Callomyia gilloglyorum Kessel
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4111.5.1 |
|
publication LSID |
lsid:zoobank.org:pub:1286E111-8C60-47AB-B2A2-36D3BFB6CA3F |
|
DOI |
https://doi.org/10.5281/zenodo.5621988 |
|
persistent identifier |
https://treatment.plazi.org/id/3C7A0266-7359-971C-C2B1-2E1519B4FE36 |
|
treatment provided by |
Plazi |
|
scientific name |
Callomyia gilloglyorum Kessel |
| status |
|
Callomyia gilloglyorum Kessel View in CoL
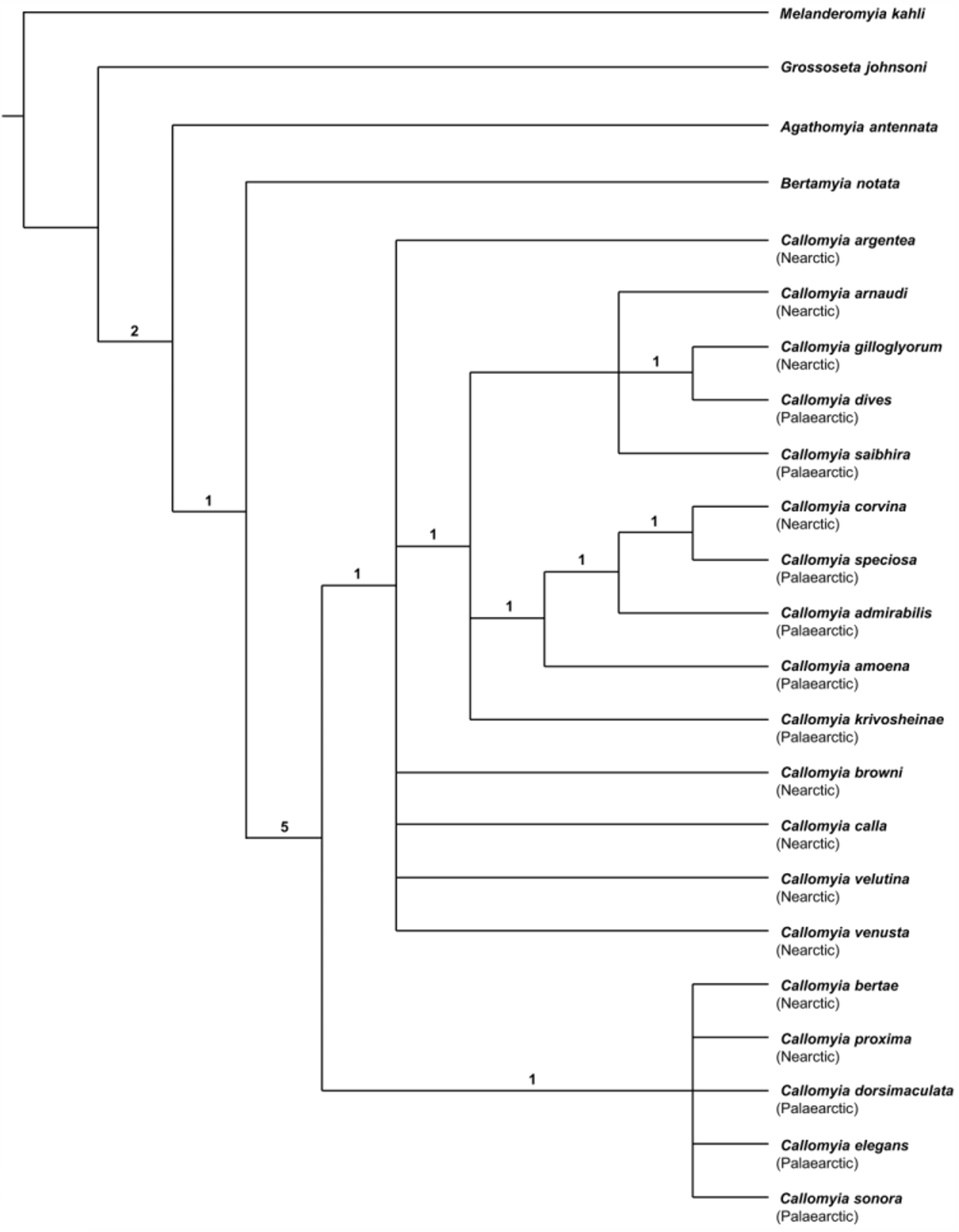
(Figs 3, 11, 21, 29, 37, 42, 44, 61, 74)
Callomyia gilloglyorum Kessel, 1961b: 4 View in CoL . Type locality: Alameda County, California, USA.
Diagnosis. This western Nearctic species is characterized by contrasting black and silver-grey abdominal colour, an elongate conical first flagellomere in the male and subtriangular first flagellomere in the female, 7–8 notopleural setae on the male scutum, male with yellowish-brown tinted wing, and male terminalia with a broadly lamellate surstylus and a trifid hypandrial process. The male is similar to the western Nearctic species C. corvina , but differs in colour, terminalia, shape of the first flagellomere, and setae on the notopleuron and mid tibia, as indicated in the key to species. The male of C. gilloglyorum also has very similar terminalia to the southwestern Nearctic species C. arnaudi , but differs by a longer ventral process on the broadly lamellate surstylus and longer projections on the hypandrial process.
Description. Male ( Figs 11 View FIGURES 9 − 14 , 21 View FIGURES 19 − 24 ). Body length 3.8–4.1 mm; wing length 3.9–4.1 mm. Head silver-grey; mouthparts including palpus brownish-yellow; antenna with scape, pedicel, first flagellomere and arista dark brown. Antenna with first flagellomere elongate-conical ( Fig. 42 View FIGURES 41 − 44 ).
Thorax mainly dark brown to black with indistinct silver-grey dusting on lateral portion of presutural scutum, notopleuron, supra-alar area of scutum and posterolateral portion of postsutural scutum; propleuron, mesopleuron, metapleuron, mediotergite and laterotergite silver-grey; postpronotal lobe yellowish-brown posteroventrally. Scutum with 7–8 notopleural setae.
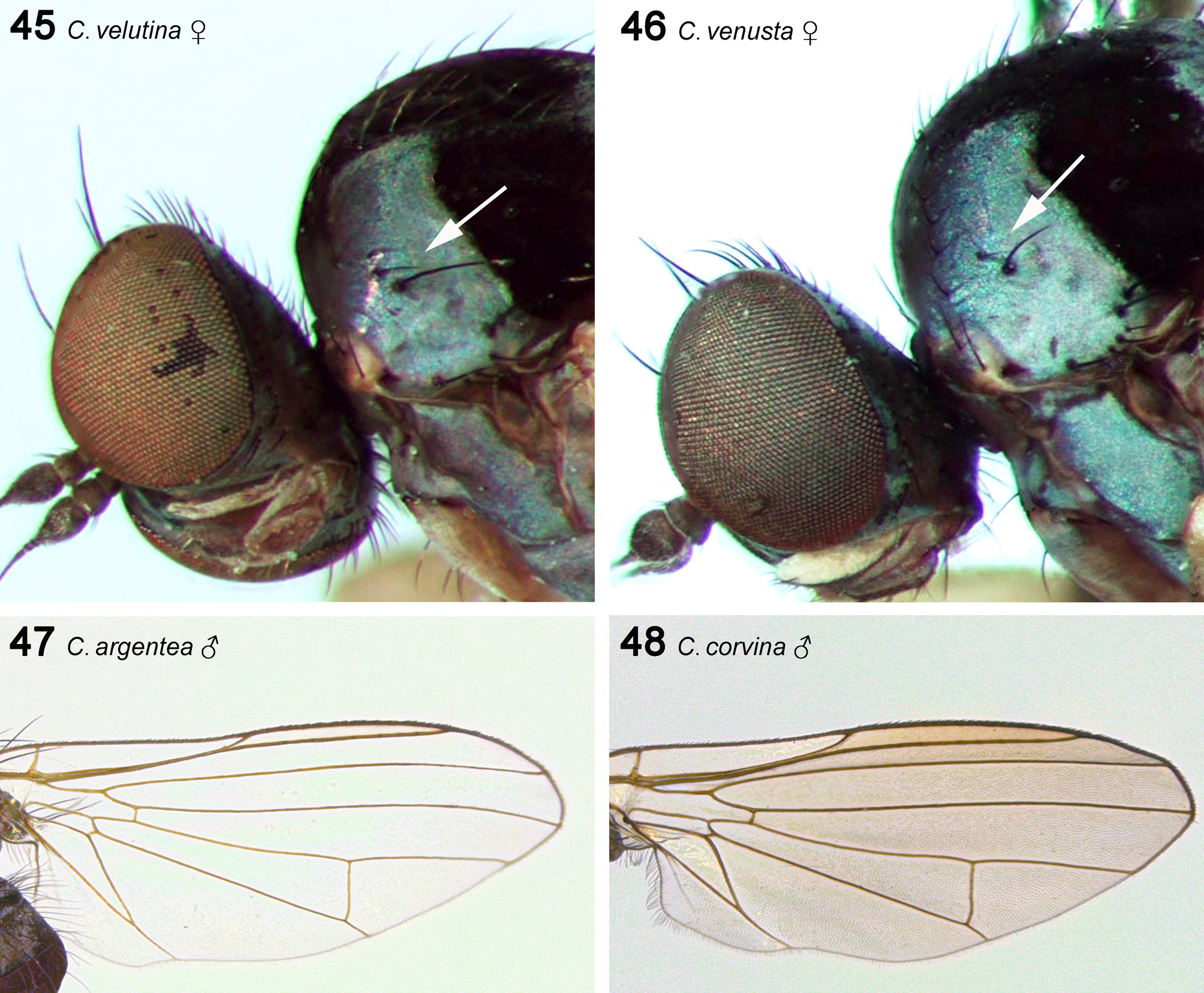
Legs yellowish-brown to brown with trochanter, base and apex of femur, and base of tibiae yellow; tarsomeres 3–5 dark brown; coxae silver-grey dusted. Mid tibia with median anterodorsal seta absent, median dorsal seta present (as in Fig 50 View FIGURES 49 − 54 ); base of hind femur with long thin posteroventral seta (as in Fig. 54 View FIGURES 49 − 54 ). Hind tarsomere 1 long, slightly narrower than apical width of hind tibia, length approximately 3X width.
Wing yellowish-brown tinted (as in Fig. 48 View FIGURES 45 − 48 ). Halter with stem brown, knob dark brown.
Abdomen dark brown to black with lateral to ventrolateral silver-grey markings on tergites 1, 2 and 4; ventrolateral silver-grey markings on tergites 3 and 5; posteroventral silver-grey dusting on tergite 6 in most specimens; tergite 7 entirely brown to grey; sternites light brown, sternite 8 brown to grey.
Terminalia ( Fig. 61 View FIGURES 59 − 62 ) with epandrium and hypandrium grey; surstylus and hypandrial process brown; cercus brownish-yellow. Epandrium with short broad ventral lobe, rounded at apex; apical process elongate, narrow, pointed at apex. Surstylus broadly lamellate with narrow ventral process; dorsal lobe lengthened with rounded apex; ventral process elongate, moderately curved dorsally. Hypandrium with elongate slender apical process; process trifid, with 2 very long narrow apical projections and long narrow basoventral lobe. Postgonite very long and narrow, rounded at apex. Phallus sharply hooked at apex. Cercus short.
Female ( Figs 29 View FIGURES 29 − 32 , 37 View FIGURES 37 − 40 ). Body length 2.9–3.5 mm; wing length 3.45–3.9 mm. Head silver-grey with vertex, occiput and face silver-brown to reddish-brown in some specimens; mouthparts brownish-yellow with palpus pale yellow; antenna with scape and pedicel yellowish-brown, first flagellomere and arista brown to dark brown. Antenna with first flagellomere subtriangular ( Fig. 44 View FIGURES 41 − 44 ).
Thorax mainly silver-grey; scutum brown dorsally; scutellum darker medially in some specimens. Scutum with 2–3 presutural intra-alar setae.
Legs brownish-yellow with tarsomeres 3–5 brown to dark brown; coxae silver-grey dusted in most specimens. Mid tibia with short median dorsal seta present in most specimens (as in Fig. 52 View FIGURES 49 − 54 ).
Wing hyaline. Halter brownish-yellow to brownish-orange.
Abdomen dark brown to black with tergites 1, 2, 4, 6 and 7 silver-grey, tergites 2, 4 and 6 interrupted by a median dorsal dark band in some specimens; tergites 3 and 5 with posterior silver-grey dusting in some specimens; sternites pale yellow, silver-grey dusted in some specimens.
Terminalia with segment 8 silver-grey; epiproct, hypoproct and cercus brownish-yellow, silver-grey in some specimens.
Type material. HOLOTYPE, ♂ labelled: “Oakland/ Alameda/ Co. Cal.; “J. Gillogly/ Collector; “Larvae/ V.1.54/ Emerged/ XI.9.54; “ HOLOTYPE / Callomyia / gillogly-/ orumKessel [red label]; “California Academy/ of Sciences/ Type No. 6446 ( CAS). ALLOTYPE, ♀ labelled: Oakland/ Alameda/ Co. Cal.; “J. Gillogly/ Collector; “Larvae/ V.1.54/ Emerged/ VIII.15.54; “ALLOTYPE/ Callomyia / gillogly-/ orumKessel [red label]; “Collection of the/ CALIFORNIA ACADEMY/ OF SCIENCES, San/ Francisco, California ( CAS). PARATYPES: USA: CALIFORNIA: same data as holotype (3 ♀, 2 ♂, CAS); same data as holotype except emerged 1.xi.1954 (1 ♂, CAS).
Additional material examined. CANADA: BRITISH COLUMBIA: King Salmon Lake, 15.vii.1960, W.W. Moss (1 ♀, CNC); Manning Provincial Park, small creek vic. Manning Park Lodge, 5.viii.1986, P.H. Arnaud Jr. (1 ♂, CAS); USA: ALASKA: Spenard, 17.viii.1959, E.L. Kessel (1 ♂, CAS); CALIFORNIA: Humboldt County, Prarie Creek, 4.xi.1960, E.L. Kessel (1 ♂, CAS); Marin County, Inverness, 14–29.xii.1963, P.H. Arnaud Jr. (1 ♀, CAS); Sacramento County (1 ♂, USNM); Stanislaus County, Del Puerto Canyon, Frank Raines Park, 1120’, 3.iv.1970, P.H. Arnaud Jr. (1 ♀, CAS); Tulare County, mi. S.E. Ash Mt., Seq. N. Park HQ, 10.iii.1984 (1 ♂, CSCA); MONTANA: Sanders County, Bull River Forest Camp, 31.viii.1964, E.L. Kessel (1 ♀, CAS); OREGON: Lane County, Paradise Forest Camp, Hwy. 126, 6.ix.1962, E.L. Kessel (1 ♀, CAS); Wallowa County, Arrow Forest Camp, Lostine River, 27.viii.1962, E.L. Kessel (1 ♂, CAS).
Geographical distribution and seasonal occurrence ( Fig. 74 View FIGURE 74 ). Callomyia gilloglyorum is a western North American species currently known from Alaska, British Columbia, Montana, Oregon and California. Larvae have been collected in early May, from which adults emerged in mid-August and early November. Adults have also been collected from early March to early April and July to late December.
Remarks. Callomyia gilloglyorum is most closely related to the widespread Palaearctic species C. dives , as alluded to by Chandler (2001) and shown in the strict consensus tree ( Fig. 79 View FIGURE 79 ). These two species have similar male terminalia and abdominal colour patterns. The male terminalia of C. gilloglyorum differ from the latter species by having a longer ventral process on the broadly lamellate surstylus, longer apical projections and a shorter basoventral lobe on the hypandrial process, and a narrower postgonite. The male terminalia of C. gilloglyorum are also similar to those of the Palaearctic species C. saibhira (comparison based on description and figures of C. saibhira in Chandler (2001) and Tkoč & Roháček (2014). Both the male and female abdominal colour patterns are similar to C. dives , however the black and silver-grey colour is duller, with C. dives having more striking silver and velvety black colour.
Callomyia gilloglyorum is the only North American species of Callomyia for which the immature stages are known (Fig. 3). Larvae were reared from white mycelia (fungus species not identified) under the bark of a fallen pine tree ( Pinus sp.) ( Kessel 1961b). The reared larvae took four to seven months to emerge as adults in the laboratory, possibly indicating a long developmental time in nature. However, seasonal records for adults collected from early March to early April and July to late December suggest shorter immature developmental times with multiple generations per year. Also, the early March and late December seasonal records, both from California, may indicate that adults of this species overwinter.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Callomyia gilloglyorum Kessel
| Cumming, Heather J. & Wheeler, Terry A. 2016 |
Callomyia gilloglyorum
| Kessel 1961: 4 |