Austrosimulium (Novaustrosimulium) bancrofti ( Taylor 1918 )
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4700.4.10 |
|
publication LSID |
lsid:zoobank.org:pub:F8C10BF0-53AD-41BC-9DBF-06FEB5086F5B |
|
persistent identifier |
https://treatment.plazi.org/id/8E1C2F5F-FF9C-FF9E-FF41-799C3F64F9BA |
|
treatment provided by |
Plazi |
|
scientific name |
Austrosimulium (Novaustrosimulium) bancrofti ( Taylor 1918 ) |
| status |
|
Austrosimulium (Novaustrosimulium) bancrofti ( Taylor 1918) View in CoL .
( Figs. 1–42 View FIGURES 1, 2 View FIGURES 3–5 View FIGURES 6–10 View FIGURES 11–13 View FIGURES 14–17 View FIGURES 18, 19 View FIGURES 20–23 View FIGURES 24–29 View FIGURES 30–31 View FIGURES 32–36 View FIGURES 37, 38 View FIGURES 39–41 View FIGURE 42 )
Simulium bancrofti Taylor 1918: 168 View in CoL (in part with Austrosimulium pestilens View in CoL ).
Austrosimulium bancrofti (Taylor) View in CoL . Tonnoir, 1925: 241 (in part with A. pestilens View in CoL ).
Simulium bancrofti Taylor. Taylor, 1927:70 View in CoL (redescription, reassignment).
Austrosimulium bancrofti (Taylor) View in CoL . Drummond, 1931: 8 (male, immatures).
Austrosimulium bancrofti (Taylor) View in CoL . Smart, 1945: 499 (taxonomy).
Austrosimulium bancrofti (Taylor) View in CoL . Mackerras & Makerras, 1948: 256 (systematics, biology).
Austrosimulium View in CoL (Novaustrosimuliu m) bancrofti (Taylor) View in CoL . Dumbleton, 1973: 484 (subgenus)
Austrosimulium View in CoL (Novaustrosimuliu m) bancrofti (Taylor) View in CoL . Colbo, 1974 (biology).
Austrosimulium View in CoL (Novaustrosimuliu m) bancrofti (Taylor) View in CoL . Bugledich, 1999: 231 (listing of citations).
Austrosimulium View in CoL (Novaustrosimuliu m) bancrofti (Taylor) View in CoL . Ballard and Bedo, 1991: 338 (species complex).
Austrosimulium View in CoL (Novaustrosimuliu m) bancrofti (Taylor) View in CoL . Ballard, 1994: 131 (RNA, adult antennae).
Austrosimulium View in CoL (Novaustrosimuliu m) bancrofti (Taylor) View in CoL . Adler, 2019: 23.
Diagnosis. A small to medium grayish species, adults normally with seven antennal flagellomeres. Female: abdomen with wide, discontinuous, ashy dorsal stripe, distinguishing the species from all others, albeit not A. (N.) pestilens . Male: antenna black, traces of yellow on scape and pedicel; hind basitarsus lacking ventral row of stout setae; abdomen with conspicuous patch of ashy pollinosity laterally on V, VI segments. Pupa: gill horn flat, spatulate, spinous, numerous fine filaments concertinaed basally; sternite IX with simple grapnel hooks. Cocoon: shoe-shaped with well-developed anterior collar. Larva: head pigmentation usually distinct; lacking ventral papillae; semicircular sclerite absent. (Note, Dumbleton 1973, in his diagnosis of the subgenus, states that antennomeres of larva are subequal—however, for bancrofti and pestilens , the basal one is shorter, for magnum the apical one is longer).
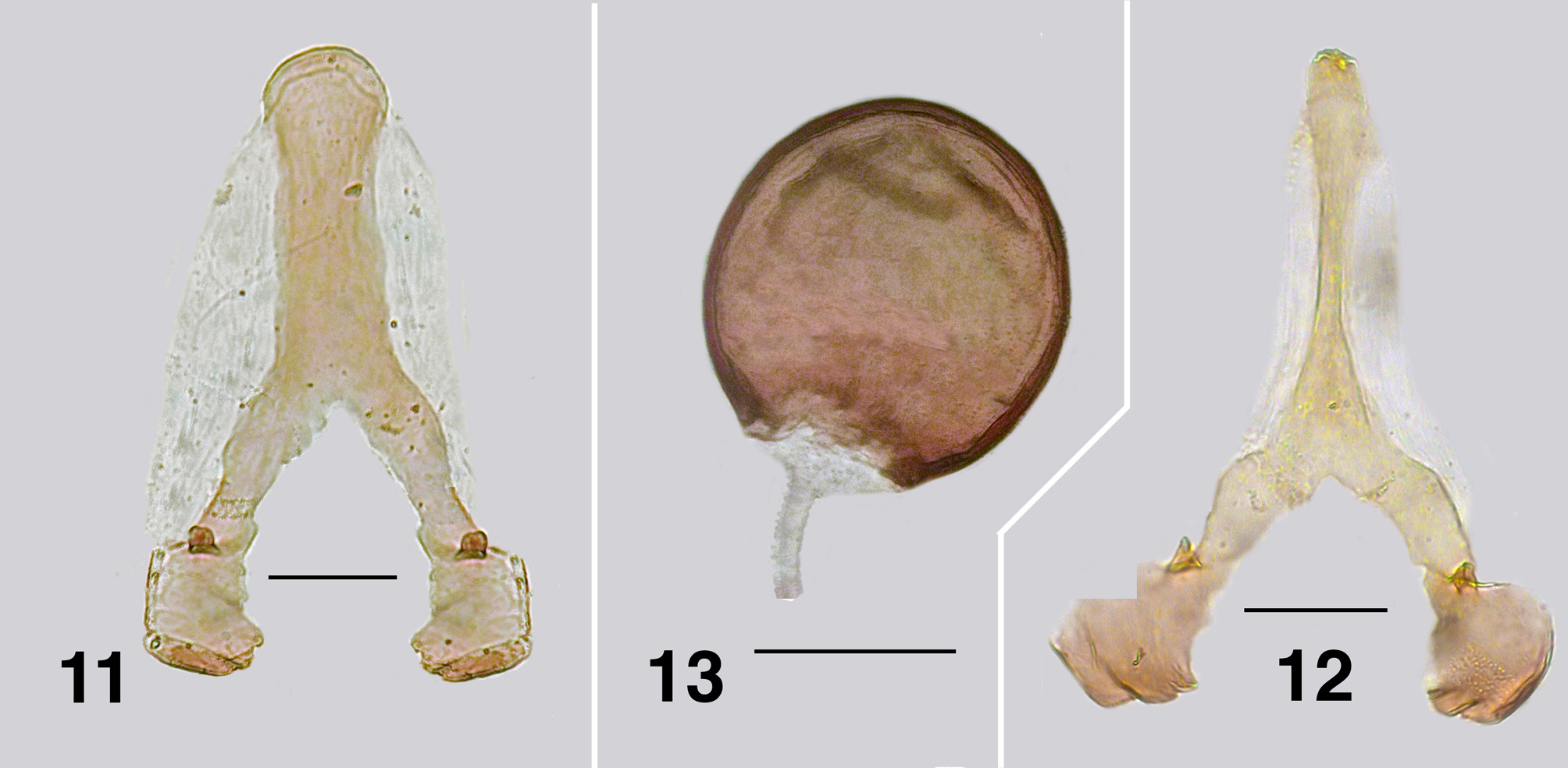
Adult female (based on types and extensive material in ANIC). Body ( Fig. 1 View FIGURES 1, 2 ): dark brown, with white patches on abdomen; total length 2.0– 2.5 mm. Head ( Fig. 2 View FIGURES 1, 2 ): overall dark brown, mouthparts paler, width 0.56–0.78 mm, depth 0.48–0.50 mm; postocciput not markedly hirsute, frons markedly broad, frons/ head ratio 1.0:3.4, under some lighting appears pollinose, cervical sclerites distinct. Eyes: interocular distance 0.2 mm; ommatidia diameter 0.027 mm; ca. 28 rows across and 40 down at mid-eye. Clypeus: width ca. 022mm; densely covered with fine gray hairs. Antenna ( Fig. 3 View FIGURES 3–5 a–d): variable; not markedly extended beyond head margin; total length 0.3–04 mm; normally with seven flagellomeres (but numbers various); scape and pedicel brownish-yellow, remainder dark brown, flagellomere I ca. twice as long as cup-shaped scape and markedly expanded apically (e.g., Fig. 3a View FIGURES 3–5 ), occasionally smaller ( Fig. 3f View FIGURES 3–5 ) even on same specimen; apical (VII) flagellomere commonly with groove at mid length (incomplete or complete division giving eight flagellomeres), markedly cone -shaped; (e.g. Fig. 3c, d, f View FIGURES 3–5 ), some specimens differ between sides. Mouthparts: well expressed; ca. 0.5× length of head depth; maxillary palpus ( Fig. 4 View FIGURES 3–5 ), total length 0.45 mm, palpomere III darker brown than remainder, proportional lengths of palpomeres III–V 1.0:0.7:1.3; sensory vesicle ovoid, large, 0.6× width palpomere III, opening 0.3× vesicle width; mandible ( Fig. 5 View FIGURES 3–5 ) substantial basally, finely tapered apically with ca. 30–45 inner markedly fine teeth, slightly increasing in size towards apex, outer teeth absent; lacinia with 16 inner teeth and 11 outer teeth; cibarial cornuae (for WA, thin basally, flared apically,) elongated, thin, strongly pigmented, central depression flat ( Fig. 6 View FIGURES 6–10 ). Thorax: length 1.1–1.2 mm; width 0.5 mm; scutum dark brown with dark pollinosity and moderately dense short hairs, showing dull golden dorsally, but silvery more laterally; scutellar depression with longer creamy hairs; scutellum black with similar hairs, but darker along apex; postnotum concolourous with scutellum, vestiture absent; pleuron and plural membrane with ashy pollinose. Wing ( Fig. 8 View FIGURES 6–10 ): clear, length 2.0– 2.5 mm; width 1.0 mm; veins yellowish; anterior veins well expressed, except subcosta, others poorly so; CuA markedly sinuous; basal cell absent; a:b ratio 1.0: 3.4; slightly dusky on anal lobe. Haltere: knob creamy-yellow to dense white, stem grayish. Legs ( Fig. 7 View FIGURES 6–10 ): evenly medium brown; hind basitarsus ca. 8× longer than width, calcipala small, 0.5× width of basitarsus, not extended as far as pedisulcus; hind basitarsus mark- edly hirsute on proximal half, ventral row of stout spines poorly expressed, even less so on distal region; basitarsus II 2.5× as long as apical width; claws ( Fig. 9 View FIGURES 6–10 ) lacking basal tooth, with small heel. Abdomen ( Fig. 10 View FIGURES 6–10 ): abdominal scale dark brown with pale hairs; tergite II rich brown with small central patch of white scales, 4×, as wide as long, tergites III–V quadratic with pale patch increasing in size posteriorly, tergites VI, VII wider than long, pale posteriorly. Genitalia: (for WA, Fig. 14 View FIGURES 14–17 ) sternite VIII heavily pigmented, hypogynial valves broadly cone-shaped, lightly pigmented, basally with dense microtrichia, apically bare and clear, medial gap straight, narrow; (for NSW, Fig. 15 View FIGURES 14–17 ) sternite VIII light brown, hypogynial valves broadly cone-shaped, lightly pigmented, particularly at valve apices with a clear band of unpigmented cuticle (also seen in A. pestilens ); genital fork, (ACT, Fig. 12 View FIGURES 11–13 ) anterior arm well defined, slightly expanded apically, lateral membranous areas very lightly expressed, posterolateral arms with small knee bend; apodeme small, with sharp apex; (WA, Fig. 11 View FIGURES 11–13 ) anterior arm short, broad, lateral membranous areas moderately expressed, apodeme small and rounded apically; spermatheca ( Fig. 13 View FIGURES 11–13 ) spherical, smooth, small scattered acanthae internally, junction with duct large with sculpted edge; cercus (VIC, Fig. 16 View FIGURES 14–17 ) ovoid, anal lobe poorly expressed, (WA, Fig. 17 View FIGURES 14–17 ) cercus broadly rounded, slightly ovoid, evenly spaced hairs, dense microtrichia, anal lobe larger, albeit not protrusive.
Adult male (based on ‘allotype’, WA; plus NSW material). Body ( Fig. 18 View FIGURES 18, 19 ): head and thorax dark, abdomen lighter; total length ca. 1.6 mm. Head ( Fig. 19 View FIGURES 18, 19 ): width 0.9 mm, depth 0.62 mm. Eyes: upper ommatidia yellowish orange, diameter 0.035 mm, ca. 23 across 26 down; lower ommatidia blackish red, diameter 0.011 mm, ca. 26 across and 30 down. Clypeus: pollinose. Antenna ( Fig. 20 View FIGURES 20–23 ): total length 0.5 mm; evenly dark brown, pedicel spherical and broader than remainder of antenna; flagellomere I elongated, 2× as long as wide, II rounded, III–IV angulate, distal (VII) markedly cone-shaped, often not completely separated from flagellomere VI. Mouthparts: poorly developed; length 0.3× head depth; mandibles and lacinia with terminal hairs; maxillary palpus ( Fig. 21, 22 View FIGURES 20–23 ) length ca. 0.5 mm, palpomere III dark, narrowed, sensory vesicle irregular in shape, 0.3× palpomere width, opening 0.2× vesicle width, palpomere IV moderately pigmented, expanded apically, palpomere V pale, proportional lengths of palpomeres III–V 1.0:0.7:1.3; there are minor differences between palpi from ACT ( Fig. 21 View FIGURES 20–23 ) and NSW ( Fig. 22 View FIGURES 20–23 )—shape of palpomere III, and length and shape of palpomere V. Thorax: length 0.9 mm; width 0.8 mm; scutum evenly velvety brown, vestiture of sparse silvery scales; scutellum and postnotum concolourous with scutum. Wing: length 2.0 mm, width 1.1 mm; anterior veins yellowish. Haltere: stem gray, knob white. Legs: evenly dark brown; hind basitarsus, 5.5× longer than width, markedly hirsute on proximal half, ventral row of stout spines present, but spaced irregularly. Abdomen: dark brown, pollinosity absent. Genitalia: ( Fig. 23 View FIGURES 20–23 ). gonocoxa as long as basal width, poorly sclerotized anteromedially; gonostylus ca. 4× as long as basal width, angulate in lateral view, slightly curved, 3–5 terminal spines, various even on same specimen; ventral plate markedly cone-shaped laterally, 1.7× as wide as long, very broadly V-shaped posteriorly, anterior arms not markedly developed, medial keel mark- edly broad, directed ventrally, with broad apex anteriorly, vestiture of dense fine hairs; median sclerite not markedly expressed, bifurcated apically; paramere as distinct curved rod with fine apical spinules; adeagal membrane with sparse microtrichia.
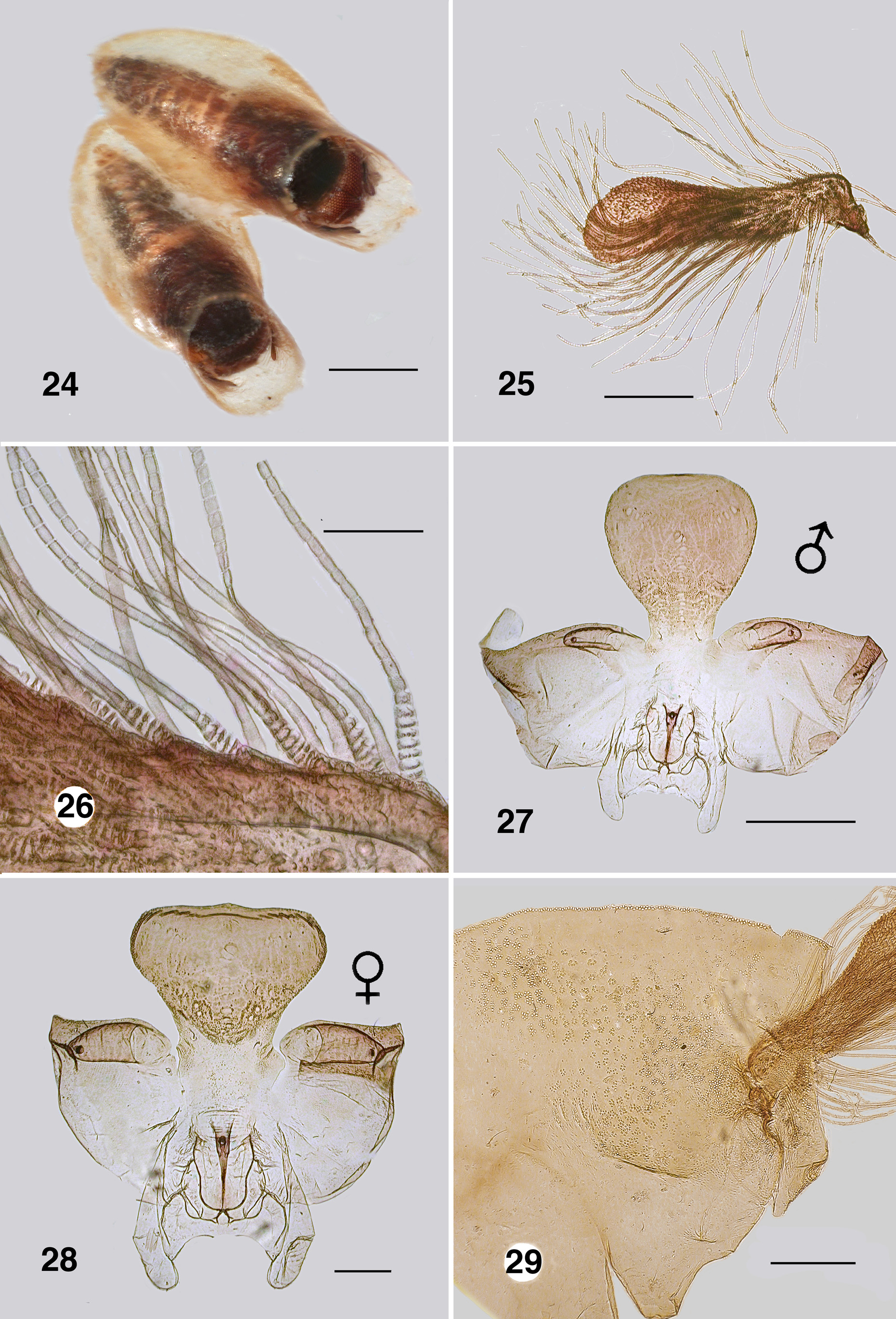
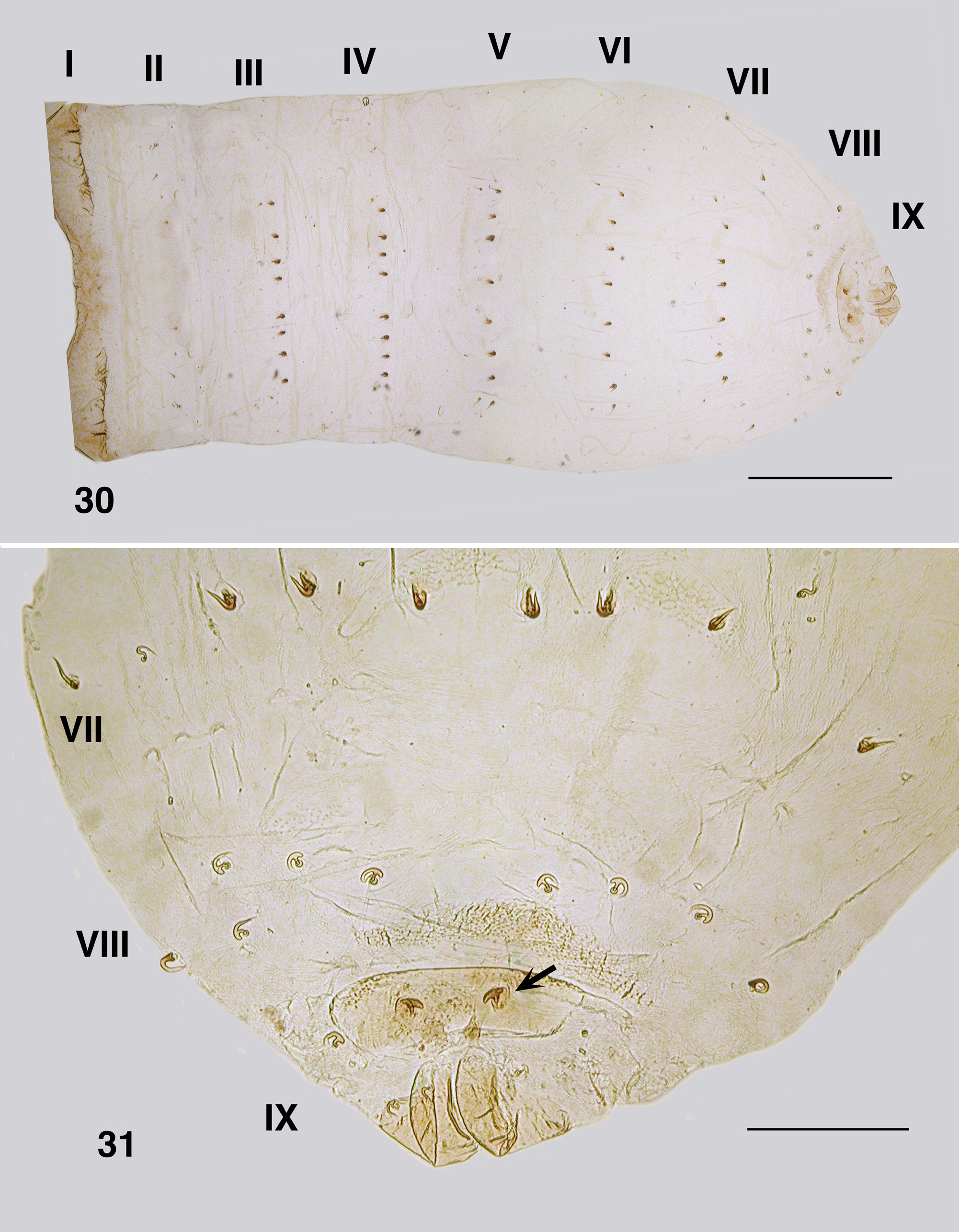
Pupa (numerous specimens, NSW, VIC) ( Fig. 24 View FIGURES 24–29 ). Body: female, length 2.3–3.0 mm, male 2.5–2.9 mm. Head: cephalic apotome lightly tuberculated with distinct tubercles arrayed along ecdysial sutures; male frons ( Fig. 27 View FIGURES 24–29 ) rounded apically, ratio of basal width to vertex width 1.0:2.2, basal width to length 1.0:2.6; frontal and facial setae present, but small; epicranial setae small, not obscured by antennal sheath; cuticle essentially smooth apically, tuberculate basally; female frons ( Fig. 28 View FIGURES 24–29 ) short broad with flat apex, ratio of basal width to vertex width 1.0:2.0, ratio of basal width to length 1.0:1.5, cuticle and setae as for female. Thorax ( Fig. 29 View FIGURES 24–29 ): cuticle with tubercles in rosettes, concentrated along medial ecdysial suture, more densely around gill base, no pattern; dorsocentral sensilla as small fine hairs. Gill ( Fig. 25 View FIGURES 24–29 ): broad flat spatulate horn, 0.65 mm in length, with short sharp spinules distally; ca. 60 fine filaments, 0.005 mm in diameter, subequal to horn length, absent from distal quarter of horn; filament surface annulated, annulations 2×longer than wide, slightly bead-shaped, concertinaed (ringed) at attachment to horn ( Fig. 26 View FIGURES 24–29 ). Abdomen ( Figs. 30, 31 View FIGURES 30–31 ): cuticle thin, colourless; armature markedly reduced; sternite armature absent; tergite I slightly sclerotized, tergite II with fine hairs, III–VII with anteriorly directed hooks, spine combs absent, but with minute scales, tergite VIII with grapnel hooks and posterocentral region of scales, tergite IX terminal spines small, directed posteriorly, sternite IX with sparse grapnel hooks.
Cocoon ( Fig. 24 View FIGURES 24–29 ): shoe-shaped, extended beyond pupa; fabric closely woven with distinct edge to anterodorsal opening, ventral collar well extended, with irregular edge.
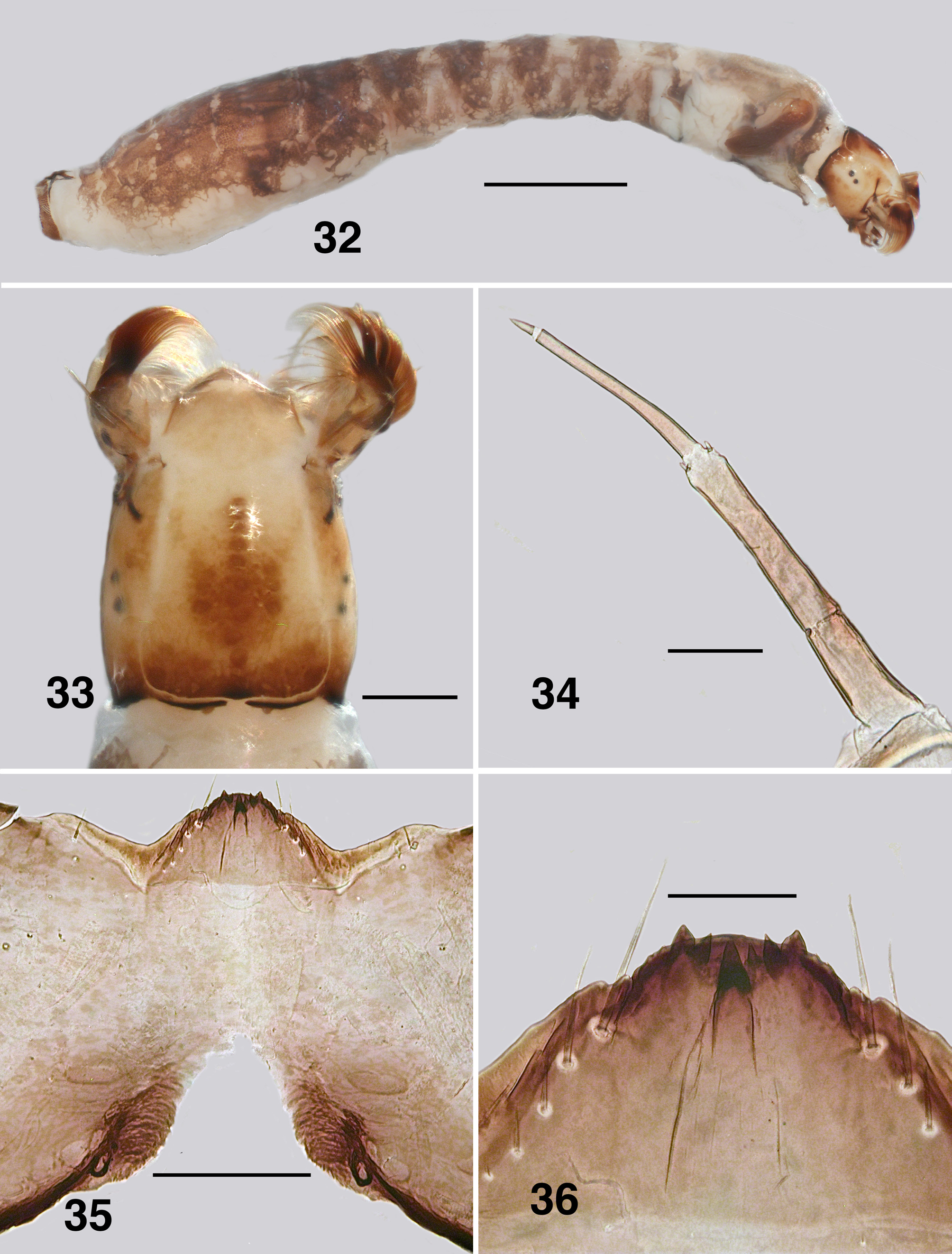
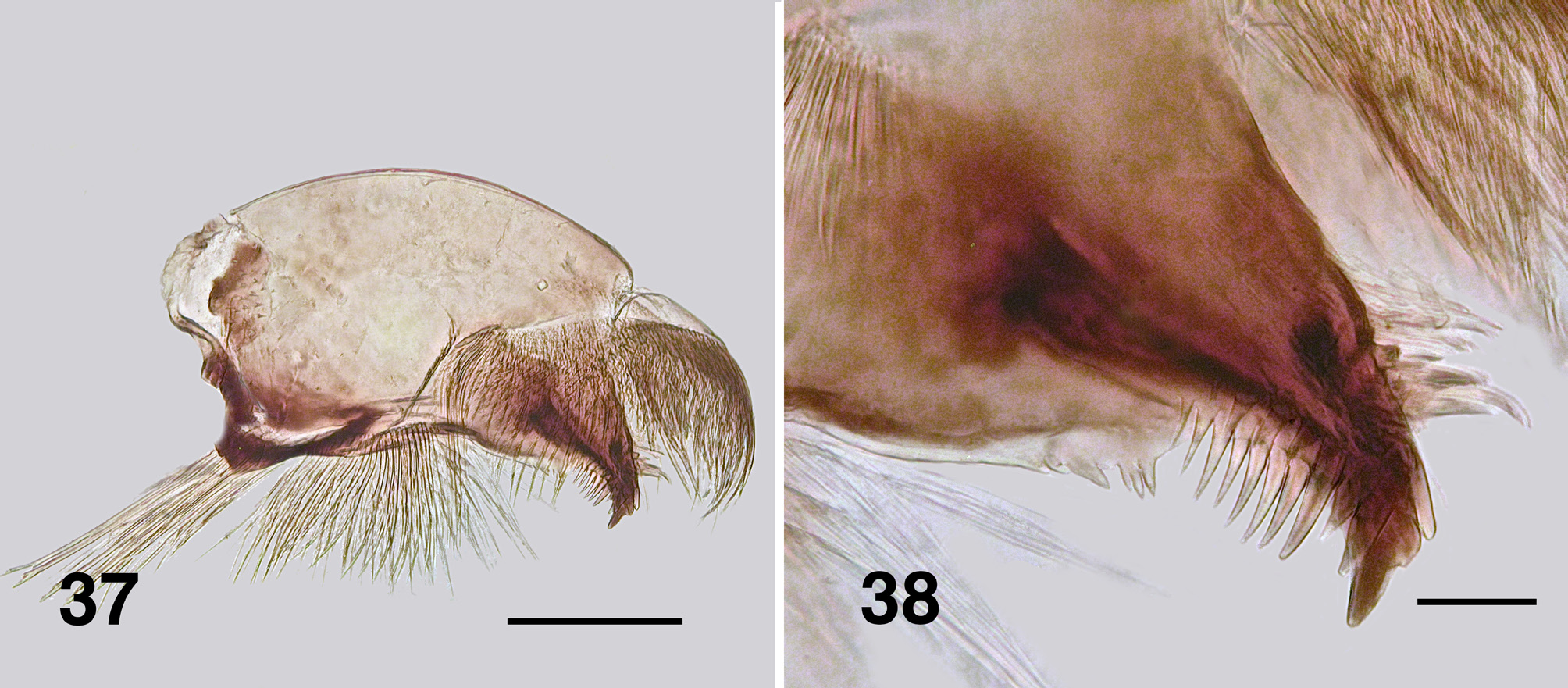
Larva (based on numerous last instar specimens). Body ( Fig. 32 View FIGURES 32–36 ): total length 5.0– 6.7 mm, creamy ventrally, segments I–III with distinct dark brown bands and pale intersegmental regions, posteriorly merging into medium brown. Head ( Fig. 33 View FIGURES 32–36 ): slightly narrowed anteriorly, margins not markedly convex posteriorly; length 0.70 mm, width 0.53 mm; distance between antennal bases 0.38 mm; anterior apotome creamy, head spot pattern positive usually dark brown, pattern variable, but commonly as a diffuse cross, darker posteriorly across the apotome; laterally two spots each just ventral to stemmata; ecdysial sutures distinct posteriorly; postocciput markedly expressed, cervical sclerites small and fused to postocciput. Antenna ( Fig. 34 View FIGURES 32–36 ): total length 0.27 mm, extended slightly beyond labral fan stem; evenly light brown, basal antennomere short, medial and distal antennomeres subequal in length, proportional lengths of antennomeres I–III, 1.0:1.8:1.8. Labral fan: stem short and broad, slightly pigmented, ca. 40 fine rays, length 0.4 mm, width 0.005mm at mid-length; microtrichia all fine, long, subequal, ca. 0.023 mm long, regularly arrayed. Postgenal cleft ( Fig. 35 View FIGURES 32–36 ): broadly V-shaped with irregular apex; postgenal bridge mottled brown, concolourous with genae; ratio of hypostoma, postgenal bridge and cleft 1.0:1.6:1.6; posterior tentorial pits small, surrounding cuticle heavily sclerotized. Hypostoma ( Fig. 36 View FIGURES 32–36 ): teeth markedly variable, largely covered by ventral edge of hypostoma; tooth 0 (median) well expressed, but not protruded, tooth 1 (first sublateral) apparently absent, tooth 2 (second sublateral) small or non existent—variable even on same specimen, tooth 3 just protruding beyond ventral edge, tooth 4 (lateral) well developed and protrusive, teeth 5–7 apparently absent, replaced by points on hypostomal edge; four or five hypostomal setae per side. Mandible ( Figs. 37, 38 View FIGURES 37, 38 ): short and broad, anterior covering brush exacerbated; outer, apical and subapical tooth on narrowed apex, with the apical brush short and protrusive; eight spinous teeth; sensillum and three to five serrations distinct, on raised base; blade region, smooth and slightly convex. Maxilla ( Fig. 39 View FIGURES 39–41 ): lobe short and broad, palp well separated from lobe, short, 2× longer than basal width, patch of short hairs basally. Thorax ( Fig. 40 View FIGURES 39–41 ): anterior proleg with small, poorly expressed lateral plates; mature pharate pupal gill elongated, directed 45º posteroventrally, filament aligned with horn posteriorly then recurved anterodorsally. Ventral papillae: absent. Anal sclerite ( Fig. 41 View FIGURES 39–41 ): anterodorsal arm short and not expanded, interarm struts distinct, posteroventral arms elongated and smoothly tapered; accessory and semicircular sclerites absent. Rectal papillae: three simple lobes. Posterior circlet: ca. 100 rows, 20–25 hooks per row (total ca. 2500).
Types. While the specific status of Austrosimulium bancrofti and A. pestilens was initially muddled, that of bancrofti types was equally so. Taylor (1918: 168) described bancrofti from females from Eidsvold. There is no mention specifically of types, except in the article’s introduction that states “ The type specimens are contained in the Institute collection. ” There are no dates.
As given previously here, Mackerras and Mackerras (1948: 258) had access to Taylor’s type series of A. bancrofti . There were nine specimens, all female. This material, originally housed in the Australian Institute of Tropical Medicine, Townsville, had been transferred to the collection of the Division of Economic Entomology, C.S.I.R., Canberra; now the Australian National Insect Collection (ANIC), C.S.I.R.O, Canberra. Only two of those specimens were of A. bancrofti , the other seven were A. pestilens . Of the two bancrofti specimens, one with “ Allotype ” on the label was accepted by the Mackerras’ as the holotype. Another with “ Paratype ” appears to have been accepted as a paratype, but that is not fully clear. Dumbleton (1973: 557) and Bugledich (1999: 331), both accepted the “ Allotype ” specimen as the holotype and that is followed herein. Both these specimens are currently housed in ANIC and the “ Paratype ” specimen is accepted as a paratype.
Holotyp e. Pinned female on pith. Label data: [EIDSVOLD, / QUEENSLAND / Dr. T. L. Bancroft.] [Allotype / Simulium / bancrofti, Taylor]. No date. The type is in good condition, albeit missing maxillary palpi, and the minuten pin is badly corroded. ANIC. Examined DAC, 2011 . Paratype . One pinned adult . Label data: [EIDSVOLD, / QUEENSLAND / Dr. T. L. Bancroft.] [PARATYPE {F} / Si- mulium / bancrofti, Taylor] . No date. ANIC. Examined DAC, 2011 . Oddly , Bugledich (1999) did not mention paratypes . Date for the types from Eidsvold is unknown—T. L. Bancroft lived in Eidsvold for twenty years (1910–1930) and had many opportunities to collect simuliid material—as well as many other organisms, and he did ( Marks, 1973 ; Mackerras & Marks, 1973 ) . Other material. There are some hundreds of pinned A. bancrofti adults in the ANIC . Of interest is that there are 12 pinned specimens from Eidsvold and seven have similar labeling as on the type specimens regarding locality and collector . The remainder of these specimens were clearly used by Tonnoir in a detailed examination, dated 1923, probably those to which he referred to as “ paratypotypes”; with little doubt for his seminal 1925 study on Australasian simuliids. In the smaller ANIC collection of A. pestilens, there are no specimens from Eidsvold, raising the question as to whether the above seven specimens are the original ‘type’ series of Taylor and are not A. bancrofti, but of A. pestilens, never having been transferred or relabeled ? These specimens were not examined for this work, apart from recording label data . As well as the large holding of A. bancrofti in ANIC, a substantial alcohol collection, of mainly immatures, was provided to the author by Hiede and Peter Zwick. As already noted this material is now housed in ANIC and the Strickland Museum , University of Alberta (UASM) .
Etymology. Named after Dr. Thomas Lane Bancroft (1869–1933), who collected the original material. T. L. Bancroft was the son of Joseph Bancroft, co-discoverer of the cause of filariasis. Thomas was himself of note ( Mackerras & Marks, 1973) for his work on dengue, filariasis and the Queensland lungfish Neoceratodus forsteri (Krefft) .
Distribution ( Fig. 42 View FIGURE 42 ). Tonnoir (1925: 219) noted that although A. bancrofti was originally known only from Queensland at Eidsvold, (S25.36700° E151.1300°) where T. L. Bancroft was located ( Marks, 1979), Tonnoir had seen NSW material from Dawson River (S24.9700° E150.0500°) and Bumberry (S33.1600° E148.5100°), collected by Dr. E. W. Ferguson, 1-x-1916. Localities consolidated below are derived from literature citations, the large ANIC collection, from the Zwick material and the Atlas of Living Australia (ala.org.au); the list should, however, not be considered definitive. Dates are given in detail when known. For each State, localities are listed from north to south. Colbo (1974) showed a distribution map (his Fig. VIII) for A. bancrofti , as did Dumbleton (1973; his Fig. 10 View FIGURES 6–10 ) and these are updated here ( Fig. 42 View FIGURE 42 ). Colbo (loc. cit.) commented that A. bancrofti , unlike A. pestilens , occurred in dryer western Queensland and he wondered how populations survived—either with a drought resistant stage such as is known for A. pestilens , or by reintroduction when conditions were suitable. The latter point is of general interest to biogeographers of Australian arid regions (Razeng, et al., 2017; Majer et a l., 2018). Indeed, Simulium ornatipes Skuse is known from southwest of Alice Springs at the George Gill Range, Northern Territories (S24.2556° E131.5702°), one of the driest regions in Australia ( Davis et al.,1993).
Queensland.Annan Riv., Cooktown (S15.5400° E145.1900°), 20-vi-1971. Mt. Spec GoogleMaps (S19.000° E146.2590°), 22- iv-1966. Don Riv. (S20.000° E148.1900°), 16-iv-1947. Cattle Crk., Eungella Valley GoogleMaps (S21.1400° E148.2100°), 16-iii- 1977. Lotus Crk. (S22.3528° E149.0961°). Mackenzie Riv. (S23.1600° E149.4900°), Apr., June. Fitzroy Riv., Rockhampton (S23.3400° E150.4900°), Jan., Feb. GoogleMaps , 19-i-1951. Don & Callide Riv., Calliungal, (S23.6400° E150.3800°), Apr., May. Rannes (S23.9500° E150.1900°), April. Burnett Riv. (S24.5600° E151.8900°), 5-v-1947. The Brook, Jacky Small’s Crk. (S24.8500 ° E150.7100°), April, May. Theodore (S24.9400° E150.0600°),?- ii-1939. Dawson Riv., near Theodore (S24.9500° E150.0700°), Apr. Gin Gin Crk. (S24.9723° E151.9454°). Ceratodus (S25.3008° E151.0891°). Burnett Riv., Eidsvold (S25.3781° E151.0836°), 1-v-1947. Lochaber Crk., Burnett Riv., Eidsvold (S25.3800° E151.1700°). Tin Can Bay Rd. GoogleMaps (S25.9400° E152.9700°), 18-iv-1949. Boonara & Upper Barambah Crk. GoogleMaps , Goomeri (S25.9400° E151.8900°), Apr., May. Lower Barambah Crk. GoogleMaps , Gayndah (S25.6200° E151.6300°), Apr. May GoogleMaps , 5-v-1947. Nambour (S26.2400° E152.5700°), Feb., Apr. Golden Beach, Noosa (S26.3100 E152.7400), 2-v-1949. Six Mile Crk, Cooroy (S26.4000° E152.7500°), 2-v-1949. Muckadilla Crk., Roma (S26.5700° E148.7900°), Mar.- Aug. Gympie (S26.6300° E152.9200°), Feb., Apr. Miles (S26.6500° E150.1800°). Brisbane, 15 km, W of Chinchilla GoogleMaps (S26.6700° E150.3900°), 12-i-1997, 16-viii-1947. Charley Crk., Chinchilla GoogleMaps (S26.7400° E150.6300°), Apr. Back Crk., Dalby (S27.2690° E151.2370°), Apr. Brisbane Riv., Wivenhoe (S27.4300° E152.6300°), Apr., May. Dunwich, Stradbroke Island GoogleMaps (S27.5100° E153.4000°), 11-viii-1951. Blunder Crk., Oxley (S27.5932° E152.9970°), 14-v-1950. Albert Riv., (S27.7700° E153.1900°), June. Nerang (S27.9900° E153.3000°), 23-iii-1947. St. George GoogleMaps (S28.0400° E148.5600°). Mudgeeraba Crk. (S28.0800° E153.3700°), Mar., May GoogleMaps , 23-iii-1947. Duraki (S28.1873° E151.6243°). Cave Crk. GoogleMaps , nr Numinbah (S28.2218° E153.2406°), 16-iii-1992. Wilson’s Peak (S28.2499° E152.4833°). Bransby, Nockatunga (S28.3700° E142.0200°), 9-xi-1949. McIntyre Riv., Goondiwindi (S28.5400° E150.2900°), Aug. Weir on McIntyre Riv. GoogleMaps , Goondiwindi (S28.5553° E150.3130°),?-?-1947. Texas (S29.0582° E150.8438°). Gravesend (S29.6500° E150.2000°), 28-ix-1952. Upper Taylors Arm GoogleMaps (S30.0700° E152.6333°).
New South Wales. Moree (S29.3000° E149.4900°), 16-v-1951. Collarenebri (S29.4200° E148.2600°), 21-ix- 1950. Yargobi Crossing (S29.5347° E150.2472°), 5-xi-1951. Bingara (S29.8300° E150.5000°), 24-ix-1952. Bourke (S30.1100° E145.9400°), May. Gooch’s Water GoogleMaps , Ebor (S30.4000° E152.3400°), 22-x-1950. McCleay Riv., Willawarrin (S30.9300° E152.6200°). Bumberry GoogleMaps (S33.1600° E148.5100°), 1-x-1923. Nyngan (S31.5571° E147.1849°),?- ix-1952, 3-iii-1956. Barrington Tops, Gummi (S31.9000° E151.4600°), 2-iii-1951. Lochaber Crk. (S33.1739° E149.9573°), 25-iv-1947. Molong Crk., Mt. Canoblas (S33.3378° E149.0181°), 8-x-1950. Forbes (S33.3900° E147.8600°), 14-xi-1964. Fish Riv., Bathurst (S33.5000° E149.6900°), April. River Lett, Hartley (S33.5277° E150.2006°) Dec. Hornsby GoogleMaps (S33.6900° E151.0800°), 23-xii-1952. Colo Vale (S34.4050° E150.4600°), 25/ 26-ii- 1958. Euston (S34.5901° E142.7532°). Minnamurra Falls GoogleMaps , nr. Wollongong (S34.6310° E150.7420°), 22-ii-2001. Tallong (S34.7500° E150.0400°). Yass, (S34.8200° E148.9000°), Aug., Nov. Goodrabigbee Riv., Wee Jasper GoogleMaps (S35.1631° E148.6868°), 18-iv-1964. Shoalhaven Riv., N. of Braidwood (S35.3428° E149.7375°), 22-x-1952, 1-xi-2007. Deniliquin (S35.5400° E144.6100°),?- ix-1926. Logbridge Crk., 19 km w of Corryong (S36.2100° E147.7100°), 13-xi-2007. Biggara (S36.3401° E148.056°). Leatherbarrel Crk., Mt. Kosziusko Nat. Prk. GoogleMaps (S36.5257° E148.1914°), 5-x-2002. Wullwye Crk., Dalgety (S36.4896° E148.8358°), 21-xii-1972.
Australian Capital Territory. Canberra (S35.2800° E149.1200°), 18-x-1929, 2-x-1931, 13-xii-1950. Molonglo Riv. GoogleMaps (S35.2800° E149.0400°), Sept-April, 18-ix-1931. Black Mt. GoogleMaps (S35.2800° E149.1100°), June, Nov., 22-x- 1929, 16-x-31. Mt. GoogleMaps Coree (S35.3000° E148.8000°), 22-iii-1955. Murrumbidgee Riv. GoogleMaps (S35.3200° E148.9500°),?- i-1934, 21-ix-1953, 25-ii-1955, 11-x-2014. Cotter Riv. GoogleMaps (S35.3200° E148.9400°), Nov-April. Cotter Riv., Bridge (S35.3258° E148.9397°), 11-xi-1929,?- viii-1972. Lee’s Spring GoogleMaps (S35.3500° E148.8000°), Oct., 21-iii-1951. Blundells GoogleMaps (S35.3600° E148.8100°), Sept, Dec, Feb., 24-x-1930, 18-i-1931, 20-xi-1934, 22-x-1938. Brindabella GoogleMaps (S35.3600° E148.5100°), Oct., 17-xi-1924, 20-iii-1930, 21-xi-1962. Pierces Crk. GoogleMaps (S35.3600° E148.9000°), 12-ix-1950. Tidbinbilla GoogleMaps (S35.4400° E148.9400°), 11-xi-1953. Mt. Gingera GoogleMaps , Brindabella (S35.5700° E148.7700°), 31-i-1951.
Victoria. Murray Riv., west of Merbein GoogleMaps , (S34.1600° E142.0500°), Dec. Jingellic GoogleMaps (S35.9625° E147.5086°). Barmah GoogleMaps (S36.0192° E144.9549°). Tallandoon, Mitta Mitta GoogleMaps (S36.4393° E147.2033°). Callaghans Creek Rd GoogleMaps (S36.5078° E147.4175°). Mitta Mitta GoogleMaps (S36.5202° E147.3703°), (S36.5624° E147.4106°), (S36.5153° E147.4349°). Lake Buffalo, (S36.7383° E146.6616°), 12-iv-1972. King Riv., Whitfield GoogleMaps (S36.7529° E146.4247°), 12-iv-1972. Lower Tablelands Rd. GoogleMaps (S36.7968 ¡ E147.6741°). Omeo Valley GoogleMaps (S36.9638° E147.6044°). Howqua Riv., Lake Eildon (S37.2366° E146.2291°), 12-vi-1972. Lee’s Spring, Mt. Franklin GoogleMaps (S37.2600° E144.1490°), 1-i-1952. Jamieson Riv. (S37.3100° E146.1100°), 16-ii-2001. Taggerty Riv. (S37.3218° E145.7080°), 11-x-2002. Acheron Riv., Buxton (S37.4168° E145.6984°), 13-x-1951. Steavenson Riv., Buxton (S37.4257° E145.7076°), 3-ii-1973. Buchan Riv. (S37.5008 E148.1734), 1-v-2012. Branxholme (S37.8400° E141.65000°), 12-viii-1958. Stratford/Avon. Gippsland (S37.9800° E147.0960°), 20-xi-1972.
Western Australia. Bullsbrook (S31.6700° E115.9900°), 3-viii-1953. Hovea (S31.8700° E116.0800°), 29-ix- 1935. Mundaring (S31.9100° E116.1600°), 23-viii-1926. Perth (S31.9500° E115.8300°),?- vii-1930. Serpentine Falls GoogleMaps , S of Perth. (S32.3679° E116.0111°), 26-x-2005. Serpentine Riv. (S32.3684° E116.0103°),?- iv-1972. Serpentine Riv. (S32.3700° E115.9900°), Apr. Groper, Harvey (S33.0800° E115.8900°),?- iii-1972. Kirup (S33.7100° E115.8500°), 29-viii-1926.S. Bridgetown(S33.9500° E116.1300°), 29-viii-1926, 27-x-2005.Bridgetown(S33.9700° E116.1300°). Donnely Riv. NW Pemberton (S34.0800° E115.9400°). Channeybearup (S34.4000° E115.9000°), 5- x-1970. Beedelup Falls GoogleMaps , NW Pemberton (S34.4189° E115.8674°), March 1972. Carey Brook, Beedelup Nat. Pk. (S34.4345° E115.7986°),?- iii-1972. Pemberton (S34.4400° E116.0300°), 15-iii-1956, 6-x-1970.
Tasmania. Brisbane River, Wivenhoe (S41.0800° E145.9200°), 5-v-1947. Devonport (S41.1700° E146.3200°), 18-x-1933. Great Forester River (S41.2673° E147.5070°). Launceston (S41.4400° E147.1200°), Apr. Launceston Gorge (S41.4510° E147.1144°), 23-iv-1946, 8-xii-1972. Meander River GoogleMaps , Birralee Road (S41.4947° E146.8172°). Ben Lomond, S Perth (S41.5600° E147.5200°), 8-xii-1972. Evendale GoogleMaps (S41.5700° E147.2400°), 1-iii-1964. St. Pauls River, upstream of Royal George (S41.8248° E147.9757°). Jordan River, Mauriceton (S42.5273° E147.1244°). Clyde River GoogleMaps , below Hamilton (S42.5623° E146.8267°). Muntain Riv., Ranelagh (S42.9378° E147.1348°).
In general, A. bancrofti distribution ( Fig. 42 View FIGURE 42 ) is markedly similar to that of other Australian Gondwanan Simu-liidae—eastern and western, absent from the arid center, some present in Tasmania. Similar to those taxa, the WA form of A. bancrofti is likely a separate sister species, as well maybe that in Tasmania; this latter alluded to by Dumbleton (1973) for Tasmanian Austrosimulium spp. These situations warrant further investigation. Of possible interest with regard to eastern A. bancrofti as a species complex, is the apparent gap in distribution north of the ACT and the northern edge of NSW. For Tasmania, the distribution of A. bancrofti appears to follow Tyler’s Line ( Shiel et al., 1989; discussed by Craig et al., 2019) where there is a sharp diagonal cutoff to the west, reminescent of that for Austrocnephia orientalis (Mackerras and Mackerras) ( Craig et al., 2019) . These distributions may represent, however, an artifact of collection.
Remarks. Given the dates, a Western Australia specimen of A. bancrofti in the ANIC, reared by Drummond and labeled as follows [From pupa/ serpintine(sic)/ 16.4.20 / coll. F.H.D.] [Type {M}/ Austrosimulium / bancrofti] [Serpintine(sic)/ 10.4.20 / coll. F.H.D.] [ Simulium / bancrofti {M}/ Drawn) Tay/ ALLOTYPE], is considered to have no status as a Type. This peculiar usage of ‘type’ raises the question as to why the specimen accepted generally as the holotype, has the word “ Allotype ” on the label when all the specimens that Taylor had were female?
Drummond (1931) when describing the pupa remarked on the “ringed” nature of gill filaments adjacent to their insertion on the gill stem. Close inspection ( Fig. 26 View FIGURES 24–29 ) indicates it is actually concertinaed and of unknown function—perhaps flexibility? As far as known, such a character state is unique within Simuliidae . An expectation here, is that it would occur in the ‘sister’ species of the bancrofti species-group, namely A. pestilens and A. magnum —indeed, distinctly in the latter, less so in the former (DAC, pers. obs.). This character is possibly definitive for the species-group.
Bionomics. Because of serious biting by female adults of A. bancrofti , the species has been well investigated. Tonnoir (1925), oddly, made no comment, but Mackerras & Mackerras (1948: 258) gave considerable details. In short, at the Eidsvold type locality, few adults were netted even though a large population of immatures was present, that suggested to them rapid dispersal; albeit this might have been a timing issue. Immature stages were found in moderately clear, swift water (velocity 1.2 m /s), attached to clean substrates, either stones or larger vegetation. Larvae formed clumps with pupae in crevices out of the full force of water. The larvae could occur to a depth of 38 cm and were commonly associated with A. pestilens and S. nicholsoni Mackerras & Mackerras. Pupae took some three days to develop before the adults emerged. Mackerras & Mackerras (1949: 396) noted that A. bancrofti occurred after the peak emergence of A. pestilens ; both associated with prior flooding. A drought-resistant stage was suggested. Mackerras & Mackerras (1952: 110) recorded that A. bancrofti females bit humans, dogs and rabbits.
Colbo (1974) made extensive investigation into the biology of Queensland simuliids, including climatic and geographic aspects. While eggs of A. pestilens had been discovered in vast numbers in substrate deposits, oviposition behaviour for A. bancrofti was not known. Determination of number of larval instars indicated that A. bancrofti had seven larval instars, of which the first instar possessed distinct labral fans—similarly known for other Austrosimulium spp. (e.g., Crosby, 1974; Craig et al., 2012). Early instar larvae drifted into faster water, which suggested oviposition might be upstream of such areas ( Colbo & Moorhouse, 1979). Velocities in larval habitats ranged from 1.2 to 1.3 m /sec., in close agreement with Mackerras & Mackerras (1948). The larval period ranged from 18–25 days under the conditions of Colbo’s observation. Of note, similar to Mackerras & Mackerras (loc. cit.), was that A. bancrofti larvae tended to clump together, even attaching to one another—given the high velocity of the water, a now well-known ploy to produce skimming flow and ameliorate drag (e.g., Moulton et al., 2018). This phenomenon is probably similar for A. bancrofti pupae, that formed groups numbering 8–85, all which were at the same stage of development. With regard to the sequence of species associated with flooding, A. pestilens occurred when discharge was high and water muddy, with A. bancrofti later at more moderate flows and clearer water; S. ornatipes occurred when flow was at base levels. Maximum breeding period was from June to September, less so during October–December when water flow was low and substrates covered with algae. Water temperatures of 20–28°C were recorded.
Colbo (1974: 189) recorded parasites from larvae of A. bancrofti . These appeared to the Blastocladiales Coelomycidium (?) simulii (Debais) . The description is similar to that given by Craig et al. (2012) for examples from larvae of New Zealand Austrosimulium . Colbo also recorded the microsporidians Thelohania Henneguy and Pl (e)istophora (sic) Gurley. Also noted were unidentified mermithids in adults, albeit at markedly low levels and other nematodes, although apparently not of Onchocerca . No nematodes were recovered from A. bancrofti larvae, although such were moderately common in associated species, so Colbo (loc. cit.: 321) concluded that even though a nematode was known from A. bancrofti , transmission or any disease significance remained unlikely. Hunter & Moorhouse (1976a) described a gynandromorph and intersexes of A. bancrofti females that were infected with, again, unidentified mermithids.
Tang et al. (1996) in a phylogenetic analysis of simuliid vectors of human and bovine onchocerciasis, used A. bancrofti as the out-group taxon. They state that no Australian simuliids transmit the causative organism.
Armaments, such as teeth on the cibaria of biting flies, are generally considered as a first line defense against filarial pathogens. Colbo et al. (1979), examined cibaria of a suite of Simuliidae female adults, including A. bancrofti . Of the five species of Austrosimulium dealt with, all were similar with only fine spine combs on the cibarial cornua at the junction with the pharynx. Reid (1994) also examined the cibaria of A. bancrofti and other simuliids. In agreement with Colbo et al. (loc. cit.) the cibarium of A. bancrofti lacks teeth, albeit possess ‘small spicules’, classified as ‘ Type 2 cibarial armature’. These are not visible in Fig. 6 View FIGURES 6–10 here. Reid (loc. cit.) lists A. bancrofti as a non-vector of filarial pathogens, not at variance to Colbo’s (1974) conclusions, or later, Tang’s (1996). On the other hand, earlier work by Lee et al. (1957), Lee et al. (1962: 374), plus Fenner & Radcliffe (1965: 198) suggested that A. bancrofti females are probably involved in transmission of livestock diseases including the causative virus of myxomatosis in rabbits.
Lichtward & Williams (1990) recorded the trichomycetes Harpella melusina Leger & Duboscq , Smittium simulii Lichtward and Paramoebidium spp. from larvae of A. bancrofti from widely disparate localities.
Sex ratio from a large sample of reared pupae was 46.4% males and 53.6% females (Colbo, 1974: 240). The major peak of adult emergence was 18:00 hrs ( Colbo, 1977) and fly activity appeared to be related to temperature, females not being very active below 14°C. Oviposition behaviour of A. bancrofti is still unknown, albeit that for A. pestilens is well established (Moorhouse & Colbo, 1973; Hunter, 1979), the latter females deposit eggs on the water. A general assumption is that A. bancrofti females do the same. Females of bancrofti appear to be non-autogenous (requiring a blood meal for egg production). Average number of eggs recovered from gravid truck-trapped females was 123 (range 52–198)—considerably lower than for other species.
Hunter & Moorhouse (1976b) in a major comparison of bionomics of A. pestilens and A. bancrofti , noted that A. bancrofti larvae inhabited more permanent waterways and the adults dispersed soon after emergence, the latter observation in agreement with those earlier. Females congregated on hill tops some distance from the breeding site, presumably where mating took place. As noted by others, trapping of adults was influenced by temperature, as well as wind, light intensity and rain. Few adults were collected below 15°C, with no reduction in numbers at temperatures up to 30°C. Oddly, during that particular study, females did not bite any of the animals tested as hosts. Mermithid-infected females were also recovered. Hunter (1977) investigated sugar feeding and survival of adults of A. bancrofti , A. pestilens and Simulium ornatipes —the former survived longer than the latter two. Considerably more information on immatures of A. bancrofti and associated simuliids can be garnered from Colbo & Moorhouse (1979).
Other aspects. Drummond (1931) when describing the pupa remarked on the “ringed” nature of gill filaments adjacent to their insertion on the gill stem. Close inspection ( Fig. 26 View FIGURES 24–29 ) indicates it is actually concertinaed and of unknown function—perhaps flexibility? As far as known, such a character state is unique within Simuliidae . An expectation here, is that it would occur in the ‘sister’ species of the bancrofti species-group, namely A. pestilens and A. magnum —indeed, well in the latter, less so in the former (DAC, pers. obs.). This character is possibly definitive for the species-group.
Lee et al. (1962) investigating the possibility of biting flies in Australia as vectors of disease in domesticated animals, obtained definitive precipitin tests for humans, horses and rabbits from field-caught blood-fed A. bancrofti . In summation (their Table 3), they list A. bancrofti as occurring in all States except South Australia and the Northern Territories and known, overall, to attack humans, horses, dogs, rabbits, kangaroos, wombats and eagles. As noted previously, they considered the species to be second in importance to A. pestilens as a pest.
The work by William J. Ballard and associates on A. bancrofti is considerable. Ballard (1988) described a method for sexing mature last larvae of A. bancrofti , using the shape of stained gonads. Ballard & Barnes (1988) in a preliminary examination of trap shape in regard to attraction for simuliid females, reasoned that a simulated shape of a host might reveal host preferences. Indeed, A. bancrofti females from Ipswich were attracted more to an elongated trap simulating a bovine host. The differences in behaviour between two populations (Ipswich and Taylors Arm) led to the suggestion that A. bancrofti was a species complex. Ballard (1989) further investigated the effect of silhouette shape of traps on capture of A. bancrofti adults. In general, higher numbers were captured under conditions of low wind speed and high cloud cover, with temperature and solar radiation also important, respectively 19°C and 620 watts/m 2 optimal, in agreement with earlier observation. Ballard & Morton (1990) examined the different trapfinding behaviour of two populations of A. bancrofti in Queensland. They found considerable differences between the two populations. Bovines were found to be the most attractive bait, with humans, bandicoots and chickens not attractive; the latter three non-attractive baits at major variance to other observations.
Of significance, Ballard & Bedo (1991) cytologically examined larvae of Austrosimulium bancrofti from four sites in eastern Australia. Eight cytoforms were recorded. Two cytoforms (A, B) were from Ipswich, another from Willawarin, plus one from Canberra and four (A, B, C, D) from Eidsvold, the type region. Some cytoforms were collected in sympatry, others not. The data suggested that A. bancrofti was a complex of at least eight cryptic species. This agreed with variation in behaviour of A. bancrofti adult females and larvae from near Ipswich, Willawarin and Canberra, previously known ( Ballard 1989; Ballard 1990; Ballard and Barnes 1988; Ballard & Morton 1990; Ballard & Bedo, 1991), supporting the hypothesis that such populations of A. bancrofti were distinct sibling species. Ballard et al. (1992) used the above data as part of an examination of relationship between arthropods and onychophorans.
Bedo (1976) successfully prepared chromosomes from pupae and adults of a number of Australian simuliids, with those of A. bancrofti showing extensive asynapsis (i.e. failure of the pairing of homologous chromosomes during meiosis).
Ballard (1991) and Ballard et al. (1991) investigated colonization of artificial substrate by larvae of A. bancrofti . Larvae colonizing Perspex (aka Plexiglas) strips in the Brisbane River were collected during the Austral summer (January) and spring (October). Larvae of S. ornatipes and S. nicolsoni were taken in conjunction. For the latter study the majority of larvae were of the Ipswich cytoform. Determination of larval instars showed that there were six or seven instars in January and October respectively. Texture of the substrate was significant, with higher colonization on the rougher. Preferred velocity was high, 1.0– 1.4 m /s, in agreement with the earlier work by Mackerras & Mackerras (1948) and Colbo (1974).
Ballard & Elder (1992), and Elder & Ballard (1993) used a major outbreak of A. pestilens and A. bancrofti , following flooding of the Fitzroy River, Rockhampton, QLD in 1991, to investigate trapping methods to quantify such outbreaks. They used two trap types with CO 2 plus a human-baited station, and used the biting rates to suggest thresholds for initiating control measures.
Ballard (1994) used 12S ribosomal RNA to investigate the problem of the variable antennae of A. bancrofti females, in relation to those of A. pestilens . Material available possessed variable numbers of flagellomeres, with some having the apical unit partially divided. Those with eight distinct flagellomeres were all shown to be definitively A. pestilens . Those with a variable number of five to seven flagellomeres and an incompletely divided apical flagellomere, were A. bancrofti . Analysis indicated that A. bancrofti from Rockhampton, Canberra and Ipswich were monophyletic (cytoform A). Further, a suggestion was made that there may well be an undescribed cytoform (Rockhampton) beyond the previously noted eight, recognized earlier by Ballard & Bedo (1991). Otsuka et al. (2007) used 16S rRNA for a phylogeny of Oriental Simulium s.l., with A. bancrofti as one of the out-group. Moulton (2000, 2003), similarly used A. bancrofti in a molecular analysis of simuliid basal relationships.
Indication that A. bancrofti is a complex of entities is also signaled by the morphological discrepancies between the eastern Australian forms and that from Western Australia. This includes the genital fork (cf. Figs. 11, 12 View FIGURES 11–13 ) plus anal lobes and cerci of females (cf. Figs. 16, 17 View FIGURES 14–17 ), and maxillary palpi of males (cf. Figs. 21, 22 View FIGURES 20–23 ). Separate species would not be surprising given the situation of other Gondwanan simuliid genera in Australia (e.g., Currie et al., 2018; Moulton et al., 2018). With, however, the small amount of material available from Western Australia, new collections will be needed to resolve this state of affairs.
At a higher taxonomic level, Austrosimulium bancrofti was included by Gil-Azevedo & Maia-Herzog (2007) in a phylogenetic analysis of Southern Hemisphere Simuliidae , based on morphological characteristics. The relationship between Austrosimulium and Paraustrosimulium was well supported.
Austrosimulium bancrofti larvae were part of a study on the effect of salinity in stream ecosystems ( Rutherford & Kefford, 2005) and similarly, part of a study on effect on aquatic ecosystems in Tasmania of insecticide drift from orchards ( Brown & Walker, 2000).
Further work on Austrosimulium bancrofti should involve elucidating mating and oviposition behaviour of the adults. An effort also should be made to determine the specific status of Western Australia and Tasmania forms, to bring a similar level of understanding to that known for other Australian Gondwanan simuliids (e.g., Craig et al., 2017, 2019; Currie et al., 2018; Moulton et al., 2018). As well, distribution and relationships between the various cytoforms and their different biting behaviours (e.g., Ballard & Bedo 1991)—of considerable practical value in regards to control measures. There is still much to be done.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Austrosimulium (Novaustrosimulium) bancrofti ( Taylor 1918 )
| Craig, Douglas A. 2019 |
Austrosimulium
| Adler, P. H. 2019: 23 |
Austrosimulium
| Bugledich, E. - M. A. 1999: 231 |
Austrosimulium
| Ballard, J. W. O. 1994: 131 |
Austrosimulium
| Ballard, J. W. O. & Bedo, D. G. 1991: 338 |
Austrosimulium
| Dumbleton & L. I 1973: 484 |
Austrosimulium bancrofti
| Smart, J. 1945: 499 |
Austrosimulium bancrofti
| Drummond, F. H. N. 1931: 8 |
Austrosimulium bancrofti
| Tonnoir, A. L. 1925: 241 |
Simulium bancrofti
| Taylor, F. H. 1918: 168 |