Atelopus onorei, Coloma, Luis A., Lötters, Stefan, Duellman, William E. & Miranda-Leiva, Alfonso, 2007
|
publication ID |
https://doi.org/ 10.5281/zenodo.178262 |
|
DOI |
https://doi.org/10.5281/zenodo.5621401 |
|
persistent identifier |
https://treatment.plazi.org/id/5A5FD25B-FA2D-FF59-FF52-00B563A7BE06 |
|
treatment provided by |
Plazi |
|
scientific name |
Atelopus onorei |
| status |
sp. nov. |
Atelopus onorei View in CoL sp. nov.
Atelopus bomolochos View in CoL (non Peters): Lötters, 1996: Fig. 6 View FIGURE 6 ; Coloma and Ron, 2001: Fig. 9 View FIGURE 9 .
Holotype. QCAZ 1860, adult female, from Ecuador: Provincia Azuay: 10–15 Km E Luz María, Río Chipla (2° 47' 24" S, 79° 21' 36" W), approx. 2500 m above sea level, obtained on 9 April 1990, by Stella de la Torre- Salvador, John J. Wiens, Felipe Campos-Yánez, and Luis A. Coloma.
Paratypes. KU 217448–57, QCAZ 1861–63, 1865–67, 1868 (cleared and double stained preparation), 32258–59, same data as holotype; QCAZ 3440, 3442, same locality as holotype, obtained on 21 April 1990, by Fabián Toral, María Augusta Bravo, and Gustavo Morejón.
Diagnosis. (1) A moderate-sized species with mean SVL in adult males 38.8 mm (35.2–41.3 mm, n = 9) and in adult females 44.8 mm (41.9–47.9, n = 4); (2) hind limbs short, mean tibia length/SVL 0.381 (0.341– 0.409, n = 13); (3) phalangeal formula of hand 2-2-3-3, basal webbing absent; (4) foot webbing formula I (0– 1)—(0–2) II (1/2–2)—(1–3) III (1–3)—(2–4) IV (2–4)—(1–3) V; (5) snout acuminate, slightly protruding beyond lower jaw; (6) tympanic membrane absent; (7) dorsal surfaces of body usually smooth, (one [out of 13] individuals bears a few scattered small spiculae); (8) yellow spiculae (creamy white in preservative) present mostly on flanks; (9) vertebral neural processes inconspicuous; (10) dorsum yellow-orange to light green (creamy yellow to gray in preservative); (11) venter uniform cream to pale yellow in preservative; (12) gular region without warts, spiculae or coni.
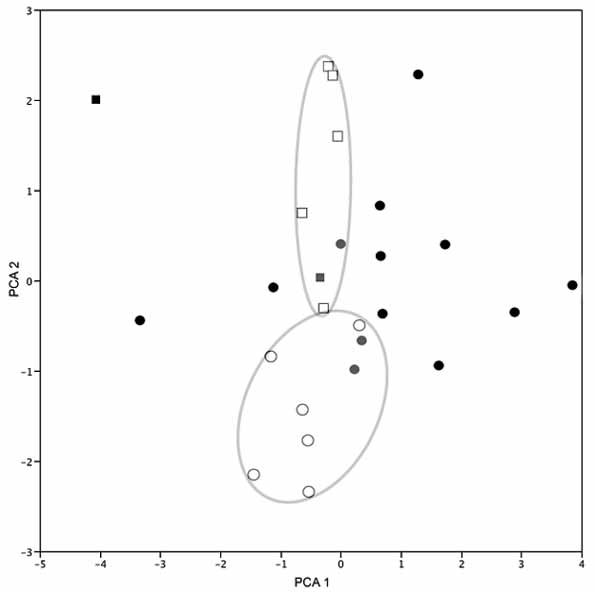
By having in life an aqua blue iris, Atelopus onorei is distinct from all other species of Atelopus . By displaying in life yellow-orange, green, or a combination of yellow-orange and green colors, A. onorei is most similar to A. bomolochos . Descriptions of the type specimens of A. bomolochos did not include colors in life. Nonetheless, Peters (1973) described the color in alcohol as “may be yellowish, yellow-green, or light brown.” Specimens assignable to A. bomolochos , collected near the type and paratype localities reveal large color variation as described by Peters. All of them have a predominantly black iris as depicted in Figs. 3 View FIGURE 3 B, C, D; although, specimens from 10 km S Cutchil show a fine green line around pupil (Field notes of John J. Wiens, 29 April 1990). Entirely yellow-orange individuals are most similar to A. bomolochos (from vicinities of Sigsig and Cutchil, Provincia Azuay; see Lötters 1996: Fig. 7 View FIGURE 7 ), some specimens of Atelopus sp. from Departamento Nariño, Colombia (formerly referred to A. ignescens ; for discussion see Coloma et. al., 2000), A. guanujo Coloma, 2002 from Provincia Bolívar, Ecuador, A. carbonerensis Rivero 1974 , A. chrysocorallus La Marca, 1994 , and A. sorianoi La Marca, 1983 all from the Andes of Venezuela (Estados Mérida and Trujillo). Atelopus onorei further differs from A. bomolochos by lacking minute black stippling regularly distributed on dorsum ( Fig. 7 View FIGURE 7 ), and by differences in hand and foot lengths of females. Herein, we compare A. onorei to populations described by Peters (1973) as A. bomolochos (specimens also used for morphometric analyses are indicated in the Appendix). The first two components with eigenvalues> 1.0 were extracted from the PCA of 7 morphometric variables of 15 males and 11 females from populations of A. bomolochos (Sevilla de Oro, Cuenca, and Juncal-General Morales) and A. onorei from Río Chipla. The axes accounted for 54.6 % of the total variation. There is no overlap among the morphometric space of females of A. onorei vs. A. bomolochos ( Fig. 8 View FIGURE 8 ), mostly because of hand and foot lengths, which are the highest loadings along PC II. Hand and foot lengths differ significantly between females of A. onorei vs. A. bomolochos (log comparisons of HAND of females t -test, t = 5.165044, df = 6.4, P> 0.0009; FOOT of females t -test, t = 4.578094, df = 8.9, P> 0.0007).
Atelopus onorei View in CoL (mean SVL of females 44.8; mean SVL of males 38.8) is significantly larger (SVL of females t -test, t = 2.992, df = 14, P <0.097; SVL of males t -test, t = 7.320, df = 36, P <0.0001) than A. guanujo View in CoL (mean SVL of females 39.6, 35.0–46.0, SD 3.0, n = 12; mean SVL of males 33.9, 29.9–37.1, SD 1.6, n = 29; after data in Coloma, 2002) and lacks white spiculae in life. Atelopus onorei View in CoL differs from Atelopus View in CoL sp. from Nariño by lacking a patch of black spiculae on females’ gular-chest region. Atelopus onorei View in CoL differs from A. carbonerensis View in CoL and A. sorianoi View in CoL in having a less protuberant snout and lacking a noticeably postocular crest. Furthermore, it differs from A. carbonerensis View in CoL and A. chrysocorallus View in CoL by having vocal slits in males and lacking a row of warts on the dorsolateral surfaces of the body in males (after data from La Marca, 1996; Rivero, 1974; Coloma, 1997).
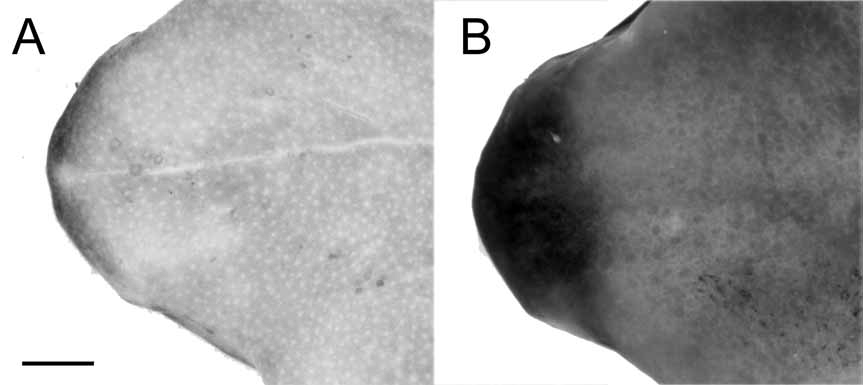
Description of holotype. ( Figs. 9 View FIGURE 9 A–C). Head about as long as wide, HLSQ and HDWD less than one third SVL (HLSQ/SVL = 0.280, HDWD/SVL = 0.282); snout acuminate, its margin slightly rounded in dorsal view; profile of tip of snout in lateral view curved and slightly protuberant to the anterior margin of jaw; no swollen gland on tip of snout; nostrils slightly protuberant, directed laterally, situated posterior to level of apex of lower jaw; canthus rostralis distinct, weakly concave from eye to nostril; loreal region concave; lips not flared; interorbital and occipital regions flat, smooth; eyelid flared without tubercles; postorbital crest slightly raised, glandular; low pretympanic and post tympanic areas warty; tympanic membrane and tympanic annulus absent; temporal area smooth; choanae small, rounded, widely separated (28% of HW); tongue about twice as long as wide, its posterior half not attached to mouth’s floor; ostia pharyngea absent.
Forearm relatively short (RDUL/SVL = 0.278); palmar tubercle round; palmar, supernumerary palmar, thenar and subarticular tubercles nearly indistinct; digital tips with round pads; thumb relatively long (THBL/ HAND = 0.717), apparently having two phalanges; webbing on hands absent, fingers lacking lateral fringes; relative length of fingers II<III<V<IV. Tibia relatively short (TIBL/SVL = 0.370); fold on distal half of inner edge of tarsus absent; inner metatarsal tubercle oval, barely distinct; outer metatarsal tubercle conical, low raised, about two thirds length of inner metatarsal tubercle; supernumerary plantar and subarticular tubercles barely conspicuous; digital pads distinct; webbing formula of foot I 1— 1 II 1— 2 III 2— 3 IV 3— 2 V; relative length of toes I<II<III=V<IV.
Dorsal surfaces smooth; flanks rugose with scattered spiculae; anterior and proximal upper surfaces of fore limbs lacking spiculae; throat rugose, contrasting to smooth chest, belly, and undersides of hind limbs; cloacal opening a tube at nearly midlevel of thighs, directed posteriorly; skin lateral to cloacal opening rugose.
In preservative (~70% ethanol), dorsum of head mostly cream with diffuse gray marks, loreal region gray; dorsum of body gray extending onto upper flanks; dorsal and ventral surfaces of limbs creamy yellow, except tibia, fingers III, V, toes IV and V, which have gray marks with diffuse borders; minute gray stippling absent on dorsum of body (viewed at 8 X magnification); spiculae white; throat, chest, belly, ventral surfaces of limbs, palms and plants yellowish cream; proximal end of tongue lacking black pigmentation, inner margin of mandible with a fine gray line.
Measurements (in mm). SVL 47.9, TIBL 17.7, FOOT 18.1, HLSQ 13.4, HDWD 13.5, ITNR 3.7, EYDM 4.5, EYNO 3.3, RDUL 13.3, HAND 10.6, THBL 7.6, SW 14.6.
Variation. Meristic variation is given in Table 1 View TABLE 1 . The paratypes resemble the holotype with the following exceptions. Sexual dimorphism is evident in that females are larger than males ( Table 1 View TABLE 1 ). Males have vocal slits and keratinized nuptial pads on the dorsal and inner surfaces of the thumb. Fore limbs are relatively long and slender in females only ( Table 1 View TABLE 1 ). Spiculae are most abundant on the flanks in a female (QCAZ 3440) and some are scattered on the dorsum of the body in that specimen.
Color variation in preservative (~70% ethanol): Dorsal colors vary from uniform yellow (QCAZ 3440) to mostly gray (QCAZ 3442). In QCAZ 2798, colors are faded because of poor preservation.
Color in life ( Figs. 3 View FIGURE 3 E–F). Data from color transparencies (no museum numbers associated) and the senior author’s field notes of specimens from the type locality. The dorsal ground color of body and limbs is orange yellow. The dorsum of one male is bright green, except the forearms, hands and feet, whereas other males and females show varying degrees of green coverage. In a few individuals the dorsum is pale yellowish brown with diffuse black blotches; the spiculae are yellow. The iris is conspicuously aqua blue with fine black reticulation and with a fine bluish-white stripe at the upper margin of pupil. Lötters (1996: Fig. 6 View FIGURE 6 ) and Coloma and Ron (2001) published the same color photograph of an amplectant pair of A. onorei (as A. bomolochos ). Nogales and Rueda-Almonacid (2005) also provided a painting and a color photo (under A. bomolochos ).
Distribution, ecology and current population status. Atelopus onorei is known only from the type locality in the Azuay Basin of the Cordillera Occidental of Ecuador, Provincia Azuay ( Fig. 6 View FIGURE 6 ) and a nearby creek (1–2 km West of Miguir, Morley Read pers. com. and his personal archive of photographs). At the latter locality, it occurs in sympatry with A. nanay Coloma 2002 . The type locality is at about 2500 m above sea level in humid cloud forest on the Pacific versant of the Cordillera Occidental. Annual mean rainfall is 1000– 2000 mm and the annual mean temperature is 12–18 ºC. Frogs were collected at Cloud Montane Forest areas (sensu Val en c ia et al. 1999), at the margin of the Río Chipla and near a small tributary. At the time of collection, during day at the type locality, most individuals were active and numerous pairs were in amplexus, while drizzle occurred (Field notes of L. A. Coloma, 9 April 1990). A female (QCAZ 3440) contained 133 ovarian eggs that are 2.14 mm in diameter (range = 1.67–2.58, n = 20, SD = 0.219).
Although this species previously was abundant at the type locality, no individuals have been found after 1990, despite at least four occasional search efforts by the senior author to locate this frog at the same site as before and surroundings on 28 November 1992, 20 February 1993, 21 April 1993, and 11 October 1993.
Etymology. The specific name is a noun in the genitive case and it is a patronym for Giovanni Onore, former Curator of Entomology at Pontificia Universidad Católica del Ecuador (PUCE). We recognize his invaluable contribution to the collections of Ecuadorian frogs and pioneer efforts on forming collections for scientific research at PUCE. His collecting efforts, especially during the eighties, resulted in the discovery of many new amphibians from Ecuador, among them Atelopus . As well, his amphibian collections in the Andean region of Ecuador provided key data, later used to document the amphibian extinctions and to shed light on its causes.
| QCAZ |
Museo de Zoologia, Pontificia Universidad Catolica del Ecuador |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.