Stiphodon carisa, Watson, Ronald E., 2008
|
publication ID |
https://doi.org/10.5281/zenodo.180994 |
|
DOI |
https://doi.org/10.5281/zenodo.5668552 |
|
persistent identifier |
https://treatment.plazi.org/id/77108797-4721-7848-FF55-F93CF86121FE |
|
treatment provided by |
Plazi |
|
scientific name |
Stiphodon carisa |
| status |
sp. nov. |
Stiphodon carisa View in CoL n. sp.
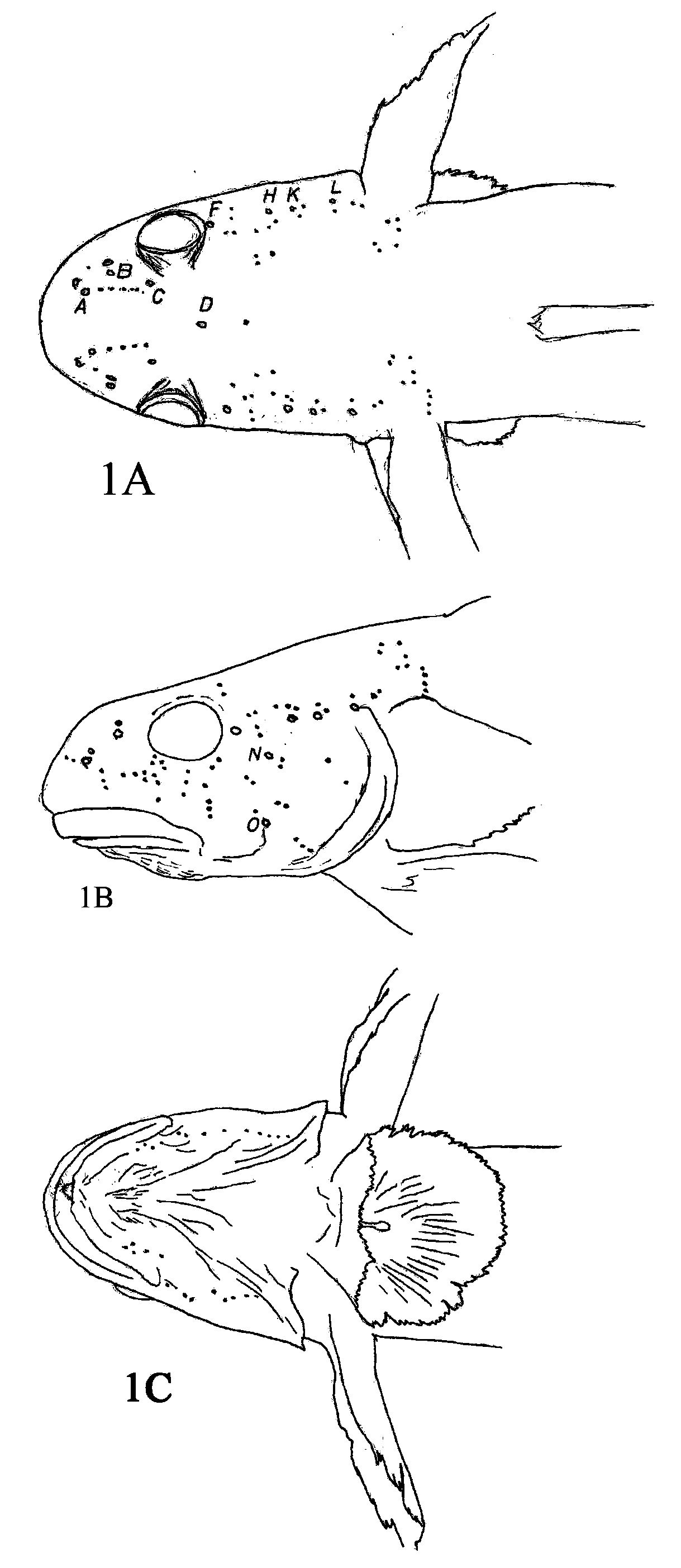
( Figs. 1–3 View FIGURE 1 View FIGURE 2 View FIGURE 3 A – D , Tables 1–3 View TABLE 1 )
Lampung Hill-stream Goby
Material examined. Seventy six specimens collected from Lampung Province, Sumatra, Indonesia, 20.9– 39.9 mm SL including 51 females and 25 males; largest male 36.7 mm SL, largest female 39.9 mm SL and smallest gravid female 23.4 mm SL.
Holotype. MZB 15194, female ( 33.8 mm SL). Indonesia: Sumatra: Lampung province: Way Ngarip ( 5° 27’ 59.22” S – 104° 31’ 3.72” E); 25 OCT 2005, L. M. Page, J. A. López, R. H. Robins, I. Rachmatika & R. Hadiaty.
Paratypes. CAS 224178, 2 males, 2 females (27.1–34.2), MNHN 2006–1604, male (35.0), MNHN 2006- 1605, male (26.7), MNHN 2006-1606, female (32.8), MNHN 2006-1607, female (30.8), MZB 15195, 12 males, 25 females (22.9–34.4), UF 162787, 7 males, 19 females (20.9–38.9), USNM 387893, 2 males, 2 females (32.4–28.6); same collection data as holotype.
Diagnosis. Second-dorsal fin with 9 second-dorsal fin rays; 15 pectoral-fin rays; 41–59 premaxillary teeth; predorsal scales sexually dimorphic in number, male with 5–11 and female with 8–16; 25–35 lateral scales; slightly embedded cycloid scales present on belly; male with a triangular-shaped first-dorsal fin with third and/or fourth spines longest but not filamentous and a patch of white fatty tissue posterior to pectoral-fin base; female usually with 5 (4–5) dusky to blackish blotches or spots along lateral midline from second-dorsal fin with usually 4 (3–4) posterior-most spots positioned close together on caudal peduncle, dusky band extending from anterior to eye to upper hypural base usually indistinct posterior to pectoral-fin base, with or without a dusky or black gular blotch, and in some females xanthism exists which fades in preservation and in life yellow with orange to bright red markings.
Description. Dorsal fins with VI-I, 9 (75), VI-I, 10 (1); first-dorsal fin separated from second. Anal fin with I, 9 (2), I, 10 (74); anal fin situated directly opposite second-dorsal fin. Pectoral-fin rays 14(2), 15 (68), 16 (6); pectoral fin oblong with posterior margin rounded, ventralmost ray usually simple (0–2). Caudal fin usually with 13 (12–14) branched rays, posterior margin rounded. Pelvic disc always with 1 spine and 5 strongly branched rays on each side; pelvic disc fused to belly between fifth rays only, frenum between spines fleshy, strong and well developed.
Scales in lateral series 27–35 ( Table 2), not extending to pectoral base, scales present dorsal to pectoral base, caudal peduncle with ctenoid scales to hypural base and cycloid beyond usually not with ctenoid and cycloid mixed, scales laterally ctenoid becoming cycloid towards vent and belly, along bases of dorsal and anal fins and close to pectoral base. Transverse forward series with all scales overlapping (10–18). Transverse back series with all scales overlapping (10–11). Zigzag series with 9 scales. Predorsal midline scales cycloid and may be evenly sized, with naked patches between scales or with tiny and larger scales mixed. Belly may be naked but usually with thin and slightly embedded cycloid scales extending forward under posterior margin of pelvic disc. No scales present on head, breast and pectoral base.
Premaxilla with tiny (ca. 8–10 teeth per mm), hinged tricuspid teeth bent at an angle with relationship to base of tooth numbering 41–59 ( Table 1 View TABLE 1 ). Dentary with recurved conical to caniform symphyseal teeth; horizontal teeth very fine and enclosed in a fleshy sheath, teeth on each side of symphysis usually correspond in number and position with premaxillary teeth. Upper lip smooth with a deep medial cleft. Rakers on inner edge of outer gill arch rudimentary, appear as short fleshy papillose projections (0–2+0–1+0). Cephalic sensory pore system always A, B, C, D, F, H, K, L, N and O, pore D singular, all others paired, oculoscapular canal separated into anterior and posterior canals between pores H and K ( Fig. 1 View FIGURE 1 ), anterior to pore D canals paired from pores A through C. Cutaneous sensory papillae well developed over surface of head and nape.
Sexual dimorphism well developed. Male with longer unpaired fins than female ( Table 3) and first-dorsal fin triangular in appearance, spines elongate, not appearing filamentous with spines third and/or fourth spine longest and membrane complete between all spines, caudal fin oblong; female with first-dorsal fin same height as second-dorsal fin and without elongate or filamentous spines, caudal fin round. Predorsal scales fewer in male (5–11) than female (8–16). Male with 2 or 3 (1–5) enlarged caniform symphyseal teeth; female usually with 1 (1–3) small conical recurved symphyseal tooth. In male a patch of white fatty tissue situated directly posterior to pectoral-fin base, but not obvious in immature material. Male urogenital papilla tapering, conical and bluntly rounded at tip ( Fig. 2 View FIGURE 2 a); female urogential papilla roughly an elongate, heart shaped organ, posterior margin fimbriate ( Fig. 2 View FIGURE 2 b).
Color in preservation. Sexual dichromatism well developed. Male: Background brown to gray. Head mostly black; gular region gray to black; branchiostegal rays generally black, membrane may appear grayish. Laterally on abdomen and onto caudal peduncle mostly gray with most scales edged in black; 3 or 4 oblique Scales in lateral series
26 27 28 29 30 31 32 33 34 35 36 37 38
carisa 3 8 11
atratus 1 2 1 7 11 16 20 12 4 atropurpureus 1 4 12 60 68 43 14 2 ornatus 1 - 6 4 1 semoni 5 4 21 28 34 33 22 24 17 8 2 1 1 * semoni 1 3 2 10 9 9 2 2
weberi 2 - 1 4 6 4 1
Scales in predorsal midline series.
2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17
ɗɗ carisa 1 5 5 6 2 3 3
ΨΨ carisa 1 - 6 10 12 7 6 3 2 ɗɗ atratus 1 - 1 2 5 3 5 13 - 1 ΨΨ atratus 2 2 3 6 10 6 1 2 1 ɗɗ atropurpureus 3 8 26 28 27 13 8 1 1 - - - 2 ΨΨ atropurpureus 2 2 7 9 12 19 10 5 5 6 1 1 ɗɗ ornatus 1 1 1 1 ΨΨ ornatus 1 3 2 2 ɗɗ semoni 3 7 30 36 31 13 9 1 1 - - 1
ΨΨ semoni 3 11 20 28 26 15 15 7 5 2 3
ɗɗ* semoni 5 6 2 - 2 2
ΨΨ* semoni 1 6 6 4 4 2
ɗɗ weberi 1 2 2 2 2 - 6 1 1
ΨΨ weberi 1
Scales in transverse forward series.
8 9 10 11 12 13 14 15 16 17 18 carisa 1 1 - - 3 9 34 20 5 1 atratus 1 - - - 1 4 8 25 11 5
atropurpureus 2 11 26 85 83 41 10 ornatus 2 3 2 2
semoni 2 1 8 5 12 47 58 43 1
* semoni 2 1 5 5 1 12 13 2
weberi 2 3 1 2 2 - 6 1 1 Scales in transverse back series.
bars between second-dorsal and anal fins and onto caudal peduncle; belly grayish. Dorsal fins blackish with whitish spotting on rays and spines, distal margin in second-dorsal fin clear. Anal-fin membrane blackish, basal half of spine and rays grayish distal margin clear. Caudal fin mostly gray to black with clear distal margin, grayish spotting present but not well developed. Pelvic disc mostly gray to black, distal margin white. Pectoral fins gray. Female: Each female similar but tending to be uniquely patterned and marked. Background mostly of body and head is cream to light tan. Blackish midlateral band originates at snout extending medially over cheek and opercle terminating at hypural base as a black spot, from posterior to pectoral fin base midlateral band usually appearing zigzag-like overlaid with dusky to blackish blotches and/or spots, midlaterally usually 6 (5–6) dusky to almost black blotches and/or spots and on caudal peduncle, usually posterior-most 4 (3–4) spots close together on caudal peduncle; posterior to pectoral-fin base and prior to origin of first-dorsal fin a faintly dusky oblique bar; posteriorly on upper pectoral-fin base a black spot, spot may be small or cover entire base as a broad bar; gular region and/or breast with a dusky blotch of varying size either present or absent; anterior to orbits a black band extending along dorsum to caudal-fin base at origin of dorsal procurrent rays terminating as a small black spot, band may appear indistinct. In dorsal fins spines and rays with black spots and streaks, membrane between spines and rays with black spots and streaks of varying shapes with largest and most conspicuous markings near posterior base of first-dorsal fin; caudal fin with crescent-shaped bars formed by black spots and dots especially prominent on and between medial rays; anal fin, distal margin clear, rays and spine whitish distally dusky, membrane between spine and rays dusky to blackish and may extend to anal-fin base; pelvic disc usually without markings; pectoral fin usually clear, blackish spots and streaks may be present on medial rays extending onto membrane close to base; in xanthic individuals coloration usually not as apparent. Xanthic female may fade leaving little pigmentation, but degrees of xanthism vary from well developed possessing few melanophores to those having a lesser degree of xanthism and a greater number of melanophores.
Color in life. Male: Olive green with brownish overtones; bright greenish gold band from tip of snout to edge of preopercle, opercle dorsally similarly colored; laterally scales with black margins, dorsal to midline most scales with an emerald green bar or spot; caudal fin brownish and marked as described in preservation. Female: Either with whitish and/or yellowish in background; markings on head, body and fins either grayish or blackish with some olive green on and near head, ventrally some scales reflect pearly white; xanthic females generally more yellow with all markings on head, body and fins orange to bright red, head appears metallic yellow on opercle and along branchiostegal rays; all xanthic individuals possess some melanophores but chromatophores dominate, eyes pigmented black.
Ecology. Stiphodon carisa was collected over gravel in a wide, shallow medium-sized river with gravel riffles and sandy runs ( Fig. 4 View FIGURE 4 ).
Etymology. The new species name is the Latin noun carisa , meaning an artful woman. The new name alludes to the unique patterns and color occurring in each female of the new species. The new name is treated here as a noun in apposition.
Affinities. The color and pattern in females of S. carisa is unusual in that this is the only species of Stiphodon so far described in which each female is uniquely patterned and/or colored. The occurrence of xanthism in a small percentage of females (ca. 5%) adds even more dimension and uniqueness in that each female not only has individualized markings but even to the point that red and yellow chromatophores are dominant in some individuals as opposed to melanophores. As noted in the color description, a female may have a dusky gular marking while another may not and in xanthic females the gular blotch may be reddish with some melanophores, as a bright red blotch or entirely absent.
In the male, presence of a triangular-shaped first-dorsal fin, and the especially dark to nearly black coloration in preservation, appear very similar to some S. atratus and S. weberi . However, S. carisa differs in having fatty tissue posterior to the pectoral base that is absent in S. atratus and S. weberi . The presence of fatty tissue posterior to the pectoral base in males of S. carisa is like that occurring in S. atropurpureus and S. semoni but S. carisa differs in having a triangular-shaped first dorsal fin with spine three and/or four elongate but not free or filamentous, ending as a point whereas S. semoni and S. atropurpureus have a rounded first dorsal and lack elongate spines. In S. atropurpureus both dorsal fins are dusky to blackish and may have black bars and spots especially prominent on spines and rays extending onto membrane between rays and spines, whereas in S. semoni both dorsal fins are colorless or nearly so. Stiphodon ornatus from Sumatra differs from S. carisa in the male having first-dorsal fin rounded without filamentous or elongate spines and without fatty tissue posterior to the pectoral base. This study does not consider S. carisa or S. weberi closely related despite similarities in overall morphometrics except for the length of the caudal fin. Stiphodon weberi is sufficiently differentiated from S. carisa in lacking fatty tissue in the male and in the range of premaxillary teeth that similarities in morphometrics can be dismissed.
Stiphodon carisa View in CoL allies with several species ( S. allen Watson, 1996 View in CoL , S. atropurpureus View in CoL , S. larson Watson, 1996 View in CoL and S. semoni View in CoL ) in having white fatty tissue posterior to pectoral-fin base in the male but differs from them in the male having a triangular-shaped first-dorsal fin and slightly longer unpaired fins. Most species of Stiphodon View in CoL with a tall first-dorsal fin and very elongate and filamentous spines tend to live in slower streams or areas not subjected to swift flows and tend not to ascend waterfalls. Most species of Stiphodon View in CoL with a rounded first-dorsal fin of the same height as the second-dorsal fin and without filamentous spines tend to live in swifter streams and often ascend waterfalls. Stiphodon carisa View in CoL is not at all similar to any species belonging to the subgroup that modally possesses 14 pectorals rays.
Preanal length.
31 32 33 34 35 36 37 38 39 40 42 42 carisa 1 - 2 12 10 26 14 6 2 - 1 1 atratus 1 3 4 10 26 12 6 - 1 1 atropurpureus 1 7 19 44 46 33 22 6 2 ornatus 1 3 2 2 1
semoni 2 8 21 39 38 39 27 11
* semoni 1 2 7 12 13 10 - 1
weberi 2 2 3 5 5 1
Head length.
19 20 21 22 23 24 25 26 27 carisa 4 23 21 19 4 2 1 atratus 2 18 19 14 6 2
atropurpureus 2 18 45 51 39 20 5 ornatus 2 2 2 2
semoni 7 24 67 42 23 17 4 * semoni 1 - 3 10 14 13
weberi 1 4 9 3 - 1
Caudal peduncle length.
18 19 20 21 22 23 24 25 26 27 28
carisa 1 4 12 13 19 18 6 2
atratus 2 17 18 15 14 2 atropurpureus 3 18 25 60 47 19 5 1 1 ornatus 1 1 1 1 3 2 1
semoni 2 25 29 45 51 20 12 2 - 1 * semoni 5 19 9 7 1
weberi 1 4 9 7 1
Body depth at origin of second-dorsal fin in males.
11 12 13 14 15 16 17 18
carisa 1 6 13 5 atratus 1 7 16 7 1 atropurpureus 2 6 35 37 8 1 ornatus 1
semoni 1 17 30 31 5 2 * semoni 4 7 4 2 weberi 7 4 5
Second-dorsal fin length.
23 24 25 26 27 28 29 30 31 32 33 34 35 36 ♂♂ carisa 1 - ΨΨ carisa 3 3 5 11 11 10 1 1 1 2 ♂♂ atratus 2 3 3 5 2 3 ΨΨ atratus 3 3 5 11 4 2
♂♂ atropurpureus 1 1 2 4 8 10 13 8 11 4 ΨΨ atropurpureus 1 2 1 5 7 18 18 15 14 5 4
♂♂ ornatus
ΨΨ ornatus 2 - - 2 2 1 ♂♂ semoni 1 2 5 7 4 10 9 12 ΨΨ semoni 1 3 6 8 19 15 18 8
♂♂* semoni 3 - 2 4 1 3 ΨΨ* semoni 2 2 7 6 1 2
♂♂ weberi 1 - 4 2 3 - Ψ weberi 1
Second-dorsal fin length (continued).
37 38 39 40 41 42 43 44 45 46 47 48 49 ɗɗ carisa - 3 1 1 2 3 6 2 2 2 - 1 1 ΨΨ carisa - 1
ɗɗ atratus 2 3 3 2 3 - - 2 1 ΨΨ atratus
ɗɗ atropurpureus 7 3 5 2 5 5 ΨΨ atropurpureus
ɗɗ ornatus 1 1 - - - - - - - 1 ΨΨ ornatus
ɗɗ semoni 9 16 13 8 2 2 3 ΨΨ semoni
ɗɗ* semoni 3 1 3
ΨΨ* semoni
ɗɗ weberi 1 2 - - 1 2 - 1 Ψ weberi
Anal fin length.
27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45
ɗɗ carisa 1 2 1 3 9 7 - 2 ΨΨ carisa 3 1 4 4 10 9 8 6 2 2 1
ɗɗ atratus 1 - 2 3 - 6 3 2 2 4 1 - 2 2 1 ΨΨ atratus 1 1 2 4 6 12 2 1
ɗɗ atropurpureus 1 2 - 14 7 10 9 13 9 6 8 5 5 - 1 ΨΨ atropurpureus 1 2 5 13 23 29 35 13 2 1
ɗɗ ornatus 1 1 - - 1 ΨΨ ornatus 2 3 1 1
Caudal fin length.
This study rejects any notion that S. carisa is a form or variation of S. semoni . Throughout the distribution of S. semoni ( Papua New Guinea to and throughout Indonesia) show almost no variation except for depth of body at the second-dorsal fin origin, which may be attributed to diet and/or water chemistry; the dorsal fins are clear and the first-dorsal fin is rounded. Similarly S. carisa is not S. atropurpureus (southern Japan, Taiwan, throughout the Philippines, and southern Malaysia) which has dusky to blackish dorsal fins and the first dorsal fin is rounded. Stiphodon carisa males show no variation, which contrasts sharply with females. Females in S. carisa vary considerably and coupled with xanthism make them unique whereas in S. atropurpureus and S. semoni markings in females tend to be fairly consistent. Stiphodon atropurpureus and S. semoni are very close phylogenetically, but the two are distinct and there is no overlap in their natural distribution. Stiphodon carisa is unique and, although a small individual of S. semoni was collected at the type locality, it appears to have been out of its element. Based on the type locality, S. carisa has a preference for slower and broader streams with riffles over gravel and sand, whereas S. atropurpureus and S. semoni have preferences for smaller and faster streams with cascades over rocks and boulders with a slab bottom substrate.
Remarks and observations. The patch of white fatty tissue posterior to the pectoral-fin base may serve at least two functions. The first appears to be as a status symbol in which larger and more dominant males have a larger white patch of fatty tissue whereas in subordinate males this is proportionally smaller and may be absent in the most immature males. White appears to be of significance within some genera belonging to the Sicydiinae; however, there are very few observations of reproductive and territorial habits and coloration within this group. White pectoral fins has been observed and reported in breeding males of species belonging to Sicydium and Sicyopterus ( Erdman 1961, 1986; Fitzsimons et al. 1993; Watson et al. 2000), and white dorsal and anal fins are found in Lentipes concolor ( Gill, 1860) ( Nishimoto & Fitzsimmons 1986) . In Stiphodon , the white fatty tissue is permanent, whereas in the few species observed of Lentipes , Sicydium and Sicyopterus , the white persists only during courtship and breeding otherwise colors are rather drab. Also the species appears to be combative and territorial, as are the vast majority of gobies. In one specimen, dentary teeth impressions suggest that the fatty tissue may serve as a cushion against vicious attacks by rival males; however, this appears minimized because dentary symphyseal teeth are strongly recurved. Another interesting observation in S. carisa is that the male is rather slimy whereas the female is not.
Observed on males of S. carisa , and present in many other small Sicydiinae is a well-developed column of cutaneous sensory papillae on the abdomen similar to a condition reported in Lentipes ( Watson & Kottelat 2006) . In Lentipes columns of sensory papillae are present in the male but absent in the female ( Watson & Kottelat 2006); the associated nervous system may also be sexually dimorphic. In S. carisa , the column of cutaneous sensory papillae is well developed in the largest males and, interestingly, the column of sensory papillae transects the posterior margin of fatty tissue.
TABLE 1. Number of premaxillary teeth to right of symphysis in selected species of Stiphodon (* S. semoni from Sumatra).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Stiphodon carisa
| Watson, Ronald E. 2008 |
S. allen
| Watson 1996 |
S. larson
| Watson 1996 |