Alpheus karplusi, Anker, 2022
|
publication ID |
https://doi.org/ 10.11646/zootaxa.5105.3.5 |
|
publication LSID |
lsid:zoobank.org:pub:0055F495-69B0-45A3-8BCD-128A569E9079 |
|
DOI |
https://doi.org/10.5281/zenodo.6343542 |
|
persistent identifier |
https://treatment.plazi.org/id/9F69E057-FFE1-6773-249E-0593FECB300A |
|
treatment provided by |
Plazi |
|
scientific name |
Alpheus karplusi |
| status |
sp. nov. |
Alpheus karplusi sp. nov.
( Figs. 1–4 View FIGURE 1 View FIGURE 2 View FIGURE 3 View FIGURE 4 )
Alpheus rubromaculatus .— Karplus et al. 1981: 6, fig. 2C (black-and-white photograph); Karplus 1987: 514, fig. 2C; Debelius 2001: 153 (part.), 1 colour photograph (shrimp from Egypt only) [nomen nudum].
Alpheus djeddensis View in CoL .— Vine 1996: 104 (colour photograph) [not A. djeddensis Coutière, 1897 View in CoL ].
See discussion for other records of Alpheus rubromaculatus [not A. karplusi sp. nov.].
Type material. Holotype: male (cl 10.5 mm), FLMNH UF 37011 , Saudi Arabia, Red Sea , Farasan Islands, Zahrat Durakah, 16°50’09.2”N, 42°18’22.7”E, fringing reef slope around sandy island, depth 2–6 m, leg. M. Berumen, A. Anker, P. Norby, 11.03.2013 [fcn BDJRS-2701]. GoogleMaps
Paratypes: 1 male (cl 11.4 mm), FLMNH UF 37016 , same collection data as for holotype [fcn BDJRS-2706] View Materials GoogleMaps ; 1 female (cl 7.4 mm), FLMNH UF 37070 , Saudi Arabia, Red Sea , off Thuwal, Shib Nazar East offshore reef, 22°19’19.2”N, 38°51’18.0”E, depth about 10 m, leg. A. Anker, P. Norby, J. Moore, 16.03.2013 [fcn BDJRS- 2827] GoogleMaps .
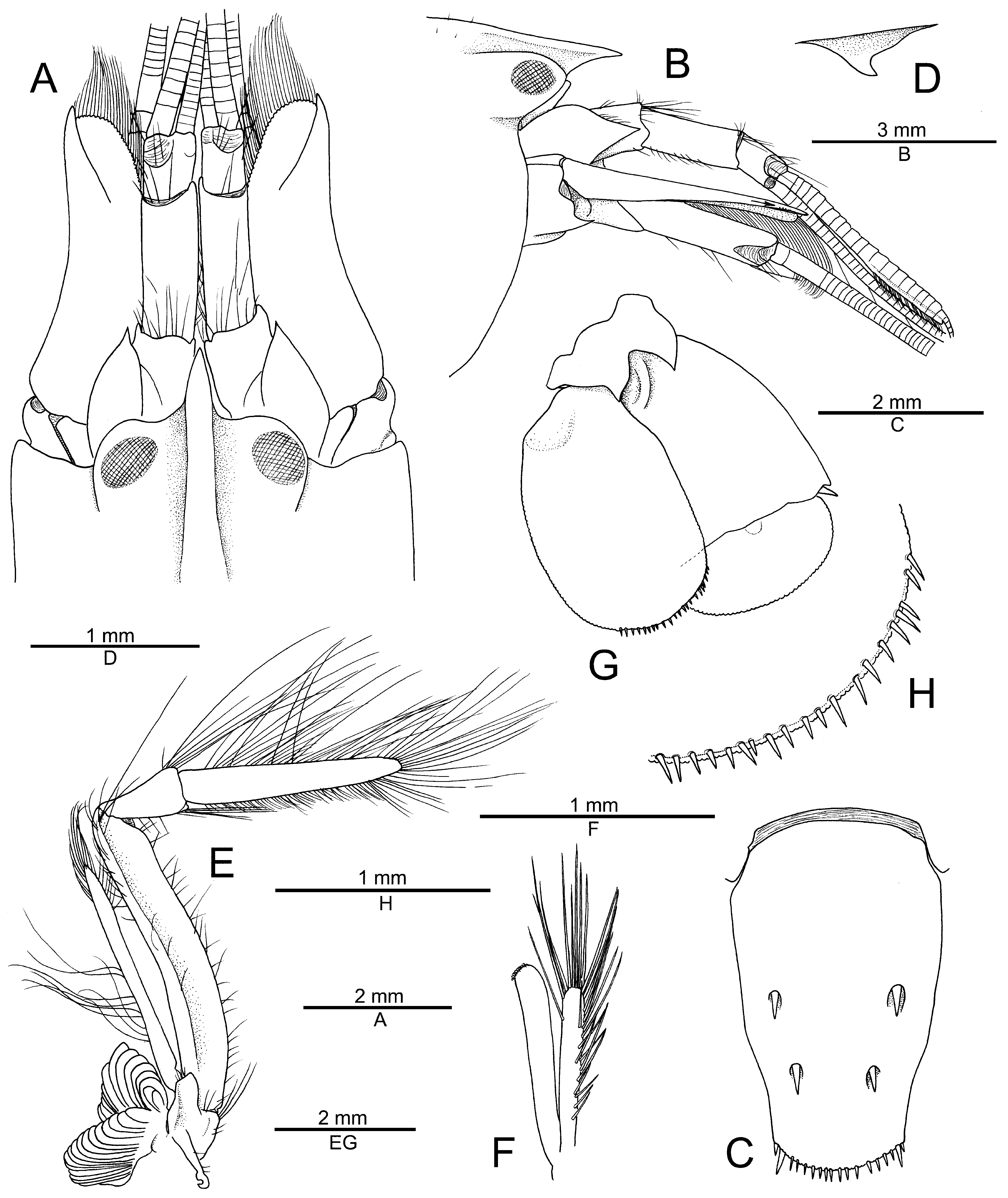
Description. Carapace glabrous, without setae, sparsely pitted ( Fig. 1A, B View FIGURE 1 ). Rostrum well developed, moderately slender, about 1.8–2.2 times as long as wide at base, straight, subacute distally, almost reaching distal margin of first article of antennular peduncle; rostral carina well developed, rounded dorsally, gently sloping into shallow adrostral furrows, continuing well beyond base of orbital hoods, gradually widening and fading, not reaching midlength of carapace ( Fig. 1A, B View FIGURE 1 ). Orbital hoods swollen, somewhat projecting anteriorly in lateral view, unarmed; frontal margin between rostrum and orbital hood shallowly concave ( Fig. 1A, B View FIGURE 1 ). Pterygostomial angle rounded ( Fig. 1B View FIGURE 1 ); cardiac notch deep.
Telson broad, subrectangular, gently tapering distally, about 1.7 times as long as maximal width, with lateral margins slightly convex at about 0.7 of its length, more noticeably tapering in posterior third; dorsal surface with two pairs of stout short cuspidate setae both inserted at some distance from lateral margin, first pair near telson mid-length, second pair between 0.7 and 0.8 telson length; posterior margin broadly rounded, with row of slender spiniform setae above plumose setae; posterolateral angles each with one pair of spiniform setae, mesial ones stouter and almost three times as long as lateral ones ( Fig. 1C View FIGURE 1 ).
Eyes with large, well-pigmented silvery cornea ( Figs. 1A, B View FIGURE 1 ; 3A, B View FIGURE 3 ; 4B, D View FIGURE 4 ). Antennular peduncle moderately long and stout; stylocerite slightly swollen laterally, ending in sharp point, latter reaching but not significantly overreaching distal margin of first article; ventromesial carina with anteriorly directed, shark-fin shaped tooth; second article about 2.7 times as long as wide; lateral antennular flagellum with secondary ramus fused to main ramus over most of its length, with numerous groups of aesthetascs distally, starting from about 13th subdivision ( Fig. 1A, B, D View FIGURE 1 ). Antenna with basicerite moderately stout, armed with sharp tooth on distoventral margin; scaphocerite with lateral margin shallowly and broadly concave; blade moderately broad, not reaching beyond stout, subacute distolateral tooth; scaphocerite overreaching both end of antennular peduncle and end of carpocerite ( Fig. 1A, B View FIGURE 1 ).
Mouthparts not dissected, typical for genus in external observation. Third maxilliped relatively slender, elongate, distally setose; coxa with bluntly projecting lateral plate; antepenultimate article flattened ventrolaterally, with distinct ridge running parallel to dorsal margin on lateral surface and almost smooth mesial margin, almost six times as long as high; penultimate article cup-shaped, not bulging ventrally, distally widening, its distoventral margin with tuft of moderately long setae, latter not reaching half-length of ultimate article; ultimate article densely furnished with rows of elongate stiff setae, dorsal and apical setae longest; arthrobranch well developed ( Fig. 1E View FIGURE 1 ).
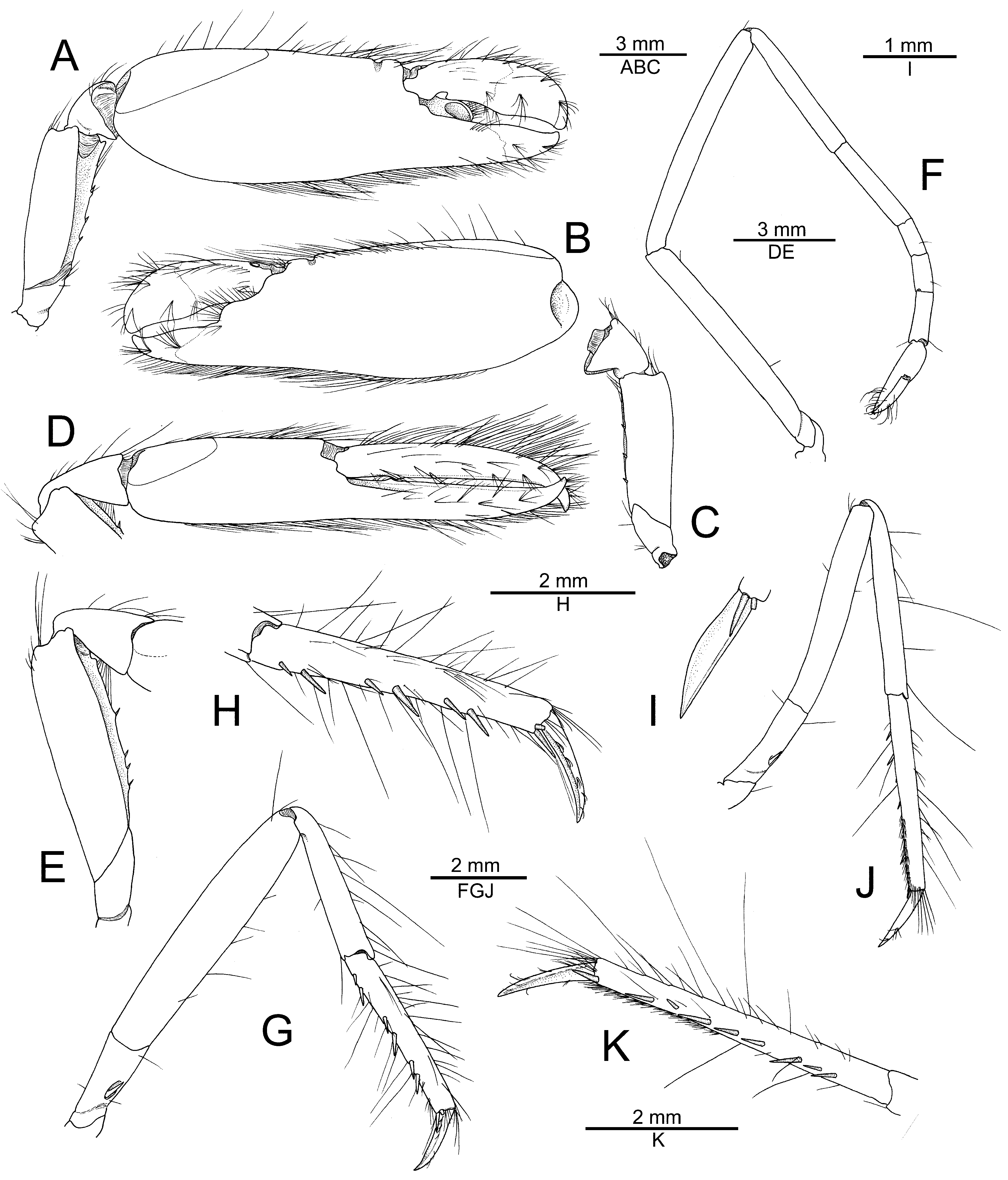
Major cheliped moderately robust; ischium stout, smooth; merus moderately stout, trigonal in cross-section, about three times as long as maximal width, distodorsal margin ending bluntly, ventromesial margin slightly rugose, with blunt distal tooth, armed with three spiniform setae; carpus short, wide, cup-shaped; chela not particularly elongate, with long setae along dorsal and ventral margins; palm strongly compressed, subrectangular in cross-section, length / maximal height ratio about 1.9–2.1; surfaces relatively smooth, not granulated; dorsal margin with well-marked transverse groove subdistally, without pronounced longitudinal ridges; ventral margin feebly convex; fingers somewhat unequal in length with dactylus slightly longer than pollex, both about half-length of palm, not twisted or significantly deviating from chela axis; dactylus distally rounded, with plunger greatly reduced, poorly demarcated from anterior cutting edge, latter feebly bulging; adhesive disks small ( Fig. 2A–C View FIGURE 2 ).
Minor cheliped relatively slender, not sexually dimorphic; ischium short, smooth; merus slightly slenderer than that of major cheliped, trigonal in cross-section, about 3.5 times as long as maximal width, distodorsal angle blunt, ventromesial margin slightly rugose, with small blunt distal protuberance, armed with four small spiniform setae; carpus longer than that of major chela, distally widening, vase-shaped; chela not particularly elongate or swollen, slender, fringed with long setae along dorsal and ventral margins; palm compressed, oval in cross-section, length / maximal height ratio about 2.2; surfaces smooth, not granulated; dorsal margin without groove; ventral margin straight; fingers subequal in length, approximately as long as palm, straight, slightly gaping when closed, not balaeniceps; cutting edges of each finger with broadly triangular protuberance proximally; adhesive disks reduced ( Figs. 2D, E View FIGURE 2 ; 3A View FIGURE 3 ).
Second pereiopod slender; ischium and merus subequal in length; carpus with five subarticles, first much longer than second, ratio of carpal subdivisions approximately equal to 2.3: 2.2: 1.0: 0.9: 1.5; chela longer than distalmost carpal subdivision ( Fig. 2F View FIGURE 2 ). Third pereiopod relatively slender; ischium with stout spiniform seta on ventrolateral surface; merus almost five times as long as maximal width, ventromesial margin unarmed distally; carpus about half-length of merus, much slenderer than merus, unarmed; propodus slightly longer than carpus, moderately setose, with about six spiniform setae along ventral margin and one distal pair of spiniform setae on distoventral angle near base of dactylus; dactylus about 0.4 length of propodus, gradually curving distally, subspatulate, moderately broadened, flattened ventrally, with longitudinal keel fringed by several tufts of setae ( Fig. 2G–I View FIGURE 2 ). Fourth pereiopod generally similar to third pereiopod, somewhat slenderer. Fifth pereiopod slenderer than third and fourth pereiopods; ischium with small spiniform seta on ventrolateral surface; merus about eight times as long as wide; carpus slenderer than merus, about 0.9 length of merus; propodus subequal to carpus, with rows of serrulate setae forming cleaning brush on distal third of ventrolateral surface, and eight or so spiniform setae along ventromesial margin, in addition to distal spiniform seta on mesial side of ventrodistal angle adjacent to base of dactylus; dactylus almost conical, only slightly broadened ( Fig. 2J, K View FIGURE 2 ).
Second pleopod with appendix masculina about 0.8 length of appendix interna, with long stiff setae, mainly along outer margin and on apex ( Fig. 1F View FIGURE 1 ). Uropod with mesial and lateral lobes of protopod each ending in sharp distal tooth; exopod broad, somewhat truncate distally, with blunt triangular distolateral tooth; diaeresis straight for most part, except for blunt lobe adjacent to stout spiniform seta, latter not reaching level of distal margin of exopod; endopod narrower than exopod, with row of small spiniform setae above plumose setae on distal margin ( Fig. 1H, I View FIGURE 1 ).
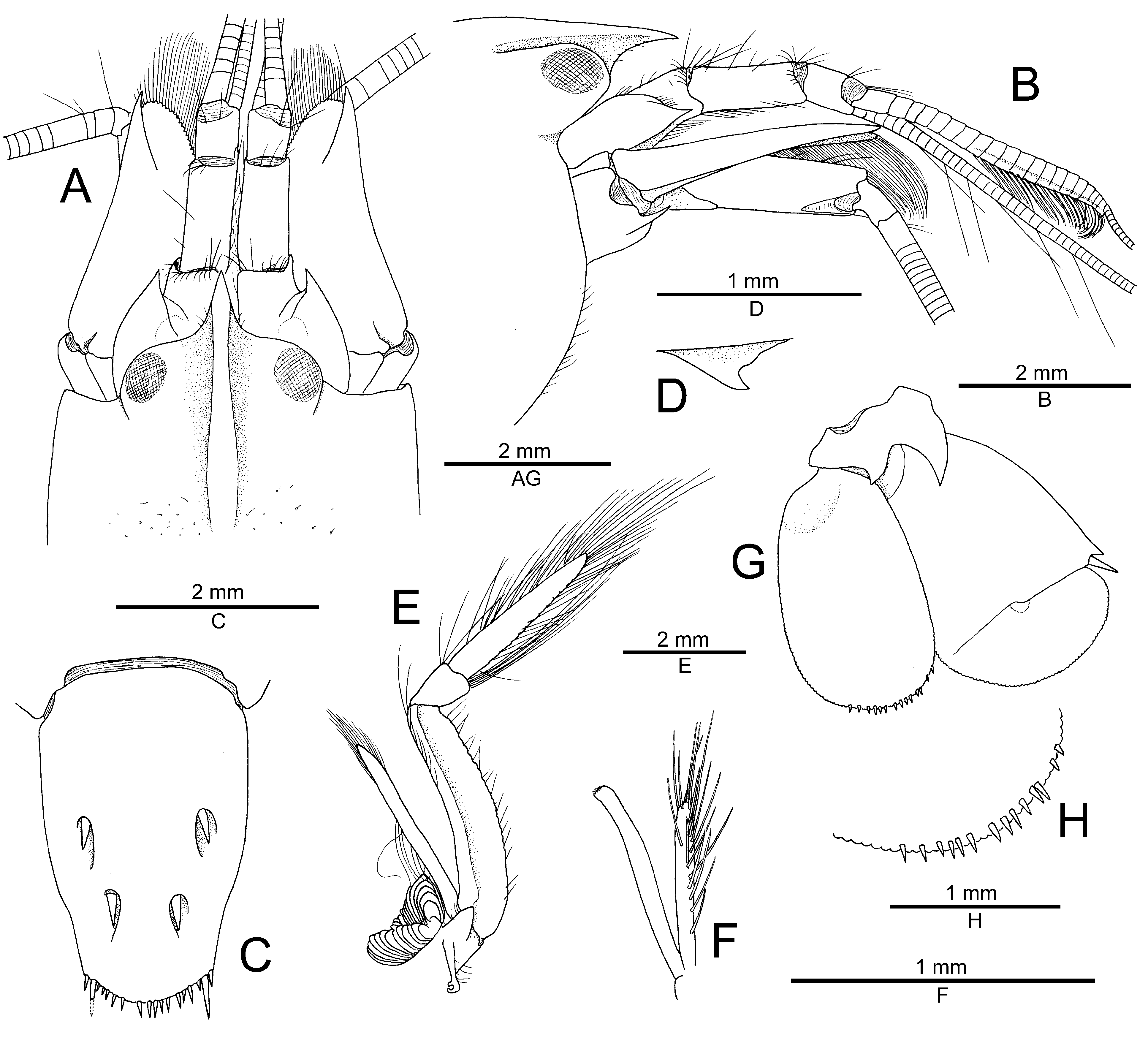
Colour pattern. General background semi-opaque pale buff, sometimes with yellowish or greenish tinge; carapace and pleon with larger or smaller, irregularly shaped (some rounded) and sized spots, blotches and short transverse bands of rusty-brown to orange-brown colour, some more or less organised in longitudinal lines, others without particular organisation; spots more numerous on pleon, tail fan and chelipeds, being somewhat larger on pleon flanks and chelae; second pereiopods and walking legs also with rusty-brown spots or transverse bands; posterior region of carapace and first pleonite with more or less conspicuous white saddle; antennular flagella yellowish; fingertips of major chela pinkish; ovaries in females (visible by translucence) olive greenish ( Figs. 3 View FIGURE 3 , 4 View FIGURE 4 ).
Etymology. This remarkable new species of Alpheus is named after Dr. Ilan Karplus for his important contribution to the knowledge of the biology and ecology of goby-shrimp symbioses ( Karplus et al. 1981; Karplus 1987, 1992; Karplus & Thompson 2011 and references therein).
Distribution. North-Western Indian Ocean: presently known only from the Red Sea, including Saudi Arabia (Farasan Islands and Thuwal, type material), Egypt (El Quseir, Safaga, Nuweiba; Fig. 4E–G View FIGURE 4 ) and Israel (Eilat, see Karplus et al. 1981; Fig. 4D View FIGURE 4 ).
Ecology. Coral reefs and adjacent reef flats with abundance of silt-covered rubble, at depths of 5–40 m (usually less than 20 m); associated exclusively with the whitecap shrimp goby Lotilia graciliosa Klausewitz, 1960 ( Vine 1996; Karplus 1987; Karplus & Thompson 2011; Debelius 2011; for the most recent review of the genus Lotilia Klausewitz, 1960 see Shibukawa et al. 2012).
Remarks. Alpheus karplusi sp. nov. has a striking and unmistakable, and therefore highly diagnostic, colour pattern, which is characterised by the presence of rusty-brown spots on a generally pale buff background ( Figs. 3 View FIGURE 3 , 4 View FIGURE 4 ). This colour pattern is in sharp contrast to all other known patterns among the goby-associated species of the A. djeddensis — A. djboutensis complex, separating A. karplusi sp. nov. at once from A. djiboutensis (sensu Holthuis 1958), A. bellulus , A. macellarius , A. fenneri , A. mannarensis , A. thompsoni , A. sciolii (see Miya & Miyake 1969; Bruce 1994; Debelius 2001; Kuiter & Debelius 2009; Minemizu 2013; Anker et al. 2015; Anker 2022), as well as the species called “ A. purpurilenticularis ” [nomen nudum] by Karplus et al. (1981). The colour pattern of A. djeddensis currently remains unknown, but the preliminary results of a morphological analysis of the Red Sea material morphologically corresponding best to A. djeddensis show that this species’ colour pattern is also very different from that of A. karplusi sp. nov. (A. Anker, in prep.). It is perhaps noteworthy that two more distantly related species of the A. brevirostris group, namely A. pubescens De Man, 1909 and A. williamsi Bruce, 1994 , also have brown spots and blotches, but not as distinct and well-separated as in the new species (cf. Bruce 1994; Anker & De Grave 2016).
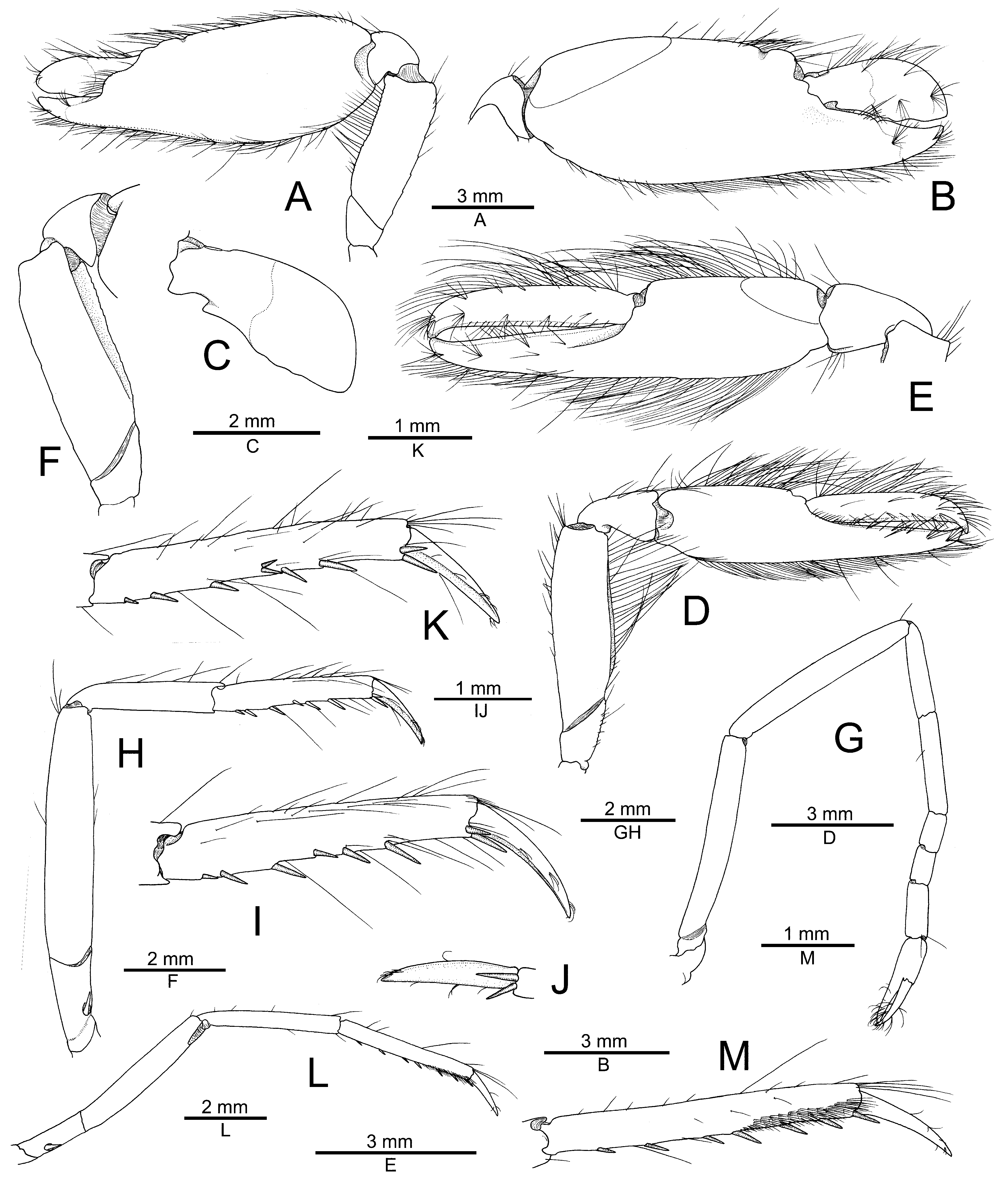
Based on morphological grounds alone, A. karplusi sp. nov. can be easily separated from A. djeddensis , A. djiboutensis , A. bellulus , A. fenneri and A. sciolii by the minor chela being much slenderer and lacking balaeniceps setae; these are present at least in males (or possibly in both sexes) in all the other species (cf. Coutière 1899; De Man 1909; Miya & Miyake 1969; Bruce 1994; Anker 2022). In addition, A. karplusi sp. nov. differs from A. djiboutensis , A. bellulus and A. sciolii by the noticeably slenderer cheliped meri, especially that of the minor cheliped; from A. djeddensis and A. fenneri by the strongly concave lateral margin of the antennal scaphocerite (vs. almost straight in the other two species); from A. djiboutensis and A. djeddensis by the glabrous carapace and pleon (vs. with more or less developed pubescence in the other two species); from A. djiboutensis and A. sciolii by the distolateral tooth of the antennal scaphocerite only slightly overreaching the blade (vs. reaching far beyond the blade in the other two species); from A. djiboutensis and A. bellulus by the noticeably slenderer major chela, with the palm twice as long as high (vs. at most 1.5 times in the other two species); from A. bellulus and A. fenneri by the less protruding rostro-orbital area and the second pereiopod with the first carpal subarticle clearly longer than the second (vs. the two being subequal in length in A. bellulus and A. fenneri ); from A. bellulus by the broader rostral carina and slenderer third pereiopod (noticeable in the width-length proportions of the merus); from A. fenneri , a much larger species at cl 19.5–23.0 mm, by the smooth major chela palm (vs. strongly granulated in A. fenneri ), the ventral margin of the male minor chela almost straight (vs. conspicuously convex in A. fenneri ), and the distoventral margin of the penultimate article of the third maxilliped furnished with a tuft of relatively few, moderately long setae (vs. with a dense field of numerous, elongate setae in A. fenneri , see also below); and from A. sciolii by the significantly longer and slenderer penultimate and ultimate articles of the third maxilliped (cf. De Man 1909; Miya & Miyake 1969; Bruce 1994; Anker 2022).
The remaining three described species of the A. djeddensis — A. djiboutensis complex, viz. A. macellarius , A. mannarensis , A. thompsoni , have similarly slender major and minor chelipeds, without balaeniceps setae or with a reduced balaeniceps condition. Nevertheless, A. karplusi sp. nov. may be morphologically separated from A. macellarius by the ventromesial margin of the chelipeds distally unarmed (vs. armed with a sharp tooth in A. macellarius ) and the shorter carpocerite, the latter not reaching the distal margin of the scaphocerite blade (vs. overreaching it in A. macellarius ) (cf. Chace 1988). The new species can be easily separated from both A. mannarensis and A. thompsoni by the second pereiopod carpus with the first subarticle clearly longer than the second (vs. with the second subarticle distinctly longer than the first in A. mannarensis or with the first and second subequal in length in A. thompsoni ) and the penultimate article of the third maxilliped furnished with a tuft of relatively few, moderately long setae on its distoventral margin (vs. with a field of more numerous and much longer setae in A. mannarensis and A. thompsoni ); individually from A. mannarensis by the mesial surface of the major and minor chelae not conspicuously granulated (vs. faintly granulated in A. mannarensis , although probably not as strongly as in A. fenneri ); and individually from A. thompsoni by the presence of at least three spiniform setae on the cheliped meri (vs. their absence or presence of only one spiniform seta in A. thompsoni ) (cf. Purushothaman et al. 2021; Anker 2022; Figs. 6 View FIGURE 6 , 7 View FIGURE 7 ).
The development of setae on the ventral margin of the penultimate article of the third maxilliped was briefly mentioned and illustrated by Banner & Banner (1981: 21, fig. 1i, j), who concluded that they represent a variable feature. However, it seems that the development of these setae is in fact species-specific and two groups can be distinguished. They are denser and extremely elongate, reaching almost to the end of the ultimate article, in some specimens identified as A. djiboutensis by Banner & Banner (1982), and also in A. bellulus , A. fenneri , A. mannarensis and A. thompsoni ( Miya & Miyake 1969; Bruce 1994; Purushothaman et al. 2021; Anker 2022; Fig. 6E View FIGURE 6 ). In contrast, they are less somewhat numerous and only moderately elongate, i.e. barely reaching the distal half of the ultimate article, in A. djeddensis sensu Banner & Banner (1982) , some specimens of A. djiboutensis sensu Banner & Banner (1982) , as well as in A. macellarius , A. sciolii and A. karplusi sp. nov. ( Anker 2022; A. Anker, pers. obs.; Fig. 1E View FIGURE 1 ).
The distribution range of A. karplusi sp. nov. appears to be restricted to the Red Sea ( Israel, Egypt, Saudi Arabia). All photographic records of “ A. rubromaculatus ” outside of the Red Sea most probably refer to a closely related, undescribed species, hereafter A. aff. karplusi (Anker, pers. obs.). These records include three colour photographs of “ Alpheus sp. 8 & 9” from Malaysia and Indonesia in Kuiter & Debelius (2009: 153); a blackand-white photograph of “ A. rubromaculatus ” from Malaysia (same photograph as in Kuiter & Debelius 2009) in Karplus & Thompson (2011: 591, fig. 4.4.10-C); two colour photographs of “ A. rubromaculatus ” from Malaysia (Mabul) and Indonesia (Bali) in Debelius (2001: 153); a colour photograph of “ Alpheus sp. 5 ” in Minemizu (2013: 103); and several previously unpublished colour photographs from the Philippines, Indonesia, Taiwan, Marshall Islands and Mauritius ( Fig. 5 View FIGURE 5 ). The main differences between the colour patterns of A. aff. karplusi ( Fig. 5 View FIGURE 5 ) and A. karplusi sp. nov. ( Fig. 3 View FIGURE 3 , 4 View FIGURE 4 ) are the clearly more yellow-green tinge of the body and appendages in the former species, including the more yellow-greenish chelipeds, bright yellow walking legs and conspicuously yellow or orange antennal flagella, as well as the spots on the carapace and pleon being more rounded and more dark red-purple in A. aff. karplusi instead of rusty-brown, as in A. karplusi sp. nov. In addition, A. aff. karplusi associates with different species of gobies and is allopatrically isolated from A. karplusi sp. nov. (see below). This widespread but as yet unnamed species, which is often incorrectly referred to as “ A. rubromaculatus ” (e.g. Debelius 2001; Pinault et al. 2015; marine aquarium trade), will be described when material becomes available for study.
Both A. karplusi sp. nov. and A. aff. karplusi are typically (but not exclusively) encountered in association with the whitecap hovering gobies of the genus Lotilia . According to Shibukawa et al. (2012), Lotilia is composed of two morphologically nearly identical species, which have been confused with each other until recently. The type species of Lotilia , L. graciliosa , is restricted to the “Red Sea and adjacent areas”, whereas L. klausewitzi Shibukawa, Suzuki & Senou, 2012 occurs in the “West Pacific” ( Japan, Australia, Papua New Guinea, Phoenix Islands), although based on numerous colour photographs, it is also widely distributed throughout the Coral Triangle ( Indonesia, Malaysia, Philippines). Based on the presently available evidence, A. karplusi sp. nov. may live exclusively with L. graciliosa in the Red Sea ( Fig. 4E View FIGURE 4 ), although this needs to be confirmed by more in situ observations. In contrast, A. aff. karplusi associates mainly with L. klausewitzi ( Fig. 5A–C View FIGURE 5 ), but also with three species of the genus Cryptocentrus Valenciennes, 1837 , viz. C. albidorsus (Yanagisawa, 1978) , C. leucostictus (Günther, 1872) and C. malindiensis (Smith, 1959) ( Fig. 5D–F View FIGURE 5 ; see also Kuiter & Debelius 2009; Pinault et al. 2015). Remarkably, and certainly not by coincidence, these three species of Cryptocentrus have conspicuously white-banded backs, as in the two species of Lotilia , suggesting the shrimps’ preference for a particular colour pattern of their goby partners. Among the shrimp-associated gobies, L. graciliosa and L. klausewitzi are unusual in that outside of the burrow they always hover above the burrow funnel ( Figs. 4E View FIGURE 4 , 5A–C View FIGURE 5 ), instead of resting on the sand in front of the burrow entrance or at the edge of the funnel, as do most other shrimp gobies, including C. leucostictus ( Fig. 5F View FIGURE 5 ). Interestingly, C. albidorsus and C. malindiensis seem to be somewhat intermediate between “bottom sitters” and hoverers ( Fig. 5D, E View FIGURE 5 ; see also Pinault et al. 2015: fig. 1). According to the author’s preliminary observations, there are no significant colour pattern differences between A. aff. karplusi associated with L. klausewitzi and those associated with Cryptocentrus spp. Nevertheless, it seems that most shrimps associated with L. klausewitzi are paler and have smaller spots ( Fig. 5A–C View FIGURE 5 ), whereas those associated with Cryptocentrus spp. are brighter yellow and have larger spots ( Fig. 5D–F View FIGURE 5 ), and also appear to be smaller in size. Whether one or two species are involved under A. aff. karplusi remains to be shown in the future.
| FLMNH |
Florida Museum of Natural History |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Alpheus karplusi
| Anker, Arthur 2022 |
Alpheus djeddensis
| Vine, P. 1996: 104 |
Alpheus rubromaculatus
| Debelius, H. 2001: 153 |
| Karplus, I. 1987: 514 |
| Karplus, I. & Szlep, R. & Tsurnamal, M. 1981: 6 |