrissae. Forearm and distal part of humerus naked. Well-developed uropatagium lacking a trail of hairs on its edge, the median extension reaching the lower one-third of tibia. The calcar is as long as foot including claws. The plagiopatagium inserts at tarsus. Tail present (ca. 8 mm), projects dorsally into first quarter of the uropatagium as a noticeable stump ( Thomas 1928; Solmsen 1998; Carstens et al. 2002; Griffiths and Gardner 2008).
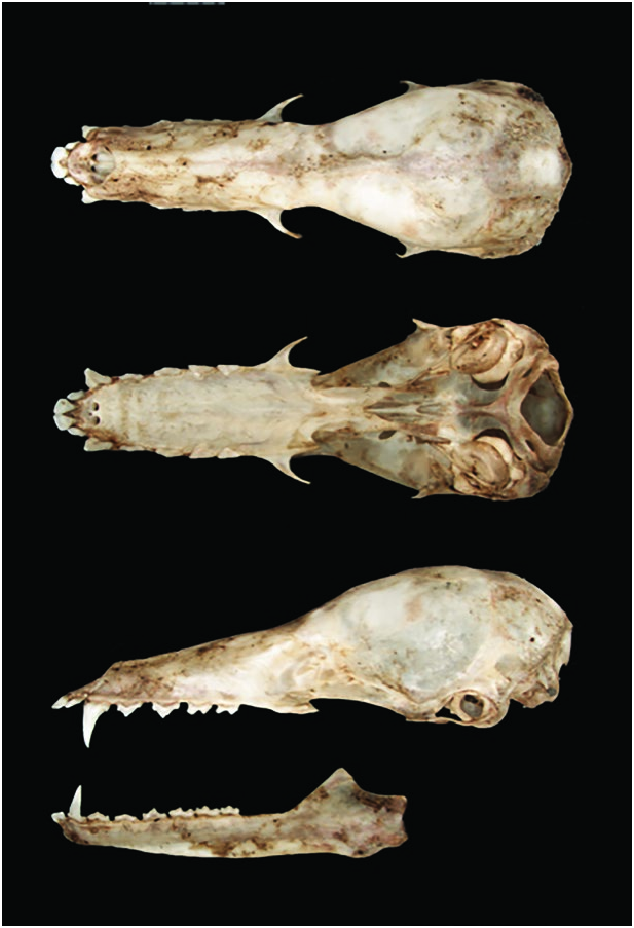
The rostrum is longer than the braincase. Premaxillary bones projected beyond the canines, producing a triangular projection. The hard palate is flat. The mesopterygoid fossa is relatively long and its anterior margin “V”-shaped. Zygomatic arches are incomplete. Pterygoids do not reach the tympanic bullae. A prominent narrow and elevated ridge divides the basioccipital along its medial axis ( Solmsen 1998; Carstens et al. 2002; Griffiths and Gardner 2008).
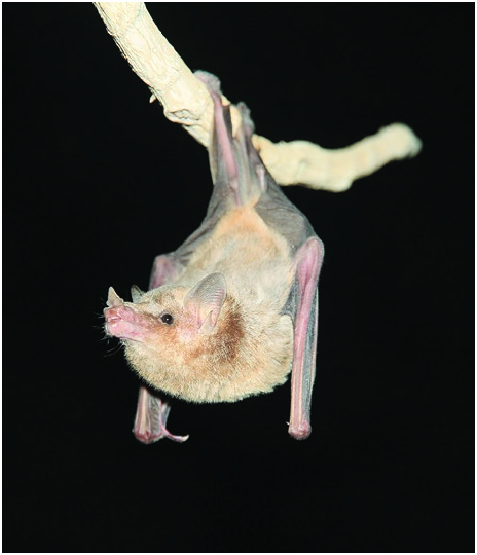
Platalina genovensium ( Fig. 1 View Fig ), the sole member of the genus Platalina , is distinguished from other lonchophyllines ( Lionycteris , Lonchophylla , and Xeronycteris ) by a combination of characters of external and skull morphology, including: greatly elongated muzzle; forearm naked and ranging from 47 to 53 mm (33–37 mm in Lionycteris , 30–48 mm in Lonchophylla , 35–38 mm in Xeronycteris ); rostrum clearly longer than the braincase ( Fig. 2 View Fig ; rostrum length equal to or less than length of braincase in other lonchophyllines); all incisors in contact; incisors not in contact with the canines; upper incisors projecting forward; flattened lingual surface of upper canines; reduced upper molars metastyles; weakly trifid lower incisors; lower canines present a lingual cingulum; and all lower molars lacking entoconulids ( Phillips 1971; Gregorin and Ditchfield 2005; Griffiths and Gardner 2008).
GENERAL CHARACTERS
Platalina genovensium , the largest lonchophylline, presents a long muzzle ( Fig. 1 View Fig ). The dorsal pelage is long (ca. 8 mm) and bicolored, dark brownish in adults and grayer in subadults ( Fig. 1 View Fig ; Velazco et al. 2013). The venter is lighter than the dorsum ( Fig. 1 View Fig ). The spear of the noseleaf is small, smooth, and triangular, lacking a central rib; two distinct narrow grooves parallel with both basal edges. Lateral edges of horseshoe swollen, fused to face along entire length with no free edge. Inferior horseshoe grades smoothly into upper lip, no distinct boundary between lip and horseshoe. Pair of dermal pads on the chin, one on each side of midline. Genal vibrissae absent. Three interramal vibrissae present. Vibrissae lateral to noseleaf arranged in two
Upper incisors are in contact with each other, large, spatulate, and procumbent. The canines are slender and lack a conspicuous cingulum. Upper premolars mesiodistally long and buccolingually narrow. The first upper premolar is mesiodistally short lacking the labial cingulum; the second upper premolar presents a weakly labial cingulum and an accessory distal cuspid. Upper molars are elongated and lack a paracone and mesostyle, but retain a comparatively well-developed parastyle and a reduced metastyle. Lower molars are elongated and lack an entoconid ( Phillips 1971; Solmsen 1998; Carstens et al. 2002; Griffiths and Gardner 2008).
Ranges (n) of external and skull measurements (mm) and masses (g) of adult females from Chile and Peru ( Galaz et al. 1999; Aragón and Aguirre 2007; Velazco et al. 2013; Ruelas and Pacheco 2018; Ruelas 2019) were: total length, 72.0–90.0 (26); tail length, 5.0–11.0 (24); hind foot length, 11.5–18.0 (28); tibia length, 19.2 (1); ear length, 13.0–20.0 (28); forearm length, 47.0–53.0 (26); greatest length of skull, 30.6–33.7 (16); condylobasal length, 28.9–30.8 (13); condylocanine length, 28.1–29.4 (13); palatal length, 17.0–18.6 (14); postorbital constriction, 4.7–5.1 (15); mastoid breadth, 10.5–11.2 (14); breadth of anterior braincase, 9.6–10.6 (13); braincase height, 7.3–7.8 (13); length of maxillary toothrow, 10.0–11.1 (15); width across M3, 4.0–5.5 (11); greatest breadth across the canines, 3.0–3.3 (13); mandibular length, 21.5–23.3 (14); length of mandibular toothrow, 10.8–11.9 (5); and body mass, 12.8–26.5 (20).
Ranges (n) of external and skull measurements (mm) and masses (g) of adult males from Chile and Peru ( Thomas 1928; Ortiz de la Puente 1951; Aellen 1966; Swanepoel and Genoways 1979; Galaz et al. 1999; Velazco et al. 2013; Ossa et al. 2016; Ruelas and Pacheco 2018; Ruelas 2019) were: total length, 72.0–90.0 (36); tail length 6.0–14.0 (36); hind foot length, 11.0–15.0 (37); tibia length, 18.0–21.0 (6); ear length, 11.7–21.0 (39); forearm length, 46.1–52.0 (42); greatest length of skull, 27.5–32.7 (38); condylobasal length, 29.5–30.7 (28); condylocanine length, 28.4–29.9 (28); palatal length, 17.3–18.8 (28); postorbital constriction, 4.6–5.2 (36); mastoid breadth,(10.4–11.3 (32); breadth of anterior braincase, 9.5–10.1 (28); braincase height, 7.2–7.9 (28); length of maxillary toothrow, 10.1–11.2 (34); width across M3, 4.1–5.8 (24); greatest breadth across the canines, 2.8–3.3 (28); mandibular length, 21.6–23.6 (30); length of mandibular toothrow, 10.9–11.4 (4); and body mass, 12.8–21.4 (25).
DISTRIBUTION
Platalina genovensium is known from several localities west of the Andes from northern Peru (6.4°S) to northern Chile (18.5°S — Fig. 3). At the eastern slopes of the Andes there are five records: two from the Department of Huánuco in central Peru, two from the Department of Amazonas in northern Peru, and one from the Department of Cajamarca in northern Peru ( Velazco et al. 2013; Ossa et al. 2016; Ruelas and Pacheco 2018). The elevational range is from near sea level (Piura) to 2,600 m (Arequipa — Velazco et al. 2013; Ruelas and Pacheco 2018). The type locality was specified only as “neighborhood of Lima ” by Thomas (1928).
Ruelas and Pacheco (2018) produced a potential distribution map based on historic records, and new records from the Peruvian departments of Amazonas, Arequipa, Cajamarca, Ica, and Lima, suggesting that P. genovensium has a continuous distribution from the western slopes to the eastern Amazonian basin crossing the Depression of Huancabamba, and on the eastern Andes from the Marañón dry valley to the relicts of dry forests from Huánuco. Additionally, the proposed distribution map suggests the presence of P. genovensium in other eastern dry forests located on Huancabamba and the Mantaro and Apurímac rivers.
FOSSIL RECORD
Platalina is known from Pleistocene deposits from the Jatun Uchco cave network in the city of Ambo, Department of Huánuco, Peru ( Shockey et al. 2009). Along with the Platalina fossil remains, other bats ( Anoura and Desmodus ), six species of carnivores, and one extinct xenarthran were also recovered ( Shockey et al. 2009).
FORM AND FUNCTION
Form:— The hairs of Platalina genovensium present long, divergent, entire coronal alternate with hastate coronal scales; melanin granules are generally dispersed, but most abundant in the distal one-third of the hairs; medulla is absent ( Benedict 1957). The third metacarpal is longer than fourth or fifth. First phalanx of digits III and IV are shorter than second phalanx on their respective digits ( Carstens et al. 2002).
Sexual dimorphism occurs in the robustness of the mandibular rami, where the females have thin and more delicate mandibular rami than the males ( Velazco et al. 2013). Although this assumption was based on a small sample size (two females and two males), it was supported by the examination of additional specimens by Ruelas and Pacheco (2018) and Ruelas (2019); however, Ruelas and Pacheco (2018) reported an outlier male specimen (MUSM [Departamento de Mastozoología, Museo de Historia Natural, Universidad Nacional Mayor de San Marcos] 44631) that presented a thin mandible.
Dental formula is: i 2/2, c 1/1, p 2/3, m 3/3, total 34 ( Solmsen 1998), although variations in the dental formula have been reported for some specimens ( Velazco et al. 2013; Ruelas and Pacheco 2018). Velazco et al. (2013) reported one adult male specimen (CEBIOMAS [Colección de Mastozoología, Centro de Ecología y Biodiversidad] 224) with only two upper molars present on each side and no sign of dental alveoli behind the second upper molar that could suggest the third molar is missing. Ruelas and Pacheco (2018) reported one male specimen (MUSM 44689) with only two upper molars present on each side and another male specimen (MUSM 38627) with two upper molars on each side, two lower molars on the right side and three on the left side. Both specimens showed no sign of the presence of dental alveoli that could suggest the third upper or lower molars are missing.
In an extensive study of the anatomy of the bony labyrinth, Velazco and Grohé (2017a, 2017b) observed that the width of the cochlea relative to the basicranium in P. genovensium is intermediate among the cochleae of other bats. The contribution of the cochlear volume to the entire bony labyrinth is 69.7%. The coiling of the cochlear canal completes 2.25 turns. P. genovensium presents the shortest number of cochlear spiral turns in the family Phyllostomidae . The estimated height of the spiral of the cochlea is 2 mm. P. genovensium lacks an apical lacuna at the apex of the cochlear spiral. The basal turn becomes thicker when approaching the vestibule and the second turn of the cochlear spiral is slightly narrower than the basal turn and overlies partially the basal turn. The common crura is shorter and wider than in other bats. The posterior limb of the lateral semicircular canal opens directly into the vestibule between the posterior ampulla and the base of the common crus. The posterior limb of the lateral semicircular canal is located more dorsally than the ventral limb of the posterior semicircular canal. The angles between the planes of the anterior and lateral semicircular canals are 90°, between the posterior and lateral are 90°, and between the posterior and anterior are 95°. The semicircular canals are mostly planar. The anterior semicircular canal is more extended dorsally than the posterior semicircular canal. The lateral semicircular canal has an oval shape in dorsal view. The anterior semicircular canal extends more anteriorly than the lateral semicircular canal. Anteriorly the anterior and lateral semicircular canals extend at the same level (Velazco and Grohé 2017a). P. genovensium exhibits the greatest aspect ratio of the cochlear spiral in bats, and among placental mammals it is second only to rodents of the genus Cavia ( Ekdale 2013; Velazco and Grohé 2017a). Three-dimensional surface models of the bony labyrinth of one individual are available in a public repository (Velazco and Grohé 2017b).
Griffith (1982) studied the morphology of the hyoid and lingual regions of the New World nectar-feeding bats, obtaining the following results: the m. stylohyoideus is absent in P. genovensium . The m. sphincter colli profundus consists of two distinct parts, both originating from the fascia of the posterior mylohyoid region. One part passes laterally to insert on the inner surface of the cervical skin. The other part passes anterolaterally to insert on the inner surface of the cervical skin at a 45° angle just lateral to the mandible. The m. stylopharyngeus originates from the posteromedial border of the stylohyal bone, at a point about 2 mm from the lateral tip; it passes medially to insert in the fibers of the pharyngeal wall, between the hyopharyngeus and thyropharyngeus muscles. The m. ceratohyoideus originates from the anterior edge of the thyrohyal bone (lesser hyoid cornu), and from the fibers of the m. thyrohyoid and inserts on the entire posterior surface of the epihyal and the lateral tip of the m. ceratohyal. The m. hyopharyngeus is almost absent. The m. cricopharyngeus consists of two slips. The m. genioglossus originates from the medial surface of the anterior 1 mm of the mandible, deep to the origin of the m. geniohyoid and inserts into the posterior one-half to one-third of the ventral surface of tongue. The m. hyoglossus originates from the insertion of the m. sternohyoideus, passes anteriorly to insert in the posterior tongue surface, medial to the insertion of the m. styloglossus. The latter originates from the expanded distal tip of the stylohyal, and from the posterior 4 mm of the lateral surface of the stylohyal bone; and inserts on the lateral surface of tongue along much of its length. The m. geniohyoideus originates via tendon from the medial surface of the mandible at a point approximately 2 mm lateral to the symphysis and splits near insertion, the deep fibers insert directly on anterior surface of basihyal, while the superficial fibers pass ventral to basihyal and insert in association with m. hyoglossus and m. sternohyoideus. The medial and lateral fibers of the m. sternohyoideus originate from the xiphoid process of sternum and insert via raphe into the fibers of m. hyoglossus and m. genioglossus. The m. sternothyroideus originates from the dorsal surface of the medial clavicle, just lateral to the sterno-clavicular articulation; runs anteriorly to insert on the lateral surface of the thyroid cartilage, just posterior to the origin of the thyrohyoideus muscle. The m. omohyoideus is robust, originates from the deep surface of the bone of the scapula immediately surrounding the scapular notch; passes ventrally and medially, curving around the cervical area deep to both the sternomastoid and cleidomastoid muscles and inserts weakly in the fibers of the hyoglossus and mylohyoid muscles. The m. mylohyoideus is undivided ( Griffiths 1982).
The tongue in P. genovensium presents medial and lateral circumvallate papillae. Papillary bodies of medial circumvallate papilla separate from valla, fossae complete. The lateral circumvallate papillae are located on lateral surface of tongue. The pharyngeal region of the tongue is covered laterally with papillae and medially bare. Lateral lingual sulci are present. Brush of hairlike papillae is present around the distal margin of the tongue. Hairlike papillae are fleshy and conical and are confined to lateral margin of distal one-third of tongue, with a single line of papillae that extends roughly to the lateral circumvallate papillae. All or most medial-posterior mechanical papillae inclined toward the pharyngeal region of the tongue. There is a small patch of mechanical papillae near the midline of the tongue. Long-tipped, bifid anterior mechanical papillae absent. Basketlike medial-posterior mechanical papillae are absent. A cluster of horny papillae is located near tip of tongue. Single large horny papilla present in center of elliptical cluster. Papillae are lacking anterior to main horny papilla. Two or more small horny papillae present posterior to main papilla. Lack of horny papillae lateral to main papilla. Paired lingual arteries present. Lingual veins not enlarged ( Griffiths 1982; Wetterer et al. 2000; De La Mar 2001).
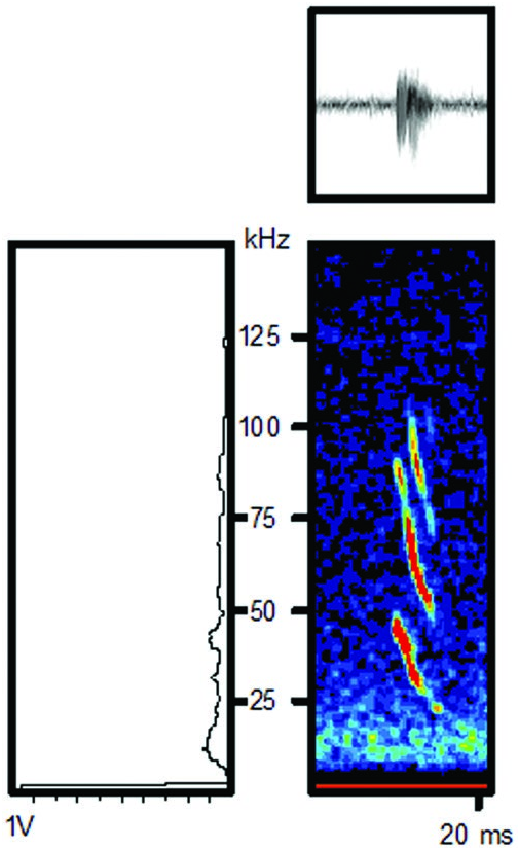
Function. —The echolocation calls of Platalina genovensium were recently described for individuals in Peru (n = 5— Malo de Molina et al. 2011) and Chile (n = 1— Ossa et al. 2016; Fig. 4 View Fig ). Calls contain three frequency modulated (FM) harmonics of low intensity (−10 to −35 dB at 1 m of distance), characteristics that make P. genovensium difficult to record. The three harmonics have a maximal energy at 42.2, 89.2, and 111.9 kHz, the duration of the pulses (mean ± SD) is short (1.98 ± 0.11 ms) and the interval between pulses is variable (90.08 ± 13.79 ms— Malo de Molina et al. 2011; Ossa et al. 2016).
ONTOGENY AND REPRODUCTION
Several craniodental features are present in juveniles which are not present in adult individuals (e.g., strongly bilobulated upper incisors, lower incisors trilobulated, convergent rostrum, and barely pompous lachrymal— Ruelas 2019). Sex ratio in colonies from Arequipa is 1:1, and pregnant females are always found in mixed-sex colonies (Sahley and Baraybar 1996). Within the geographic distribution of Platalina genovensium , reproduction rates are very sensitive to drought, showing a significant decrease before and after El Niño events in southern Peru (Sahley and Baraybar 1996). The presence of food sources could allow P. genovensium in different areas to reproduce in different periods during the year, which is associated with the phenology of the different species of columnar cactus throughout its distribution (Maguiña and Amanzo 2016). Sahley and Baraybar (1996) reported three pregnant females in October. Baraybar (2004) reported five pregnant females, two in March and three in December 1990. Zamora (2005) reported a lactating female in December. Zamora et al. (2013) reported a female in early stage of pregnancy and another in lactation in March; while in June (dry season), they registered a female during the lactation period.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Platalina genovensium Thomas, 1928
| Ossa, Gonzalo, Zamora, Hugo T & Velazco, Paúl M 2020 |
Platalina genovensium
| THOMAS, O. 1928: 121 |