Phellopsis LeConte
|
publication ID |
https://doi.org/ 10.5281/zenodo.180605 |
|
DOI |
https://doi.org/10.5281/zenodo.6235450 |
|
persistent identifier |
https://treatment.plazi.org/id/753687D8-FFBA-FFAB-1DC1-7DA9FA07FB07 |
|
treatment provided by |
Plazi |
|
scientific name |
Phellopsis LeConte |
| status |
|
Phellopsis LeConte View in CoL View at ENA
( Figs. 1–6 View FIGURE 1. A View FIGURE 2 View FIGURE 3 View FIGURE 4 View FIGURE 5 View FIGURE 6 )
Phellopsis LeConte, 1862: 216 View in CoL . (Type species Bolitophagus obcordatus Kirby 1837 , subsequent designation by Casey, 1907b: 470). Horn, 1870: 271, 273. Henshaw, 1881: 203, 255. LeConte and Horn, 1883: 365. Champion, 1884: 44. Fairmaire, 1894: C1. Lewis, 1887: 218 –220. Lewis, 1894: 379, pl. xiii, fig. 1. Champion, 1894: 114. Lewis, 1895: 447. Casey, 1907a: 44 –46. Casey, 1907b: 470, 480–481. Reitter, 1916: 130 –131. Leng, 1920: 223. Bradley, 1930: 183, 322. Böving and Craighead, 1931: 41, pl. 52. Gebien, 1936: 668. Crowson, 1955: 127. Arnett, 1962: 650, 668. Boddy, 1965: 77 –78, pl. x. Arnett, 1968: 650, 668. Arnett, 1971: 650, 668. Keleinikova and Mamaev, 1971: 125 – 128. Arnett, 1973: 650, 668. Doyen, 1976: 267, 270–271. Doyen and Lawrence, 1979: 341 –345. Papp, 1984: 162 – 163. Arnett, 1985: 350. Masumoto, 1990: 87 –91. Campbell, 1991: 252. Lawrence, 1991: 518–519. Egorov, 1992: 504 –505. Steiner, 1992: 25 –30. Lawrence, 1994: 341 –344. Ś lipiński and Lawrence, 1999: 21, 23. Steiner, 1999: 125, 138–139. García-París et al., 2001: 145. Ivie, 2002: 460.
Pseudonosoderma Heyden, 1885: 305 . (Type species Pseudonosoderma amurensis Heyden 1885 by monotypy). Semenow, 1893: 499. Synonymy by Champion 1894: 114.
Diagnosis: The members of this genus are easily distinguished from all other large (> 9 mm) Zopherinae by the presence of 11-segmented antennae and slightly open procoxal cavities.
Description (male): Length 11–22 mm. Body elongate, parallel sided; elytra 1.9–2.4X longer than pronotum; reddish brown to black; dorsal surface granulose, covered in small dark tubercles, each with single inserted setae; secondary vestiture setose; elytral and pronotal sculpture forming similar pattern in all species ( Figs. 2–4 View FIGURE 2 View FIGURE 3 View FIGURE 4 ).
Head with suprantennal ridges distinctly raised above widely separated antennal insertions ( Fig. 6 View FIGURE 6 ); not constricted behind eyes; dorsal surface of head with varying intensity of tubercles between suprantennal ridges and frons; margin of suprantennal ridge emarginate, flat, or convex. Antennae 11-segmented, not reaching beyond middle of prothorax; capitate with moderate 3 segmented club; antennomere 2 transverse, shorter than 1 or 3; antennomere 3 slightly elongate but less than twice as long as wide, shorter than 4 and 5 combined; antennomeres 9 and 10 with lateral patch of sensilla, 11 with apex covered in sensilla. Subgenal ridges present. Eye emarginate, coarsely faceted, frontal margin with golden setae; area directly behind with small elevated glabrous piece. Labrum visible, transverse, punctate, apical margin with dense fringe of setae. Mandible acutely bidentate, apex curved mesally; median tooth, setose-fringed membranous prostheca, and mola present. Maxillary surfaces with setae inserted in small punctures, variably sculptured; maxillary palpifer and basistipes with long bristle-like setae; apical maxillary palpomere rounded; galea and lacinia densely setose, lacinia hooked laterally, with one or two small teeth. Labial palps broadly separated, inserted laterally; ligula shallowly emarginate to smooth, setose. Submentum with setose pit ( Fig. 7 View FIGURE 7 ). Gula strongly narrowed or not; posterior tentorial pits present along suture, often indistinct.
Pronotum with lyriform ridge divided by midline; large apicolateral nodule; lateral margin slightly explanate or thickened; pronotum widest anterior to midline, anterior angles produced and broadly rounded; posterior angles obtuse; base narrower than elytral base; lateral margin of pronotum variably arcuate, with dense intertuberculate setae at margin, presence of setae varying in species on hypomeron. Hypomeron lacking any hint of antennal cavities. Prosternum in front of procoaxe longer than midlength between procoxal cavities; prosternum anterior to procoaxe at midline longer than prosternal process; prosternal process gradually expanded then narrowed, apical margin concave or biconcave; strongly elevated and curved dorsally behind coxae; procoxal cavities circular, widely separated, and narrowly open.
Scutellum abruptly elevated, notched anteriorly to rounded. Elytron with scutellary striole; with 7 rows of punctures distinct to obscure, rounded or irregular; epipleuron complete; distinct paired tubercles on elytron at start of apical declivity, a single tubercle near apex, apex rounded and slightly emarginate; elytra not fused. Mesepisternum widely separated, with round fovea or vermiculate, occasionally with small tubercles; mesocoxal cavities closed laterally, moderately separated; mesoventral process extending to middle of mesocoxal cavity.
Exposed portion of metepisternum long and narrow; metaventral median line long; metacoxal extending laterally to reach elytron; cavities moderately separated. Brachypterous, flight wings reduced to small elongate or rounded membranous pads.
Tarsal formula 5-5-4; tarsi and claws simple; tarsal setae on ventral surface of tarsomeres variably shorter and thicker than dorsal surface; all tibia with paired apical spurs; apical margin ringed with small spines; femora and tibia with length of pro<meso<meta; length of meta- tibia 0.06–0.12X longer than the femur; ventral surfaces of all femora with elongate glabrous area.
Intercoxal process of Ventrite 1 (V1) broadly truncate; abdomen with 5 ventrites; first 4 connate, V1 weakly to strongly depressed behind the coxae, V5 with preapical groove divided into two sinuous pits ( Fig. 4 View FIGURE 4 E–F); V3 and V4 with laterally expanded lobes, V2 occasionally with hint of expansion; laterotergite 3 variably expanded and coupled with internal surface of elytron. Aedeagus as in Fig. 8 View FIGURE 8 A–F.
FEMALE: The female lacks the setose pit on the submentum, but is similar in all other external morphological characters. The female genitalia distally terminate in a single-segmented, elongate gonostylus that is setose on the apex. Tergite eight and the proctiger are densely clothed in appressed setae. The pleated membrane lacks setation, and the coxite is setose laterally and at the apex.
LARVA: Not defined at the generic level (see description of the larva of P. obcordata below).
NOTES: The generic identity of this group has been relatively stable since LeConte’s description. The only issue was Heyden’s description of Pseudonosoderma , which contained P. amurensis and P. c h i n e n s i s for a short time. Pseudonosoderma is clearly synonymous with Phellopsis ( Champion 1894) .
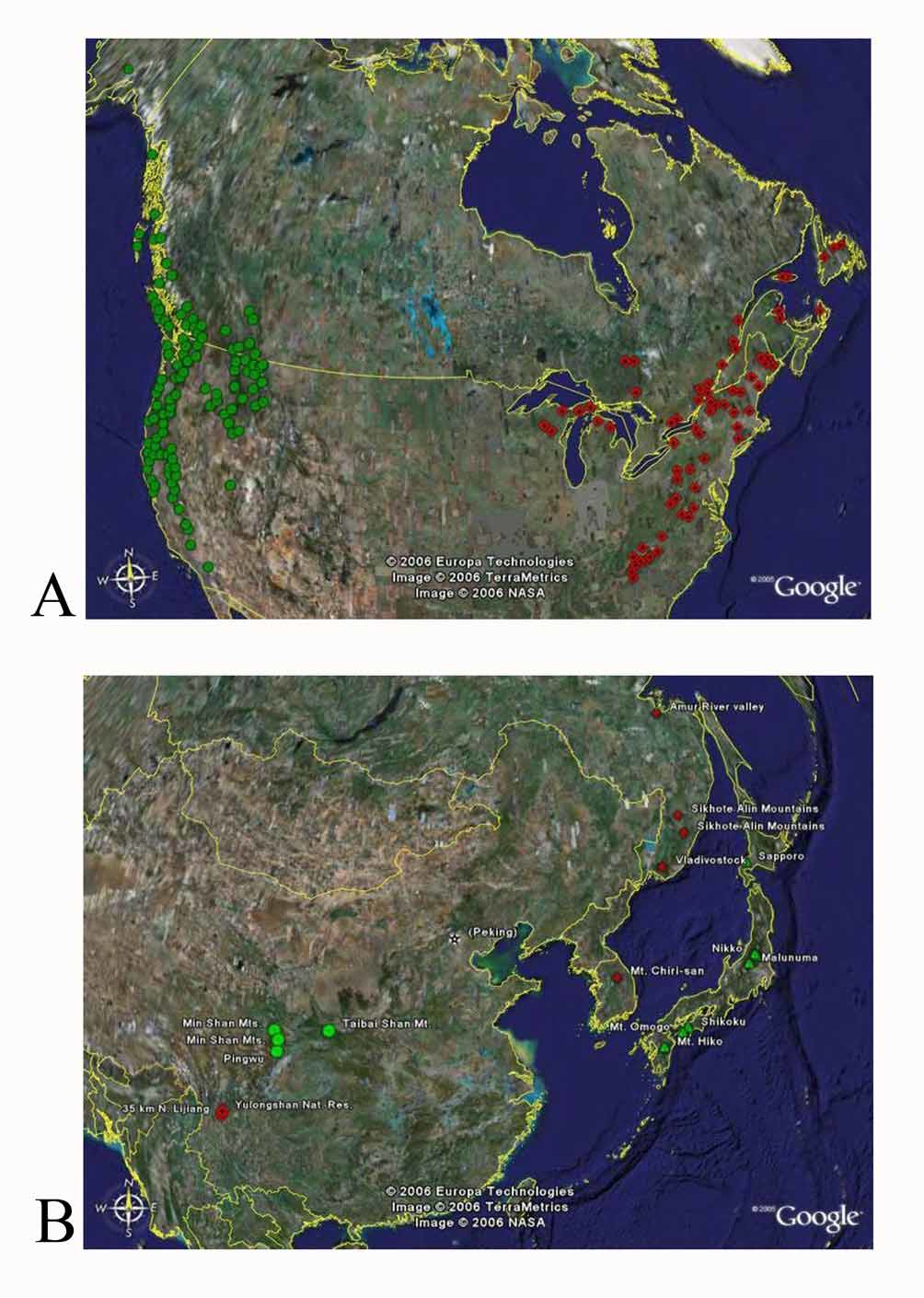
Biology. Adults and larvae both feed on fungi associated with decaying trees in old growth boreal forests ( Steiner 1992, Ivie 2002). Adults are surface feeders, while larvae burrow into the substrate.
Adults of the genus have been collected on a variety of xylophilous fungi growing on both coniferous and deciduous trees in various states of decay (label data, pers. obs., Steiner 1992, Ivie 2002). Several reports in the literature have associated adults with specific habitats. Phellopsis obcordata adults have been reported feeding on Piptoporus betulinus (Bull.:Fr.) P. Karst. on a paper birch log ( Betula papyrifera Marsh ), and Fomes annosus (Fr.) Cooke on dead balsam fir ( Abies balsamea Mill. ) in Maine, and from P. betulinus on sweet birch ( Betula lenta L.) in Maryland ( Steiner 1992). Phellopsis porcata adults have been associated with fungi on western hemlock ( Tsuga heterophylla (Raf.) Sarg. ) on Vancouver Island ( Guppy 1951) and on Lentinus fungus in Montana ( Russell 1968) . In Japan, P. s u b e re a was described from xylophilous bracket fungi of the genus Boletus on large oak trees ( Quercus sp.) ( Lewis 1894). Phellopsis amurensis was reported on Poriaceae fungi from a mixed forest of spruce, fir, birch, and other deciduous trees in the Sikhote-Alin Mountains of southern Primorskii Krai, Russia ( Lafer 2002).
Adults use thanatosis (death feigning) as a predator avoidance mechanism, a behavior that has been documented in several groups of beetles ( Chemsak and Linsley 1970, Allen 1990, Oliver 1996, Miyatake 2001, and Miyatake et al. 2004) and specifically in the Zopherinae ( Evans and Hogue 2004). When disturbed, adults drop to the ground with appendages retracted, and remain motionless for a significant time period (pers. obs., Steiner 1992). Their rough bodies blend perfectly with bark chips and detritus at the base of trees or litter on the ground, making a good search image critical to collecting species of this group.
The larvae of Phellopsis obcordata have been found living in shelf fungi in dense woodland ( Peterson 1951) and conks of the fungi Piptoporus betulinus ( Polyporales : Fomitopsidaceae ) ( Steiner 1999). In Western North America, the larvae of P. porcata bore through soft wood, where they feed on white sheet fungi between the laminae of large rotting spruce ( Picea sp.) stumps (Ivie 2002). Wood boring has also been reported for the larva of P. a m u re n s i s ( Keleinikova and Mamaev 1971), but this is probably another case of fungal association.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
Phellopsis LeConte
| Foley, Ian A. & Ivie, Michael A. 2008 |
Pseudonosoderma
| Champion 1894: 114 |
| Semenow 1893: 499 |
| Heyden 1885: 305 |
Phellopsis
| Garcia-Paris 2001: 145 |
| Steiner 1999: 125 |
| Lawrence 1994: 341 |
| Egorov 1992: 504 |
| Steiner 1992: 25 |
| Campbell 1991: 252 |
| Masumoto 1990: 87 |
| Arnett 1985: 350 |
| Papp 1984: 162 |
| Doyen 1979: 341 |
| Doyen 1976: 267 |
| Arnett 1973: 650 |
| Arnett 1971: 650 |
| Keleinikova 1971: 125 |
| Arnett 1968: 650 |
| Boddy 1965: 77 |
| Arnett 1962: 650 |
| Crowson 1955: 127 |
| Gebien 1936: 668 |
| Boving 1931: 41 |
| Bradley 1930: 183 |
| Leng 1920: 223 |
| Reitter 1916: 130 |
| Casey 1907: 470 |
| Casey 1907: 44 |
| Casey 1907: 470 |
| Lewis 1895: 447 |
| Lewis 1894: 379 |
| Champion 1894: 114 |
| Lewis 1887: 218 |
| LeConte 1883: 365 |
| Henshaw 1881: 203 |
| Horn 1870: 271 |
| LeConte 1862: 216 |