Halistemma striata Totton, 1965
|
publication ID |
https://doi.org/10.11646/zootaxa.3897.1.1 |
|
publication LSID |
lsid:zoobank.org:pub:CB622998-E483-4046-A40E-DBE22B001DFD |
|
persistent identifier |
https://treatment.plazi.org/id/03FC87BC-FFF5-FFC2-FF62-ADF46144FA84 |
|
treatment provided by |
Felipe |
|
scientific name |
Halistemma striata Totton, 1965 |
| status |
|
Halistemma striata Totton, 1965 View in CoL
Halistemma striata Totton, 1965, pp. 59–60 View in CoL , text.—fig. 24; Margulis, 1969, p. 23, fig. 2 (4,5); Stepanjants, 1977b, p. 61, fig. 3В–Д; Pugh, 1999a, p. 482, fig. 3.12.
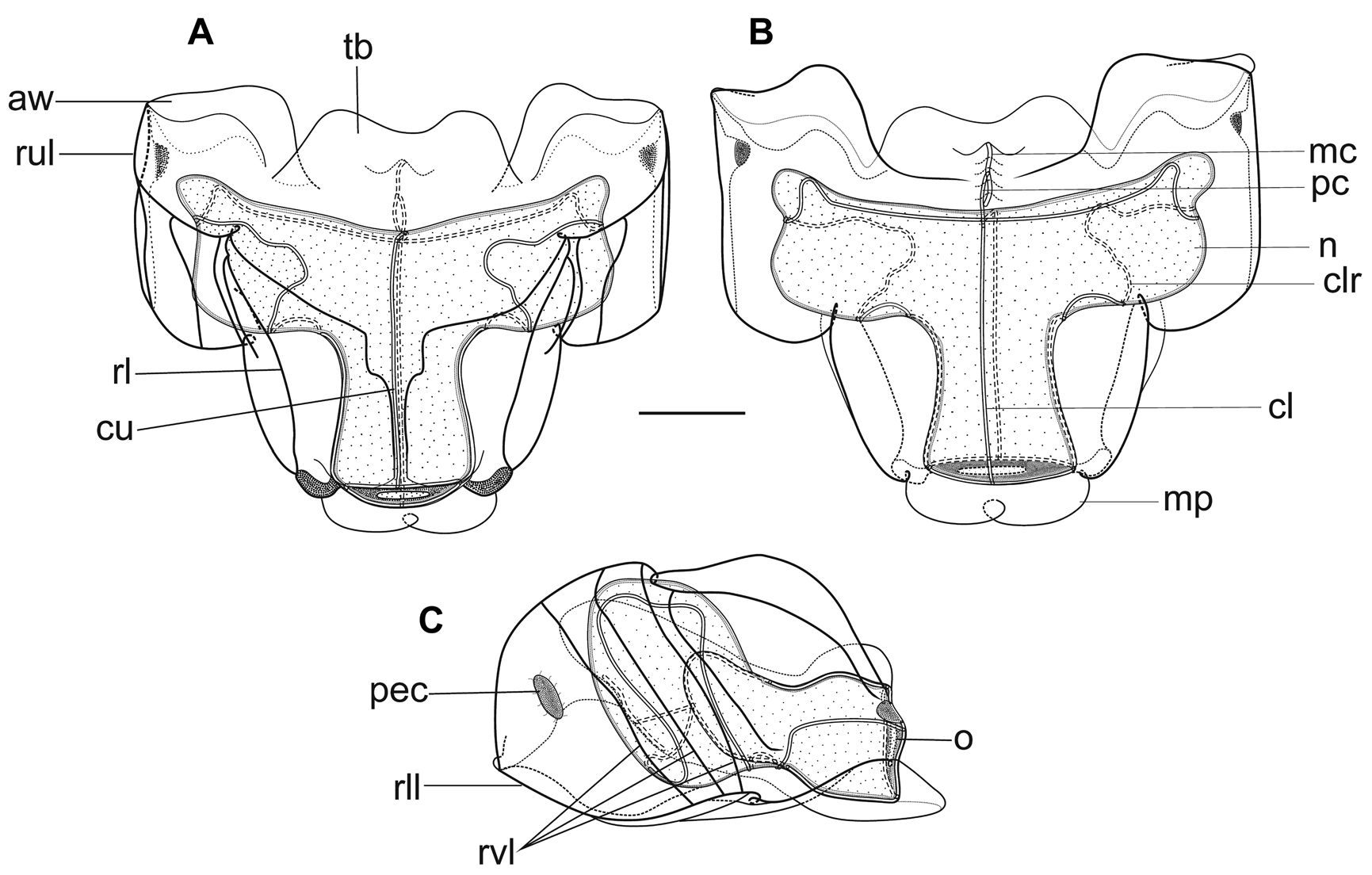
Diagnosis. Nectophores with four pairs of vertical lateral ridges. The proximal three are complete, uniting with both the upper and lower lateral ridges, while the most distal arises from the lateral ridge, a short distance after the latter divides from the upper lateral ridge, and does not join with the lower lateral ridge. The lateral ridge is complete, reaching the ostium. Close to the ostium the upper lateral ridges diverge and run laterally before petering out. Two types of adult bract, with the Type A bracts having two different forms. Tentilla with small involucrum covering the proximal 1.5 spirals of the cnidoband. Terminal filament without cupulate process at distal end.
Material examined: Two specimens collected by the Johnson-Sea-Link (JSL) submersibles; one from the Bahamas and from off the Florida coast in the Gulf of Mexico.
JSL II 990 - SS12 29 October 1984 25°22.5'N 77°54.5'W depth 706 m GoogleMaps
JSL I 2664 -DS8 16 September 1995 c. 27°0.0'N c 85°0.0'W No depth recorded
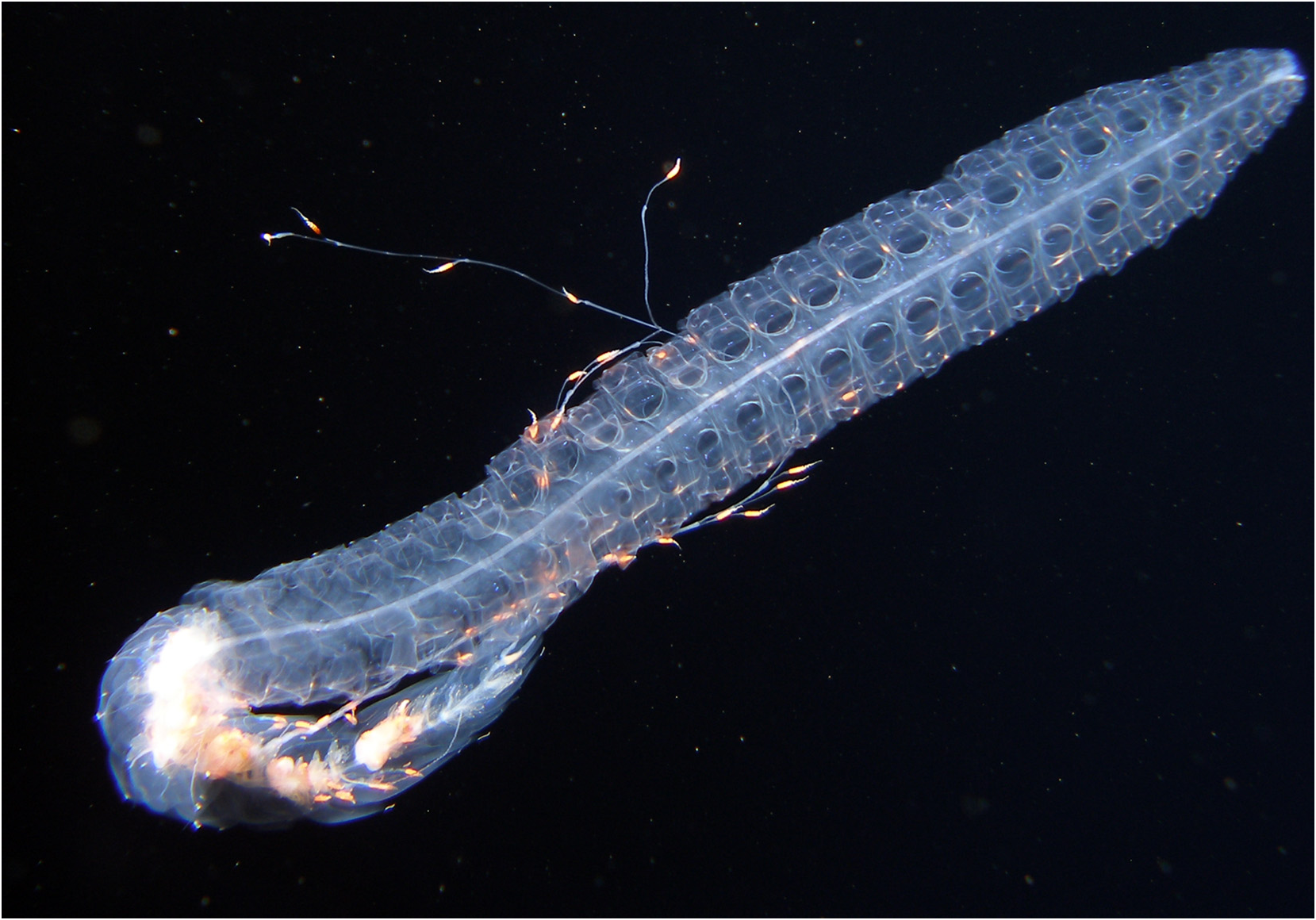
Description: The description will be based entirely on the JSL II Dive 990-SS 12 specimen, although not all of the specimen was preserved, as the other is even less complete and in poor condition. Photographs of this specimen were taken, but they are not good enough to reproduce. However, an in situ photograph of what most likely is Halistemma striata , although it was not collected, was kindly sent to me by Dr. Mark Benfield and is reproduced as Figure 39 View FIGURE 39 . It was taken in the Gulf of Mexico at 26°52.69'N, 93°16.13'W at 768 m, during the Gulf SERPENT Project. The whole specimen and, in particular, the robustness of the bracts, together with the long patches of nematocysts at their distal tips bears a close resemblance to the photographs of the JSL II Dive 990-SS 12 specimen. Although the bracts of H. rubrum also have relatively long patches of nematocysts the bracts are small and less robust (see video).
Nectosome: The nectosome and pneumatophore for both specimens was not found with the preserved material and so it was not possible to confirm that nectophores were budded off on the dorsal side of the stem. However, at least six specimens of Halistemma striata were collected at Discovery St. 10523#6 (see below) and these had pneumatophores measuring c. 1.45 mm in length and 0.75 mm in diameter, and it was also clear that the nectophores were attached to the dorsal surface of the stem.
Nectophores: No nectophoral buds or young nectophores were found with the specimen, and the eighteen preserved nectophores were all of a similar size, with a maximum length of 23 mm and a maximum width of 25 mm ( Figure 40 View FIGURE 40 ). The ridge pattern is characteristic for the species. There were four vertical lateral ridges on each side. The three more proximal ridges were complete, running obliquely down the lateral sides of the nectophore from the upper lateral ridge to the lower one at an angle of c. 30°, as Totton (1965) noted. The most proximal bent slightly distad close to its junction with both the upper and lower lateral ridges. The next bent distad close to the upper lateral ridge, while the third was almost straight. In the preserved state, this pair of ridges was found in the depths of furrows running down the sides of the nectophore, but this was probably a preservation artefact. The distal-most ridge that branched from the upper lateral soon bifurcated to form the fourth vertical lateral ridge and the lateral one. The vertical lateral one continued obliquely down the side of the nectophore but before reaching the lower lateral it curved toward the ostium and petered out. The lateral ridge ran in a broad arc toward the ostium, and formed part of the edge of the large lateral ostial process.
The axial wings were prominent, but quite short, and were truncated proximally, while laterally they were triangular in shape. Approximately in the middle of this lateral facet was an elongate concavity that was lined with ectodermal cells, which probably were sites of bioluminescence. These were the only patches of cells found on the nectophores. The broad thrust block did not extend, proximally, as far as the axial wings and had a wide median emargination.
The ascending branch of the mantle canal was slightly longer than the descending one, and in the preserved state it ran along a raised process on the lower side of the thrust block. The pedicular canal was about half its length. The latter reached the nectosac on its lower side, just below its apex, and immediately gave rise to the four radial canals. The lateral radial canals followed the typical Halistemma course, looping outward and slightly downwards toward the lateral sides of the nectosac; then looping up and down the lateral sides, passing briefly onto the lower side of the nectosac, before looping up to the mid height of the nectosac and travelling directly to join the ostial ring canal. Minor sub-loops were noted in several specimens.
Siphosome: The siphosome was relatively wide and robust, with the large bracts entirely covering the other zooids. The cormidia, even in the contracted state, were quite clearly separated from each other; although the arrangement of the zooids within each cormidium was difficult to discern.
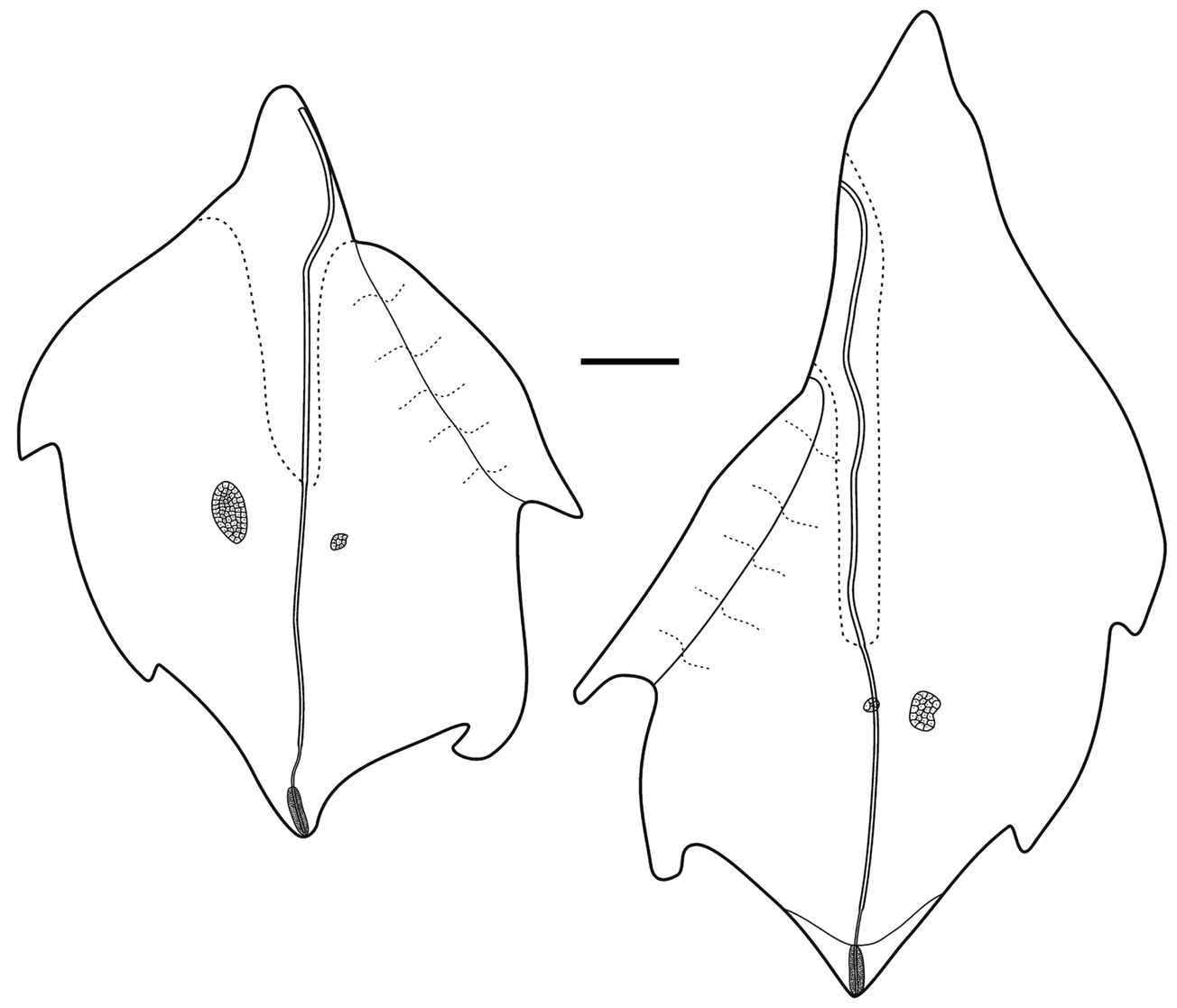
Bracts: There were basically two types of bract, although one, the Type A was present in two forms.
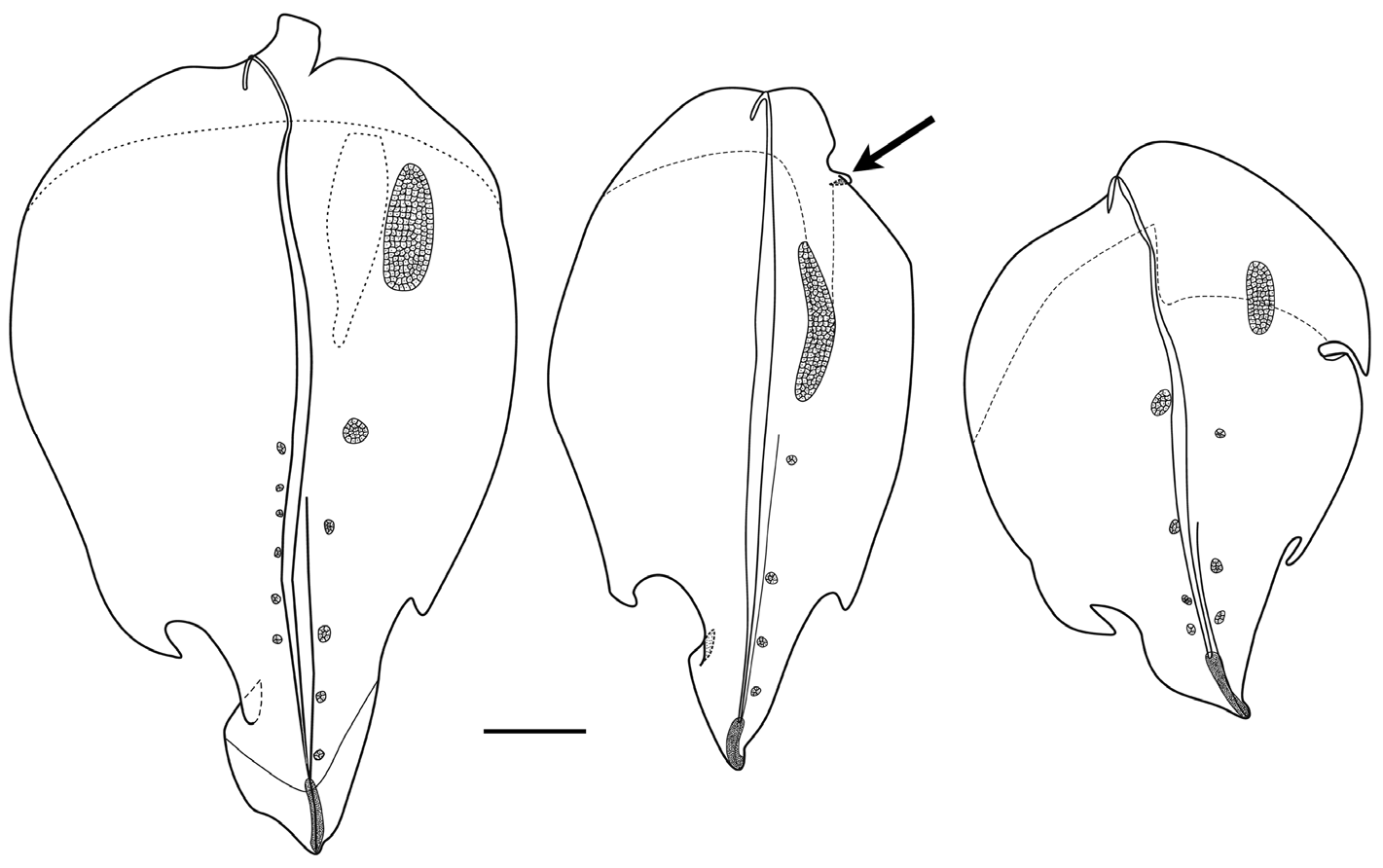
Type A —These bracts were robust measuring up to 63 mm in length and 32 in width ( Figures 41–42 View FIGURE 41 View FIGURE 42 ). They were widest toward the proximal end, while the distal end came to a point. Both forms of this bract had a weak median ridge in the distal half, where a pair of prominent, asymmetrically placed lateral teeth was present. There was a large patch of ectodermal cells, probably sites of bioluminescence, on one side of the bract in its proximal half, and up to six pairs of smaller rounded patches on either side of the mid-line in the distal half. At the distal end of the bract in the mid-line, there was a long narrow strip of nematocysts. The bracteal canal ran the full length of the bract and, particularly in the younger bracts ( Figure 41C View FIGURE 41 ) it was thickened for most of its length, but narrowed both proximally and distally. Proximally it ran up onto the upper side of the bract for a short distance and remained in contact with the lower wall of the bract for most of its length. Just before reaching the level of the proximal end of the patch of nematocysts it penetrated into the mesogloea and as a narrow canal continued obliquely upwards to the upper surface, where it continued below the nematocysts to the distal tip.
Type A-1. In addition to the pair of lateral teeth, on one side of the triangular distal process there was a distinct point, where the lateral walls of the bract overlapped each other. Usually the proximal patch of ectodermal cells was more elongate and less wide than that on the Type A-2 bracts. On the lower side of the bract the mesogloea was distinctly thickened close to the proximal end, being more extensive on the side where the ectodermal patch was present on the upper side. There was also a distal extension of thickened mesogloea on that side that continued for some distance down the bract; either forming part of the proximal thickening or completely separate from it. The distal strip of nematocysts was shorter than in the Type A-2 bracts.
One of the younger bracts ( Figure 41 View FIGURE 41 centre, arrowed) had a small, lateral cusp-like structure close to the proximal end, which somewhat resembled the situation in the Type A-2 bracts. However, this was unusual and, by the distal lateral process, it clearly was a Type A-1 bract.
Type A-2. The main difference that these bracts showed from the previous ones was the presence, proximally and on one side, of a distinctive circular indentation. The thickening of the mesogloea on the lower side was also arranged differently, being also exclusively on the same side as the indentation and proximal to it. A shorter distal extension was also present, but it lay closer to the mid-line.
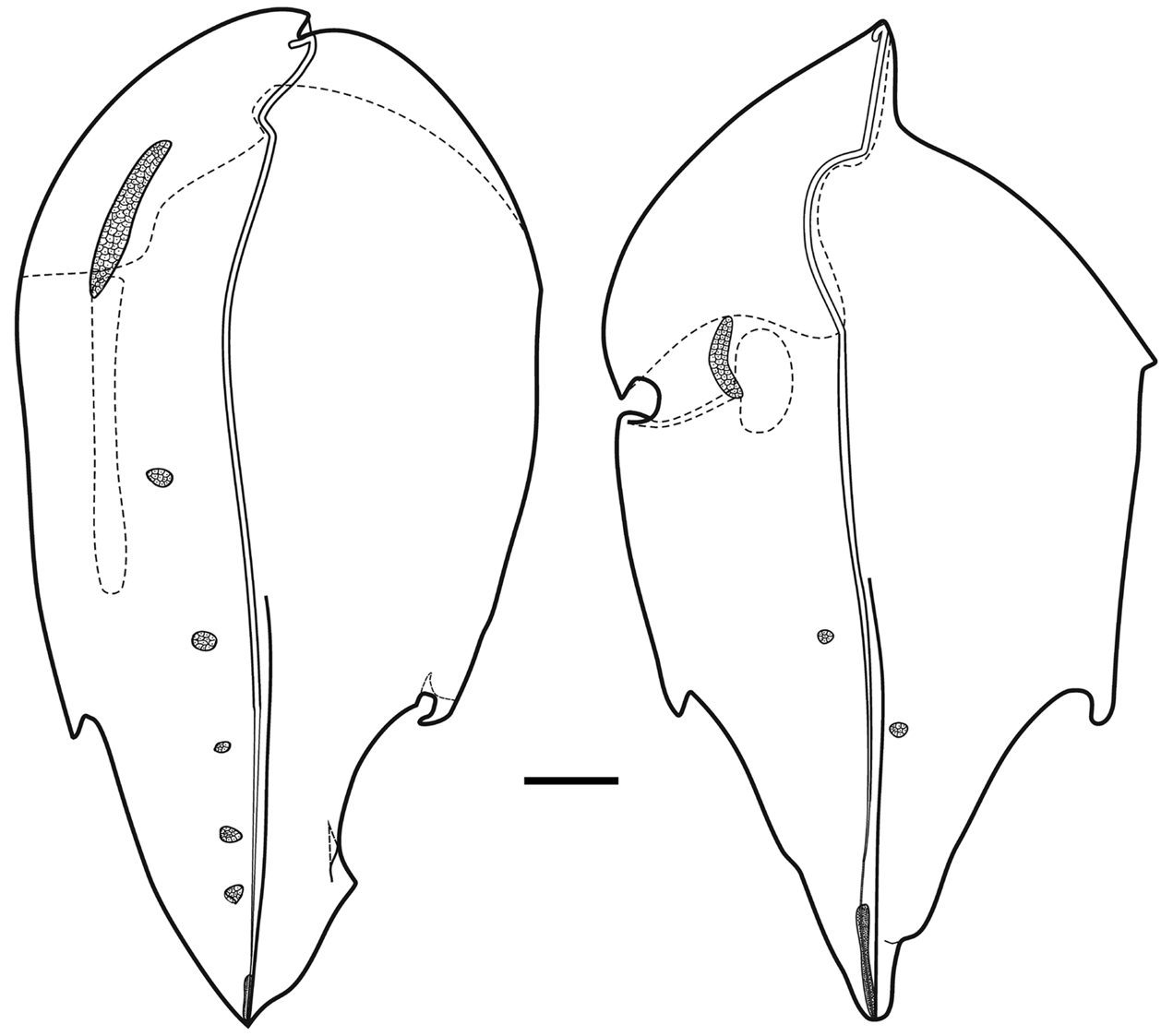
Type B —These bracts measured up to 52 mm in length and 29 mm in width ( Figure 43 View FIGURE 43 ). They were distinctly asymmetrical, foliaceous and pointed at both ends. There were two pairs of large asymmetrical lateral teeth, and a pair of ectodermal cells, of distinctly different sizes on the upper side in its distal half. The inner side of the bract, from distal to the origin of the bracteal canal to the more proximal lateral tooth, was distinctly thinner than the rest of the bract and formed a flap The bracteal canal arose on one side of the pointed proximal end and, as with the Type A bracts it ran along the median lower surface for most of its length, before narrowing and running obliquely up through the mesogloea to reach the relatively short strip of nematocysts, under which it continued to the distal tip. The type of nematocysts present was not investigated. In the proximal half of the bract, the canal was raised on a thickened mesogloeal process.
Gastrozooid and tentacle: The large gastrozooids showed no unusual features, while the tentacles, in their preserved and contracted state, were relatively thickened with musculature ( Figure 44 View FIGURE 44 ). The photographs of the JSL II Dive 990-SS 12 specimen show that the basigaster was deep red in colour, while the stomach region was milky white .
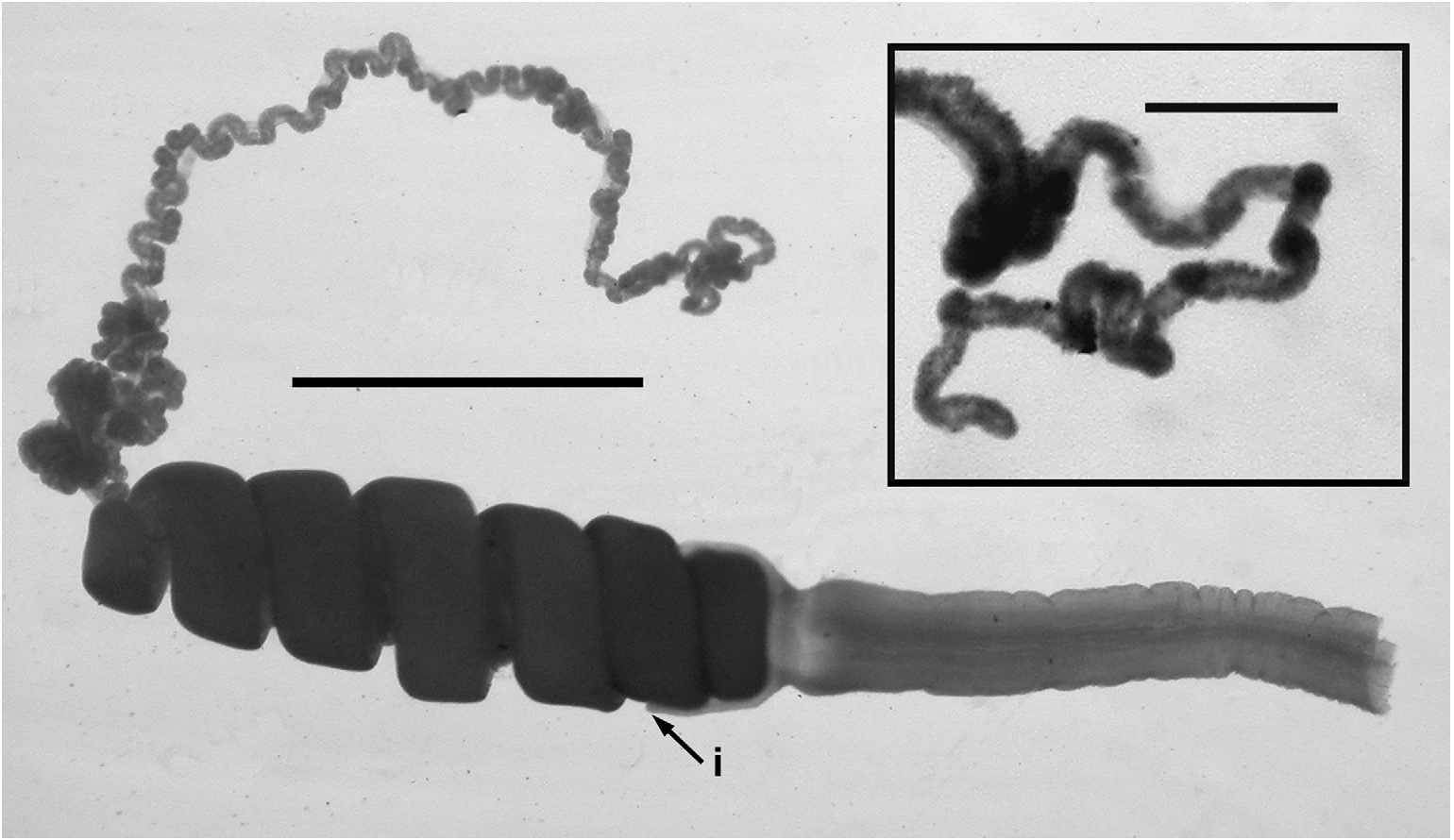
Tentilla: A mature tentillum is shown in Figure 45 View FIGURE 45 . In life the cnidobands were orange in colour, while the terminal filaments were white. The long, thick, rigid in preservation, pedicel ended distally in a small involucrum, which covered the proximal 1.5 spirals of the cnidoband ( Figure 45 i View FIGURE 45 ). The latter consisted of up to ten spiral coils, particularly in the JSL Dive 2664 specimen, although c. 7 was more common in the JSL Dive 990 one. There were relatively few, but large stenoteles, measuring c. 92 x 40 µm, confined to the sides of the proximal end of the cnidoband. The remainder of the cnidoband was filled with innumerable banana-shaped nematocysts, presumably anisorhizas that measured c. 81 x 12.5 µm. Within the cnidoband the double elastic band was very prominent. The terminal filament contained two types of nematocysts, which as discussed above were presumably acrophores and desmonemes, measuring c. 31 x 11 and c. 25 x 10.5 µm, respectively. The long, loosely and irregularly coiled terminal filament did not end, distally, in a cupulate process, but was just rounded off ( Figure 45 View FIGURE 45 , inset).
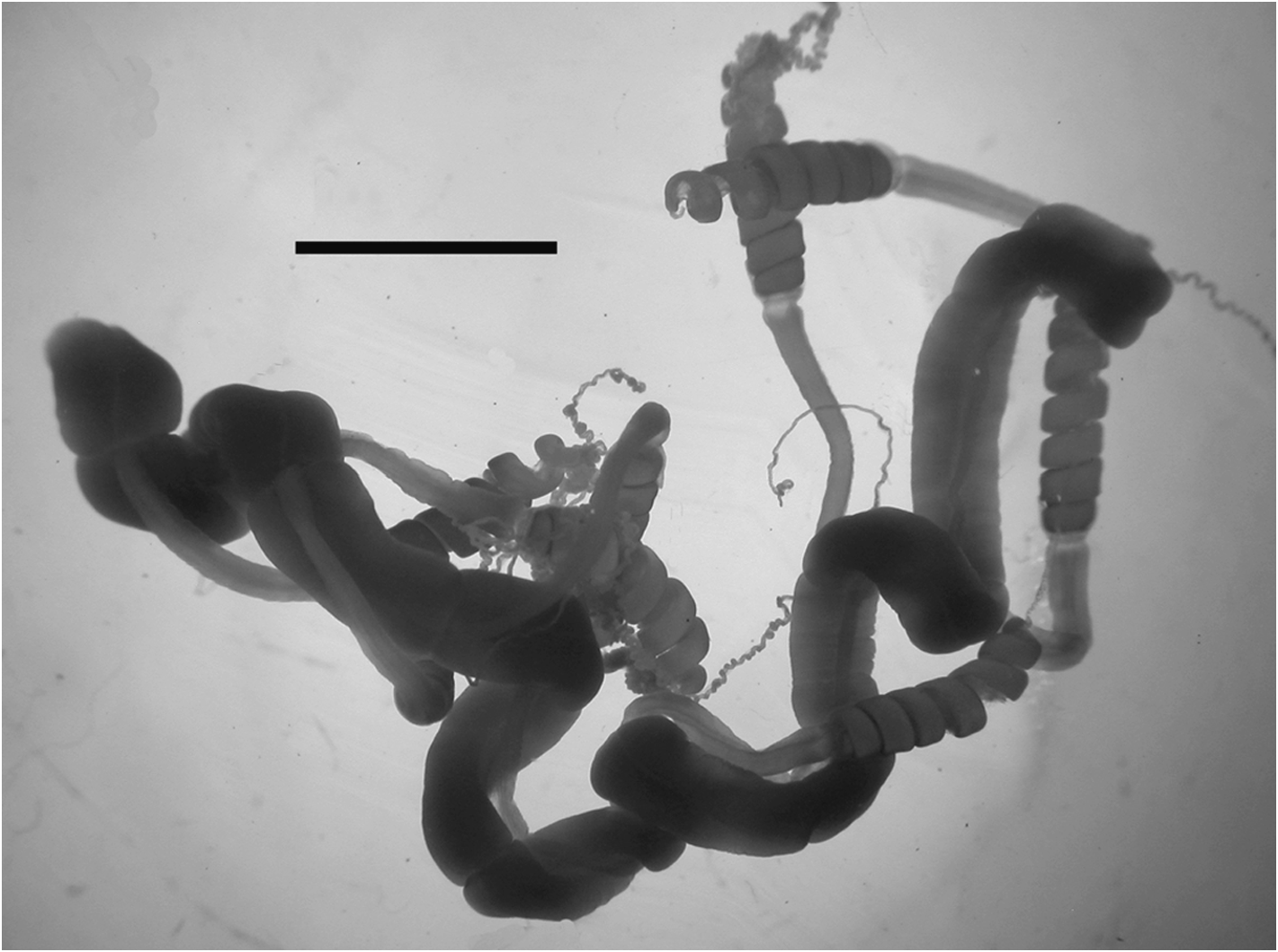
Palpons: The thin-walled palpons ( Figure 46) were generally featureless, but were clearly divided into a narrow tubular proximal region and an expanded distal part, with a distinct terminal proboscis surrounding the terminal opening. In their preserved state many palpons exceeded 15mm in length. At their base they bore a long palpacle. No nematocysts were found on either the palpon or the palpacle.
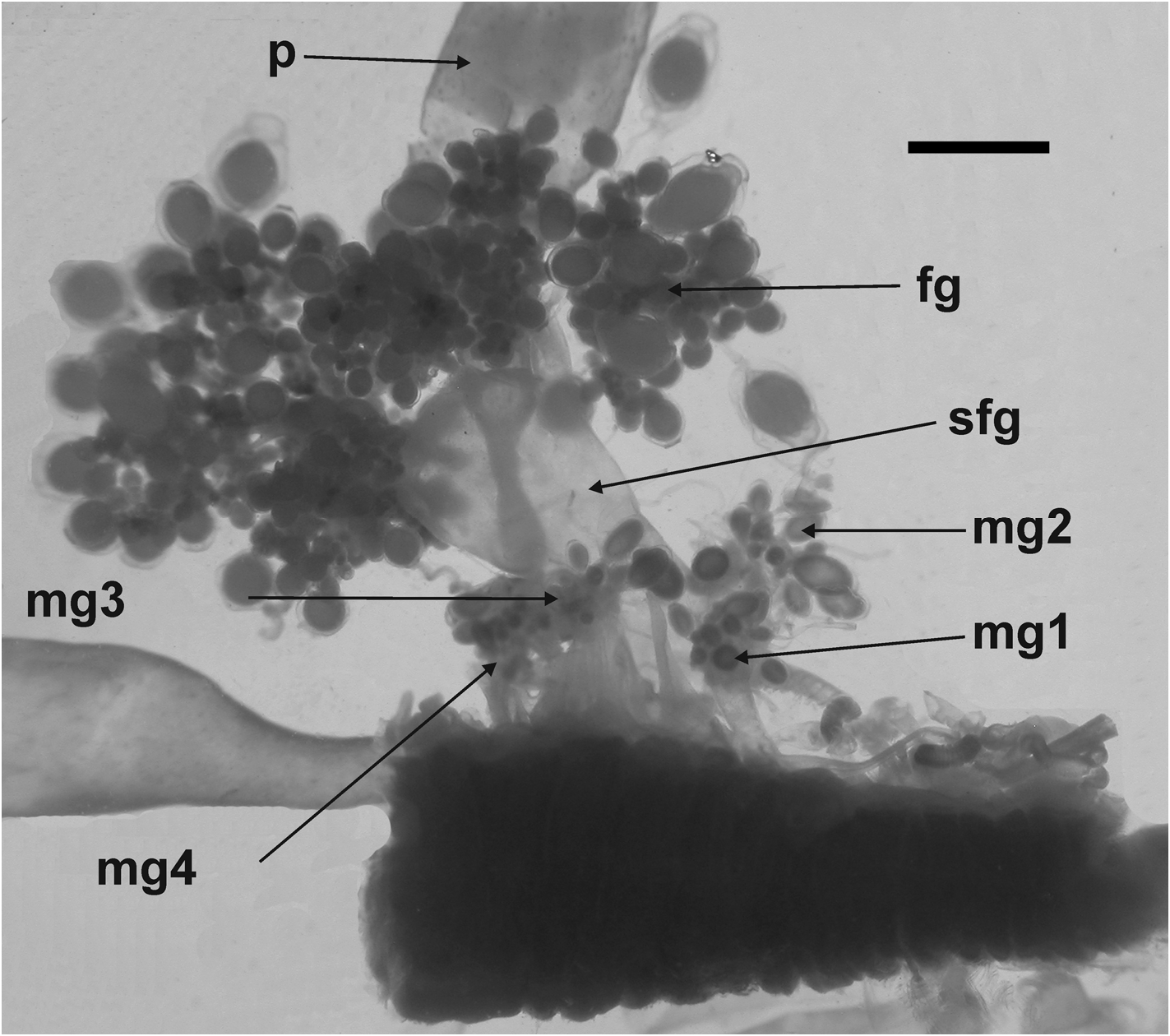
Gonophores: The female gonodendron was borne on a very broad, long but thin-walled stalk ( Figure 47 View FIGURE 47 ), with strengthening rings towards its base. This usually divided into two main branches on which the racemes of female gonophores were arranged. Mature female gonophores were quite variable in shape measuring up to 3 mm in length and 2 mm in width, and had a broadly open distal orifice ( Figure 48).
There were four male gonodendra in each cormidium, which is the most that have been observed for any Halistemma species. H . rubrum is the next most complex (see also the discussion regarding gonodendra in H. foliacea ), which has three. All the other species, where known have a single male and a single female gonodendron, with the male one anterior to the female. For H. striata there is one male gonodendron anterior to the female one, alongside which sits the second male one. Then there are two further male gonodendra posterior to the female one.
The male gonophores were long and cylindrical, measuring up to 4.5 mm in length and 1.6 mm in diameter ( Figure 49), and possessed long proximal pedicels. Distally the mouth opening was wide open, like that of the female gonophores. The gonodendral cluster usually consisted of a small bundle of gonophores, usually consisting of a mature one distally and a group of developing gonophores proximally.
Nectalia - stage: Neither of the JSL specimens was at the Nectalia post-larval stage.
Remarks: The nectophores of Halistemma striata are easily identified by the presence of four pairs of vertical lateral ridges. They are also the largest of any Halistemma species , and the largest nectophores in the present material, measuring 25 m in width, were considerably larger than the range of 8–19 mm for Totton's (1965) original material. They are slightly larger than the nectophores of H. foliacea , but that species has just two pairs of vertical lateral ridges. The Type A-2 bracts are also very distinctive, as is the distal end of the terminal filament of the tentillum. In most Halistemma species there is a cupulate process present, which differs morphologically from species to species. In H. rubrum the terminal filament ends in a tightly coiled tapering process, while in H. striata no distinct process is present.
Distribution: Totton's (1965) original material came from the Beebe collections centred around 32°12'N, 64°36'W south of Bermuda, and two Discovery stations, in the central south ( Discovery St. 81, 32° 45'S, 8° 47'W), and equatorial north Atlantic ( Discovery St. 704, 3° 37'7'N, 29° 14'W). Margulis (1969) found Halistemma striata at 18 sites throughout the Atlantic Ocean, all but two of which were found between 40°N and 20°S; the others
being at c. 56°N, 21.5°W and 42°S, 38.5'W. Stepanjants (1975) had another Atlantic record, from north of Puerto Rico. It has also been found in eight recent Discovery samples, shown in decreasing latitudinal order GoogleMaps :
Station Nect Bract Date Latitude Longitude Depth Range (m)
7711#09 5 18-May-1971 52°50.5'N 20°09.3'W 605–700
9791#10 4 12 06-May-1978 49°28.5'N 14°00.8'W 500– 600
53305#32 1 21-Apr-1994 49°14.1'N 12°15.2'W 200–310
11058#03 1 11-Apr-1984 45°34.9'N 13°36.4'W 95–200
8508#33 48 107 16-Apr-1974 44°04.8'N 13°07.6'W 302–400
7800#02 55 138 18-Feb-1972 19°46.7'N 22°32.7'W 0–1000
7089#23 29 16-Nov-1969 17°40.7'N 25°19.7'W 110–200
6662#11 102 271 15-Feb-1968 11°03.0'N 19°52.6'W 510–590
10523#06 139 232 10-May-1982 05°41.9'S 00°29.4'E 400–500
The Beebe collections, from Bermuda, contain at least four specimens, namely :
Specimen No. 29365 600 fm 22 nectophores
Specimen No. 29372 700 fm 23 nectophores
Specimen No. 301371 600 fm 1 nectophore.
Specimen No. 312196 600 fm 6 nectophores, 2 bracts.
Stepanjants (1977a) gave the first Pacific Ocean record, from south-east of the Solomon Islands, and Margulis (1987) gave the second from the region of 41– 48°S 106– 110°W. Recently, Yu (2006) has added a record from Taiwanese waters, and it has also been found from Japanese waters (Dhugal Lindsay, personal communication). It has not been noted during the extensive observations in the Monterey Bay, California, region, nor in the Gulf of California. In conclusion, it appears that Halistemma striata is a rare species with an extensive geographical distribution, generally in the warmer waters of the World's oceans.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Halistemma striata Totton, 1965
| Pugh, P. R. & Baxter, E. J. 2014 |
Halistemma striata
| Pugh, P. R. 1999: 482 |
| Stepanjants, S. D. 1977: 61 |
| Margulis, R. Ya. 1969: 23 |