Saevitella Bobies, 1956
|
publication ID |
https://doi.org/ 10.5281/zenodo.211022 |
|
DOI |
https://doi.org/10.5281/zenodo.5667278 |
|
persistent identifier |
https://treatment.plazi.org/id/8B497008-BE73-FFC2-AE8C-FE10FDD3FEA5 |
|
treatment provided by |
Plazi |
|
scientific name |
Saevitella Bobies, 1956 |
| status |
|
Genus Saevitella Bobies, 1956 View in CoL
Saevitella Bobies, 1956: 251 View in CoL ; Zágoršek 2010: 162. Type species: Saevitella inermis Bobies, 1956 View in CoL .
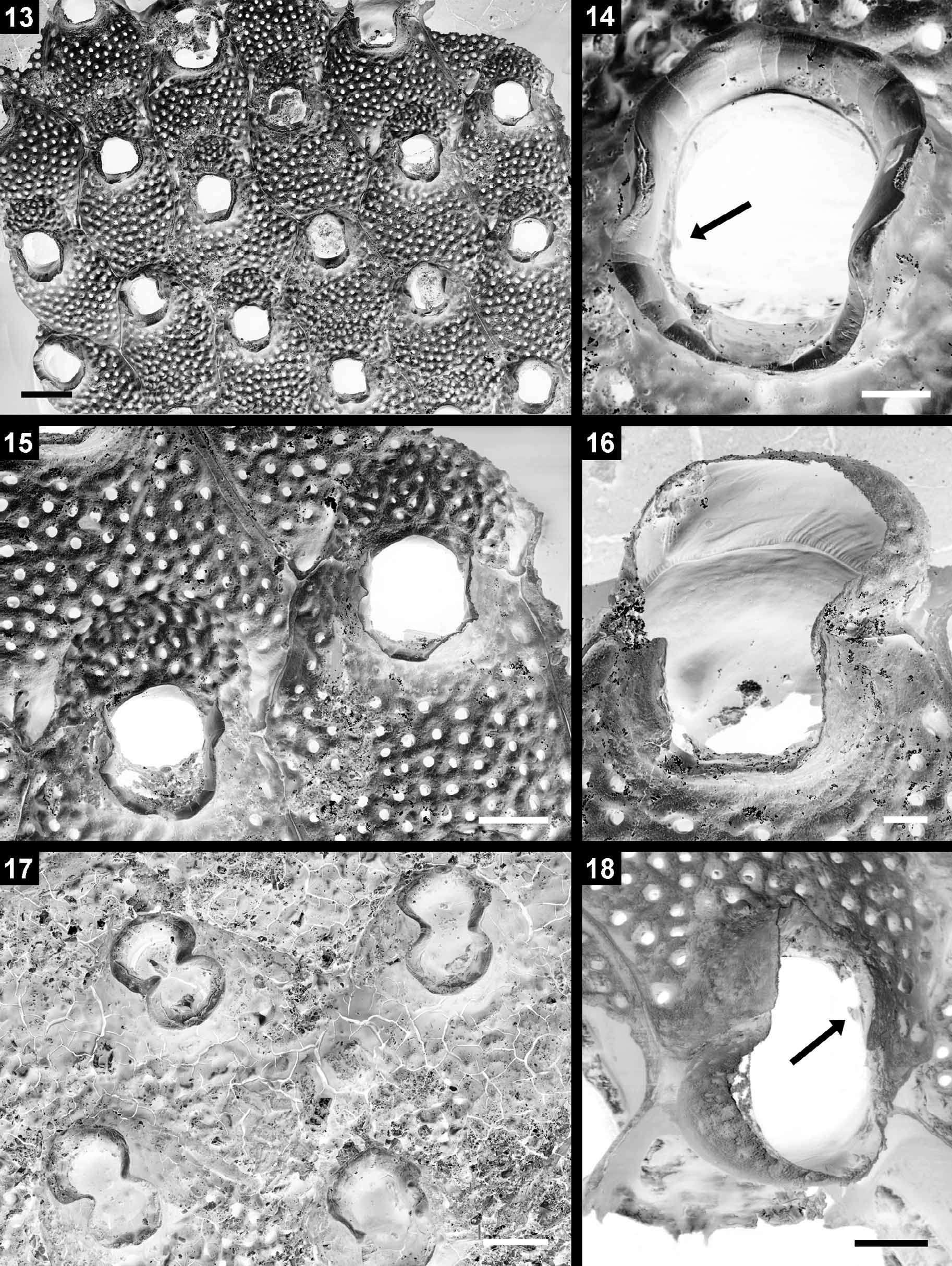
Diagnosis (amended). Colony encrusting. Zooidal frontal shield a pseudoporous cryptocyst. Basal pore-chambers absent. Primary orifice in autozooecia suborbicular with indistinct condyles and a prominent peristome, no oral spines; orifice in maternal zooids slightly dimorphic. Ovicell subimmersed in maternal zooid, hyperstomial ooecium produced by distal zooid and fused onto distal zooecial margin of maternal zooid; calcified endooecium pseudoporous and similar to frontal zooidal shield, ectooecium membranous and continuous with frontal ectocyst, ovicell opening closed by zooidal operculum. Avicularia absent. Ancestrula unknown.
Remarks. The genus Saevitella was introduced for the Miocene Paratethyan species Saevitella inermis Bobies, 1956 . Although S. inermis remained the only species in the genus until now, three little-known species from the Mediterranean Pliocene should also be placed in Saevitella – Lepralia gibbosula Manzoni, 1869 from Italy, Alysidotella cipollai Buge, 1956 from Tunisia, and an undescribed species from the Carboneras basin in southern Spain (pers. observ.). However, the relationships between these species are unclear at present. Owing to very similar morphologies, it could be that L. gibbosula is a senior synonym of A. cipollai and even of S. inermis (C. Pizzaferri, pers. comm. 2010).
The absence of Recent Saevitella from the Mediterranean Sea and its presence around Madeira may suggest that this archipelago acted as a refuge for Paratethyan/Mediterranean taxa during the Messinian salinity crisis and/ or the Pleistocene temperature minima (cf. Berning 2006).
Orificial outline and size in maternal zooids are not markedly distinct from non-maternal ones in both S. inermis and the Madeiran Saevitella peristomata ( Waters, 1899; see below), although apertures in ovicellate zooids may be slightly larger. However, the distal margin in a maternal zooecium does not form the distal rim of the primary orifice but is a distally bulging vertical wall. The ooecium, which is produced by the distal zooid, is a terminal continuation of this wall ( Figs 16, 18 View FIGURES 13 – 18 ). The operculum is, therefore, likely to rest on the proximal ooecial margin and ovicell closure is of the cleithral type (cf. Ostrovsky 2008). As there is also no calcified basal ooecial wall the brooding sac is likely to extend deeply into the distal part of the maternal zooecium, i.e. whereas the ooecium is hyperstomial, the ovicell as a whole is subimmersed.
The systematic placement of the genus remains doubtful. Whereas originally assigned to the Phylactellidae Canu & Bassler, 1917 (= Smittinidae Levinsen, 1909 ) by Bobies (1956), Gordon (pers. comm. 2011) rightly removed the genus from this family owing to differences in ovicell formation and tentatively placed it in the Hippopodinidae Levinsen, 1909 . However, there are also certain similarities, particularly concerning the structure of the zooid, orifice and ovicell, with the genus Cheilopora Levinsen, 1909 and another fossil species that has been recorded as Cheiloporina campanulata ( Cipolla, 1921) from the Mediterranean Sea and eastern Atlantic (see Berning 2006: 96, figs 121, 122). Both genera are currently placed in the Cheiloporinidae Bassler, 1936 , although it is unclear to which genus C. campanulata actually belongs as it differs from the type species of Cheiloporina , C. circumcincta ( Neviani, 1896) , in having a pseudoporous endooecium. Revision of the Hippopodinidae and Cheiloporinidae are certainly needed. Saevitella peristomata has also been placed in the genus Cosciniopsis Canu & Bassler, 1927 (e.g. Cook 1968: 182). However, although some superficial similarities exist with some of the species in that genus, e.g. with Cosciniopsis lonchaea ( Busk, 1884) , the type species of Cosciniopsis ( C. coelatus Canu & Bassler, 1927 ) differs in several aspects, including ovicell formation and the presence of avicularia.
Zágoršek's (2010: 161) decision to transfer C. campanulata to the cheiloporinid genus Hagiosynodos Bishop & Hayward, 1989 is not accepted here as there are distinct differences – in C. campanulata the endooecium is entirely pseudoporous, basal pore-chambers are absent, and budding is of the zooidal type (sensu Lidgard 1985), whereas in Hagiosynodos the endooecium is perforated by marginal pores only, basal pore-chambers are present, and budding is of the intrazooidal type. In fact, owing to the different budding types, it is arguable whether they should belong to the same family at all.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
Saevitella Bobies, 1956
| Berning, Björn 2012 |
Saevitella
| Zagorsek 2010: 162 |
| Bobies 1956: 251 |