Herpetoreas burbrinki, Guo & Zhu & Liu & Zhang & Li & Huang & Pyron, 2014
|
publication ID |
https://doi.org/ 10.11646/zootaxa.3873.4.5 |
|
publication LSID |
lsid:zoobank.org:pub:3B3F6C1E-BBD9-4ED3-91CC-4B89FBC9E348 |
|
DOI |
https://doi.org/10.5281/zenodo.5123280 |
|
persistent identifier |
https://treatment.plazi.org/id/03F587CE-FFC5-106C-A98D-FE87CD9ECD53 |
|
treatment provided by |
Felipe |
|
scientific name |
Herpetoreas burbrinki |
| status |
sp. nov. |
Herpetoreas burbrinki View in CoL sp. nov.
( Figs. 1 View FIGURE 1 & 3 View FIGURE 3 )
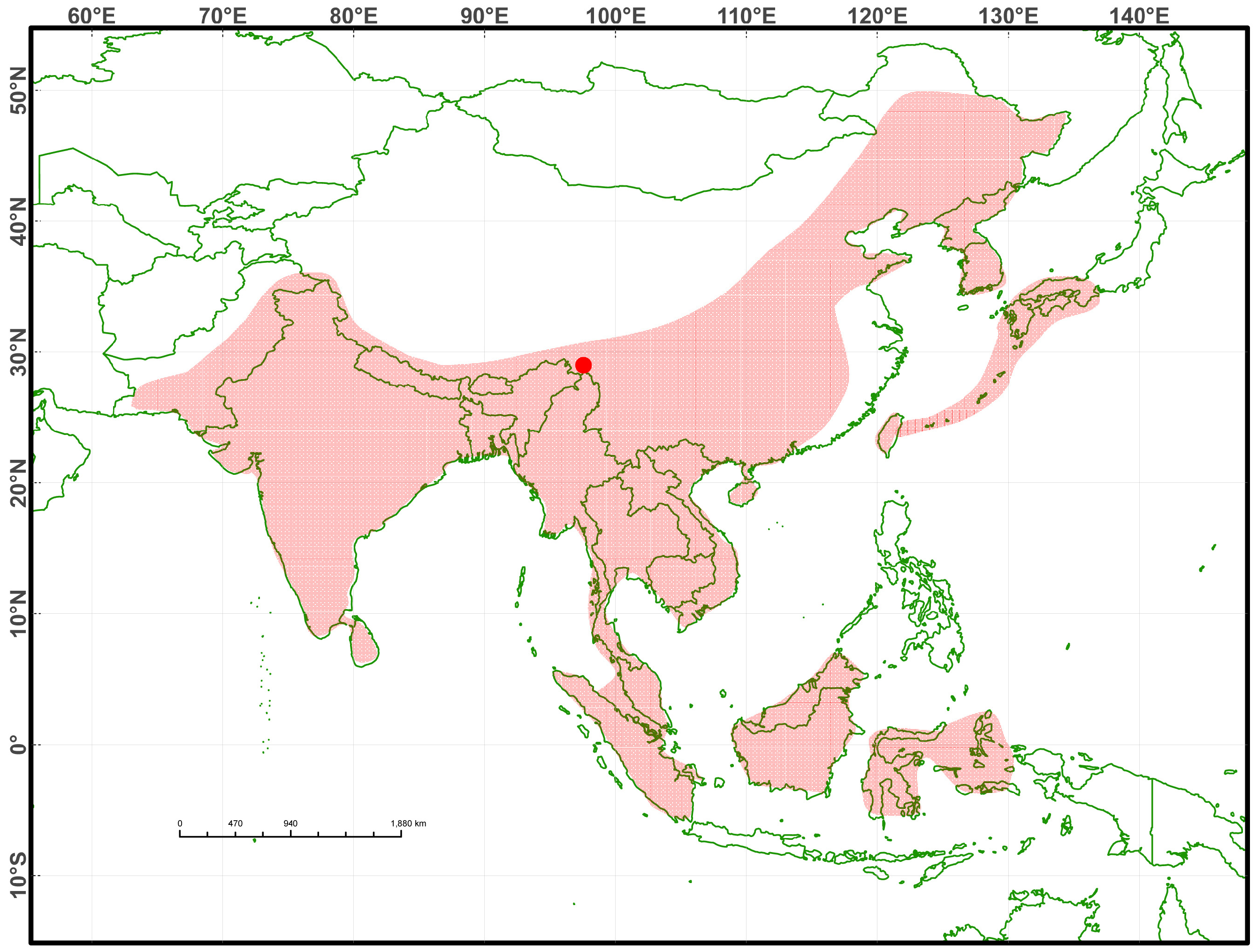
Holotype. YBU 071128, an adult male, collected in Sep. 2007 in Zayu County, Xizang A. R., China, at an elevation of 1889 m above sea level ( Fig.1 View FIGURE 1 ).
Diagnosis. Herpetoreas burbrinki can be distinguished from its relatives on the basis of the following combination of characters: 1) TL/SVL ratio in the single male 0.26; 2) one or two preoculars; 3) three postoculars; 4) three anterior temporals followed by two posterior temporals; 5) eight supralabials, 3 rd to 5 th in contact with the eye, 7 th supralabial largest; 6) ten infralabials, the first five bordering the anterior chinshields; 7) 172 ventrals (plus two preventrals) in the one single male; 8) anal plate divided; 9) 96 divided subcaudals in the one single male; 10) reduction of dorsal scales rows from 19 to 17 scale (VS19TO17) occurring above ventral scale position 108; 11) reduction of the tail dorsal scales rows from 8 to 6 (SC6TO4) occurring above subcaudal 63; 12) dorsal scales in 19-19-17 rows, all distinctly keeled.
Description of the holotype. Dorsal scale rows 19-19-17, all distinctly keeled. Scales on posterior part of head and temporal region keeled. Reduction from 19 to 17 dorsal scale rows occurs at the level of the 3 rd and 4 th dorsal scale rows and between the 107 th and 109 th ventrals. There are 172 ventrals and 2 preventrals with 96 pairs of subcaudals. Anal divided. The scale rows reduction formula on the tail is:
9(4 + 5) 23(3 + 4) 63(2 + 3) 10 8 6 4 9 (4 + 5) 23 (3+ 4) 63 (2 + 3)
Rostral visible dorsally. Internasals subtriangular, wider than long. Prefrontals large, extending laterally onto side of head. Frontal longer than wide. Supraoculars broadly in contact with prefrontal, with supraocular two-thirds as broad as frontal. Single loreal. Preoculars 2 (left) or 1 (right); postoculars 3/3. Temporals 3+2 on both sides. Supralabials 8/8, the 3 rd to 5 th bordering orbit. Infralabials 10/10, first 5 contacting anterior chinshields. Nostrils lateral, nasals are undivided. In preservative, the dorsal body and upper tail surfaces are dark gray. Labials and ventral surface of head faint white. A faint white stripe is present on each side of the body. SVL 495 mm, TL 130 mm, ratio TL / SVL 0.26.
Etymology. This species is named after the herpetologist Frank T. Burbrink of Peoria, Illinois, in recognition of his contributions to snake systematics and evolution. We suggest the common name of this new species be Burbrink’s Keelback in English and Chayu Fulianshe (Chinese).
Distribution. This species is currently known only from the type locality ( Fig.1 View FIGURE 1 ). The specimen was collected on a road located in an evergreen forest close to a town ( Fig. 3 View FIGURE 3 ). It is likely that the new species also occurs in northern Burma and/or extreme eastern India given the proximity of the type locality to their borders. No data on its feeding and reproduction are available.
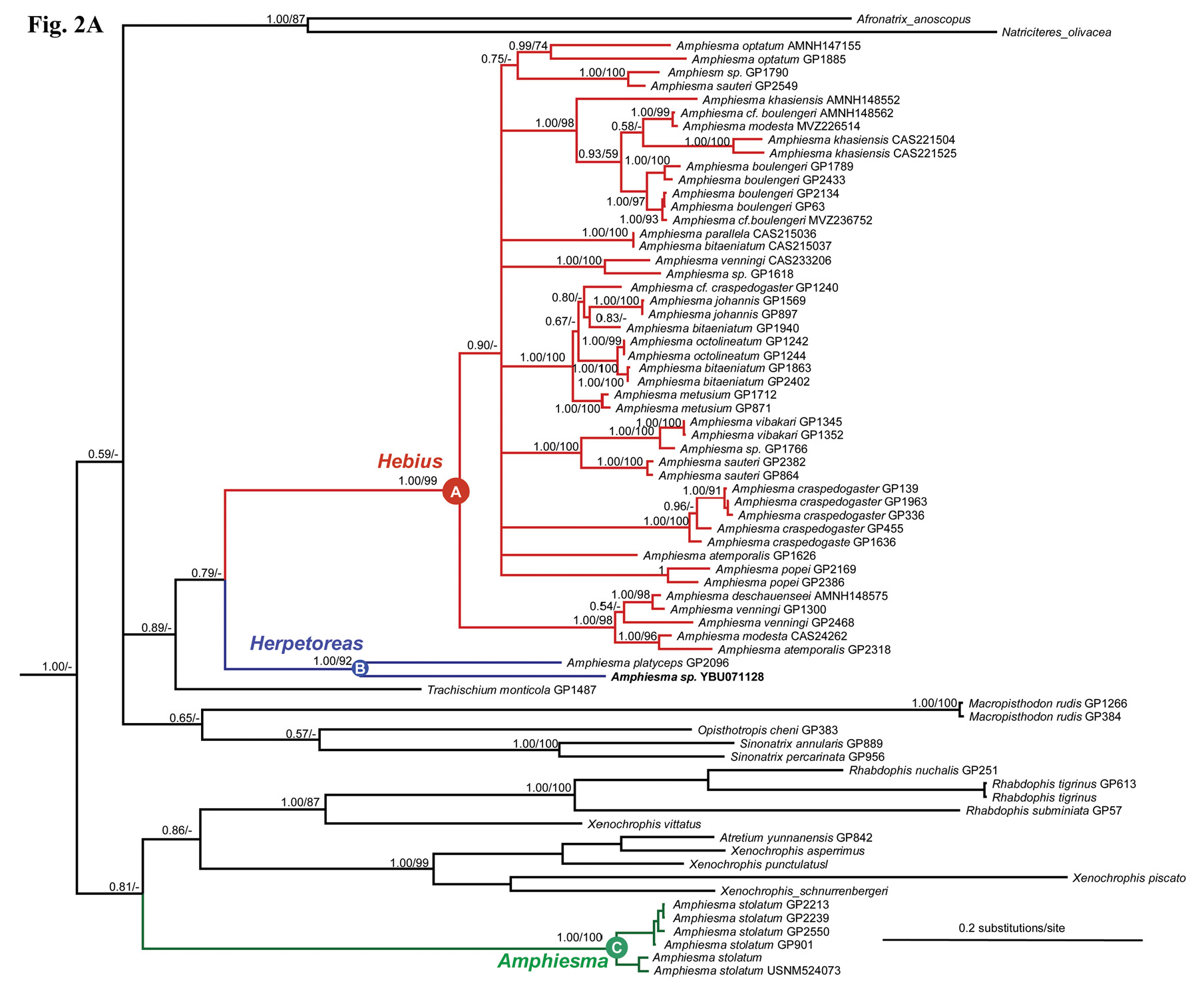
Remarks. In molecular phylogenies, H. burbrinki is closely related to H. platyceps ( Fig. 2 View FIGURE 2 ). Besides their differences in external morphology, these species were significantly different in genetic distance. Based on cyt b sequences, the average divergence (P-distance) between them (13.6%) is much higher than those between some other species in clade A (e.g. 13.1% between H. optatum and H. parallelum , 4.6% between H. octolineatum and H. metusium ).
The new species is very similar to H. sieboldii in external morphology. But the former can be distinct from the latter by having: fewer ventrals (172 vs. 216), more subcaudals (96 vs. 90), more postoculars (3 vs. 2), and a lower TL / SVL ratio (0.26 vs 0.32). In addition, a faint white stripe is present on each side of the body in H. burbrinki , but is absent in H. sieboldii .
Notes on Hebius . In this work, it is not our main aim to explore and address species level taxonomy, but several species are apparently non-monophyletic, and should be examined in future studies. Previously, morphological work indicated that some species of Amphiesma sensu lato were poorly diagnosed and quite variable, contributing to frequent misidentification and confusion between these species ( David et al. 2007, 2013). This misidentification problem was also apparent in this work based on multilocus molecular sampling. It is particularly true for the species within the genus Hebius . These species include H. bitaeniatum , H. sauteri , H. craspedogaster , H. venningi , and H. modestum ( Fig. 2C View FIGURE 2 ).
The species H. sauteri is widely distributed in China (including Taiwan) and Vietnam ( Zhao et al. 1998). Based on the counts of ventals and subcaudals, Malnate (1962) divided it into three subspecies (see Zhao et al. 1998). The populations from Taiwan and southeastern China are assigned to H. s. sauteri , and those from Sichuan belong to H. s. maxima . However, molecular results presented here suggest they are not close relatives. So, it is likely that these populations represent two different species, and that H. s. maxima should be given a full species rank as H. maxima . Another example is H. modestum . A sample of H. modestum from Yunnan, China (CAS 234262) is much distinct from another one from Vietnam (MVZ 226514). Because most samples analyzed here were loaned from museums and colleagues, we are not sure whether these questions have arisen from misidenfification or they represent undescribed new taxa. A comprehensive study with more complete samples as well as various methods is highly desirable in resolving the taxonomies at species level.
Finally, taxonomic information on the revised genera Amphiesma , Hebius , and Herpetoreas are provided below. However, we note that the generic diagnoses of these genera have been modified from published sources cited herein, and thus may not be precise. More complete diagnoses will depend on comprehensive morphological studies, but even then, the evident degree of convergence may complicate this substantially ( Pyron & Burbrink, 2009).
| R |
Departamento de Geologia, Universidad de Chile |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.