Microzoanthus kagerou Fujii & Reimer, 2011
|
publication ID |
https://doi.org/ 10.11646/zootaxa.3796.1.4 |
|
publication LSID |
lsid:zoobank.org:pub:66323922-2C76-4AB7-98E6-59205AF86DBA |
|
DOI |
https://doi.org/10.5281/zenodo.5672480 |
|
persistent identifier |
https://treatment.plazi.org/id/32546E5C-A523-FFFF-FF52-F916E012FBEA |
|
treatment provided by |
Plazi |
|
scientific name |
Microzoanthus kagerou Fujii & Reimer, 2011 |
| status |
|
Microzoanthus kagerou Fujii & Reimer, 2011 View in CoL
Figure 7, 8. Morphbank species collection 829717.
Material examined. USNM 1150463, paratype.
Diagnosis. Colonial Microzoanthus with endodermal marginal musculature; marginal muscle to 331 Μm length, composed of as many as 16 mesoglea pleats. Mesenterial arrangement macrocnemic. Columnar mesoglea adjacent siphonoglyph to 21 Μm width. Occurring at 1–20 m throughout the temperate and tropical Pacific Ocean, free-living, and lacking scleroprotein skeleton. Coenenchyme and polyps transparent or faintly red. Tentacles and mesenteries 20–26, oral disk may have fluorescent green pigments and is calathiform when expanded, capitular ridges 10–13 when discernible. Largest expanded polyps 10 mm long, 3 mm diameter.
FIGURE 7. Histology of Microzoanthus kagerou (10 Μm sections). Labeled features include actinopharynx (A), column wall (CW), dorsal directives (DD), endodermal marginal musculature (EMM), encircling sinus (ES), fifth mesentery (5th), oral disk (OD), peristome (P), siphonoglyph (S), tentacles (T), ventral directives (VD); measurements of capitular tissue width made at black arrow, measurements of column tissue width made at broken arrow, measurements of siphonoglyph tissue width made at gray arrow. A. Longitudinal section of contracted polyp at capitulum showing endodermal marginal musculature. B. Longitudinal section of contracted polyp. C. Cross-section of contracted polyp at level of actinopharynx showing dorsal directives and fifth mesenteries. D. Cross-section of contracted polyp at level of actinopharynx showing ventral directives and siphonoglyph.
Description. Colony. Coenenchyme transparent or faintly red and connects polyps (often> 1 cm apart) as a narrow stolon; infiltrated with sediment. Not known to associate with other invertebrates ( Fujii & Reimer 2011).
Polyp. Capitular ridges largely imperceptible (Morphbank 830702), 10–13 when observable ( Fujii & Reimer 2011). Tentacles and column transparent or faintly red; oral disk same color (but can have fluorescent green pigments) and is distinctly calathiform as margin extends above oral disk greater than one-fourth its diameter ( Fujii & Reimer 2011). Polyps to 3 mm in diameter (expanded) extend to 10 mm from coenenchyme; column wall infiltrated with sediment. Tentacles 20–26, dicyclic, and expand in length to 2–3 times diameter of column ( Fujii & Reimer 2011).
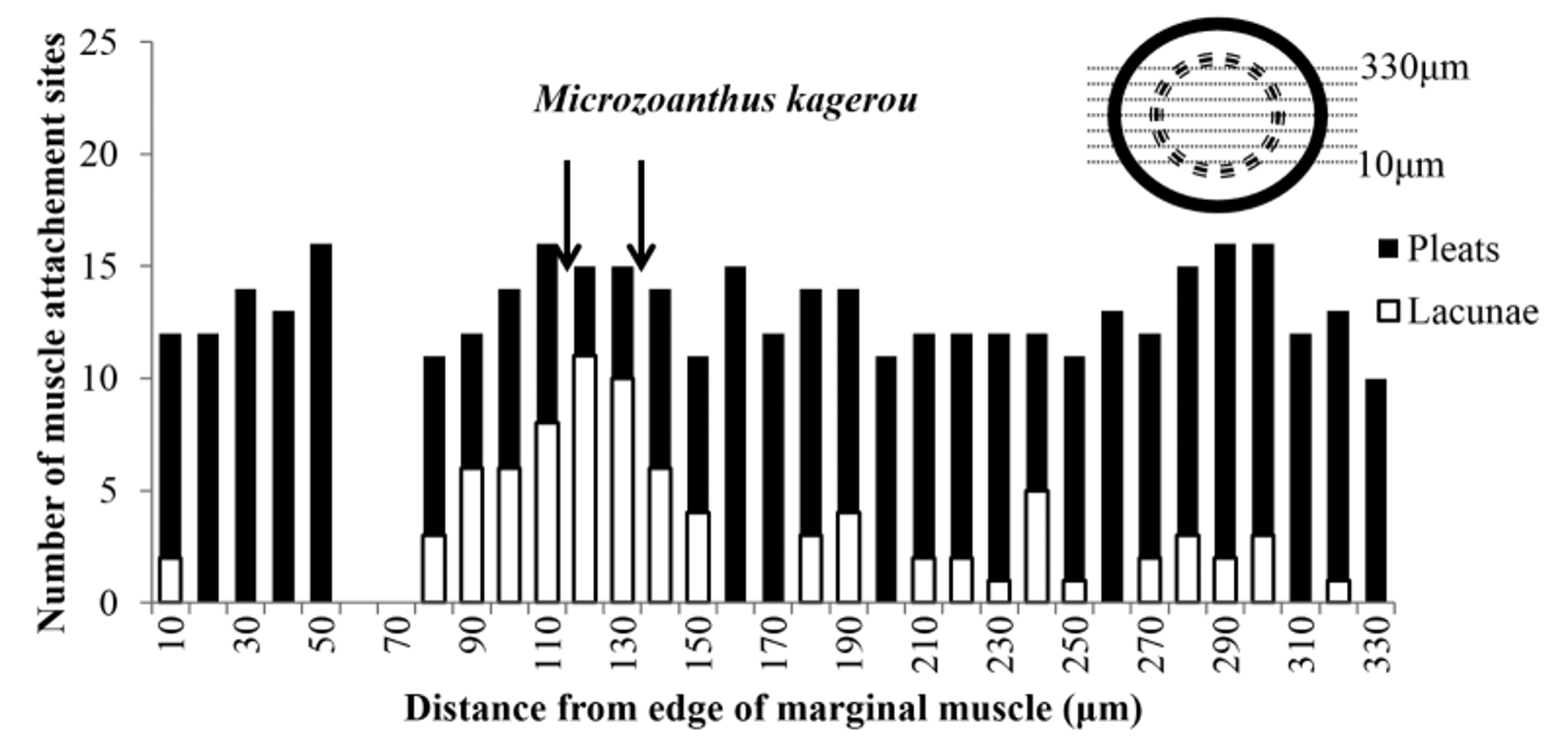
Internal Anatomy. In longitudinal section (Morphbank collection 829719), marginal musculature endodermal, muscle fibers anchored to 10–16 (x = 13, n sections = 31) unbranched mesogleal pleats (Fig. 7A). Mesogleal pleats decrease in size proximally. Fibers enclosed in lacunae in few sections as muscles transverse mesenteries (Fig. 7A, 8). Length of marginal musculature (Fig. 7A) 268–331 Μm (x = 298, n sections= 10), width at widest point (Fig. 7A) 34–65 Μm (x = 52, n sections = 10). Length of largest mesogleal pleats supporting the muscle fibers (Fig. 7A) 15–36 Μm (x = 27, n sections = 10). Large lacunae resulting from dissolution of encrustations throughout capitular ectoderm which occasionally distort ectodermal surface of mesoglea (Fig. 7B). In the region of capitulum (proximal to terminus of marginal musculature; Fig. 7A) ectoderm is 6–22 Μm (x = 12, n sections = 10), mesoglea 7–16 Μm (x = 11, n sections = 10) and endoderm 8–15 Μm (x = 12, n sections = 10) width.
In cross section at actinopharynx (Morphbank collection 829718), mesenteries 22, fifth mesenteries macrocnemic (Fig. 7C). Dorsal directives with slight flair of mesoglea at the free border, similar to non-directive imperfect mesenteries (Fig. 7C). Ventral directives (Fig. 7C, D) supported by mesoglea 105–164 Μm (x = 134, n sections = 10) from column to siphonoglyph, 2–4 Μm (x = 3, n sections = 10) width, at retractor muscles 6–15 Μm (x = 11, n sections = 10) width, and homomorphic at column; similar to non-directive perfect mesenteries (Fig. 7C, D). Actinopharynx without esophageal furrows (Fig. 7C, D). Siphonoglyph distinct and U-shaped (Fig. 7D); ectoderm is 33–88 Μm (x = 46, n sections = 10), mesoglea 4–8 Μm (x = 6, n sections = 10), and endoderm 6–10 Μm (x = 8, n sections = 10) width. Adjacent siphonoglyph (Fig. 7D), column ectoderm is 9–45 Μm (x = 27, n sections = 10), mesoglea 11–21 Μm (x = 14, n sections = 10), and endoderm 10–14 Μm (x = 12, n sections = 10) width. Elliptical lacunae form an encircling sinus just beneath ectodermal surface of mesoglea (Fig. 7D). Large lacunae resulting from dissolution of encrustations throughout columnar ectoderm, which occasionally distort ectodermal surface of mesoglea (Fig. 7C, D).
Cnidae. Tentacles and pharynx: basitrichs, holotrichs, spirocysts; filament: basitrichs, mastigophores, holotrichs, spirocysts; column: holotrichs (see Table 1 View TABLE 1 of Fujii & Reimer 2011 for size and frequency).
Distribution. Colonies free-living under rubble and interstices at 1–20 m throughout the temperate and tropical Pacific Ocean ( Fujii & Reimer 2011).
Remarks. Microzoanthus kagerou differs from Microzoanthus occultus Fujii & Reimer, 2011 in colony morphology ( M. kagerou has larger colonies whose polyps are connected by well-developed stolons), cnidae (microbasic p-mastigophores are absent from the actinopharynx of M. kagerou ), and nucleotide (COI, 16S rDNA, and ITS rDNA gene) sequences ( Fujii & Reimer 2011). The original description of M. kagerou included traditional (higher-taxon defining) morphological features in addition to cnidae and nucleotide sequences, but our observations of the paratypes are not entirely consistent with those of the original authors. We concur with Fuji and Reimer (2011; see Fig 6C) on the number of marginal muscle-attachment points (16) and the lack of mesogleal encrustations. We differ from Fuji & Reimer (2011) in their interpretation of the marginal musculature as being transitional from mesogleal to endodermal, whereas we interpret it as endodermal musculature that is sporadically and partially enclosed in lacunae as muscles traverse mesenteries ( Fig 8 View FIGURE 8 ). Of the 31 serial sections we examined, 9 sections (not in a series) are completely endodermal (pleats only), 2 have attachment sites composed of more than half lacunae, and most (21) have small portions of sites enclosed in lacunae ( Fig. 8 View FIGURE 8 ). The location of these lacunae appear to be randomly distributed and are observed in proximal, central, and distal portions of the marginal muscle, a situation inconsistent with the interpretation of a transitional musculature. Furthermore, Fujii & Reimer (2011) describe mesoglea in an unspecified region of the polyp as “approximately 90 Μm and show histology with mesoglea that approximates that width at the capitulum (judging by the scale bar in Fig. 6B of Fujii & Reimer 2011) while measurements from histology reported here do not exceed 21 Μm (65 Μm if pleats of marginal musculature are included). We consider the discrepancies in measurements to expand the circumscription of morphological variation of M. kagerou .
| USNM |
Smithsonian Institution, National Museum of Natural History |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.