Paralithoxus mocidade, Rapp-Py-Daniel & Oliveira & Bastos & Ito & Zuanon & Briglia-Ferreira, 2019
|
publication ID |
https://doi.org/ 10.1590/1982-0224-20190041 |
|
DOI |
https://doi.org/10.5281/zenodo.3663829 |
|
persistent identifier |
https://treatment.plazi.org/id/A9178793-FF8D-D826-8CCB-F913D2206C29 |
|
treatment provided by |
Carolina |
|
scientific name |
Paralithoxus mocidade |
| status |
sp. nov. |
Paralithoxus mocidade , new species
urn:lsid:zoobank.org:act:B5B7D3D6-F267-4ED5-A752-E5F64B461397
( Figs. 1–4 View Fig View Fig View Fig View Fig , 8–9 View Fig View Fig ; Tab. 1 View Tab )
Holotype. INPA 54745 View Materials , 50.5 mm SL, Brazil, Roraima State, Caracaraí Municipality, stream tributary to Ajarani River, Branco River basin, upstream of large waterfall, Serra da Mocidade , 01°42’52.70”N 61°48’17.64”W, 29 Jan 2016, D. A. Bastos, J. Zuanon, P. M. M. Ito and S. R. Briglia-Ferreira. GoogleMaps
Paratypes. All from Brazil, Roraima State, Caracaraí Municipality. INPA 52422 View Materials , 17 View Materials , 20.6–47.2 mm SL (1 c&s, 47.2 mm SL), stream tributary to Ajarani River , Branco River basin, downstream of large waterfall, Serra da Mocidade , 01°42’50.26”N 61°48’18.65”W, 29 Jan 2016, D. A. Bastos, J. Zuanon, P. M. M. Ito and S. R. Briglia-Ferreira . INPA 52424 View Materials , 17 View Materials , 15.0– 54.7 mm SL (1 c&s, 54.7 mm SL, 16 alc.) , MNRJ 51531 View Materials , 2 View Materials , 23.0–26.0 mm SL , MPEG 38600 View Materials , 2 View Materials , 27.0–30.0 mm SL , MZUSP 125192 View Materials , 2 View Materials , 25.0–28.0 mm SL , ANSP 206987 About ANSP , 2 About ANSP , 26.0–33.0 mm SL, all collected with holotype GoogleMaps .
Non-types. INPA 59028 View Materials , 14 View Materials , 13.8 View Materials –19.0 mm SL, collected with the holotype .
Diagnosis. Paralithoxus mocidade differs from all congeners, except P. bovallii , by having truncate teeth (vs. pointed to acute teeth). It differs from P. bovallii by the truncate caudal fin (vs. caudal-fin forked). Adittionaly, P. mocidade differs from P. pallidimaculatus and P. surinamensis by the presence of adipose fin (vs. absence). It differs from P. planquettei and P. stocki by having a more depressed and elongate body: body depth 10–13.9% of SL (vs. 15.3–16.8% in P. planquettei and 15.7–16.9% in P. stocki ), and body width at dorsal fin 19.3–23.0% of SL (vs. 23.3–26.5% in P. planquettei and 24.2–25.1% in P. stocki ); from P. raso by the larger interorbital distance, 28.8–33 % of HL (vs. 27.1–28.6%) and larger dentaries 5.9–9.3% of HL (vs. 4.4–5.3%); from P. stocki and P. boujardi by the caudal fin conspicuously mottled, with alternating dark and light blotches (vs. caudal fin dark with clear distal band); and from P. jariensis by having seven to eight plates between dorsal and adipose fins (vs. four) and 24–26 lateral median plates (vs. 23–24).
Description. Morphometrics and counts in Tab. 1 View Tab . Small species of Hypostominae , largest specimen examined 54.7 mm SL. Body very depressed and short. In lateral view, dorsal profile slightly convex from snout to dorsal-fin origin, descending almost straight to dorsal procurrent rays of caudal fin. Greatest body width at pectoral-fin origin; body of similar width between posterior base of pectoral fin and origin of pelvic fin, slightly narrowing from this point towards caudal-fin base. Caudal peduncle trapezoid in cross section. Ventral profile straight from snout to caudal-fin origin.
Head short, depressed and rounded in dorsal view. Snout tip hispid, completely covered by plates. Eye moderately small, dorsally positioned; interorbital region narrow and flat. Cheek plates with up to 20 hypertrophied odontodes. Parieto-supraoccipital large, hexagonal, with process posteriorly bordered by four plates. Predorsal area with three pairs of plates, misplaced in some specimens. Nuchal plate half-moon shaped; small but conspicuous. Seven to eight plates between dorsal and adipose fins. Body almost completely plated; abdomen naked. Body plates arranged in four longitudinal series anteriorly, five series between dorsal and anal fin, and five series on caudal peduncle. All plates covered by odontodes of similar size without carinae, except for two low keels on dorsal plates between dorsal and adipose fins. Ventral series of plates largely smooth, showing large median area without odontodes, except on nuptial male. Seven plates along dorsal- fin base; low preadipose plate. Median series with 24–26 plates not carinate; mid-ventral series not forming prominent keel. First anal-fin pterygiophore exposed, located anterior to anal-fin origin.
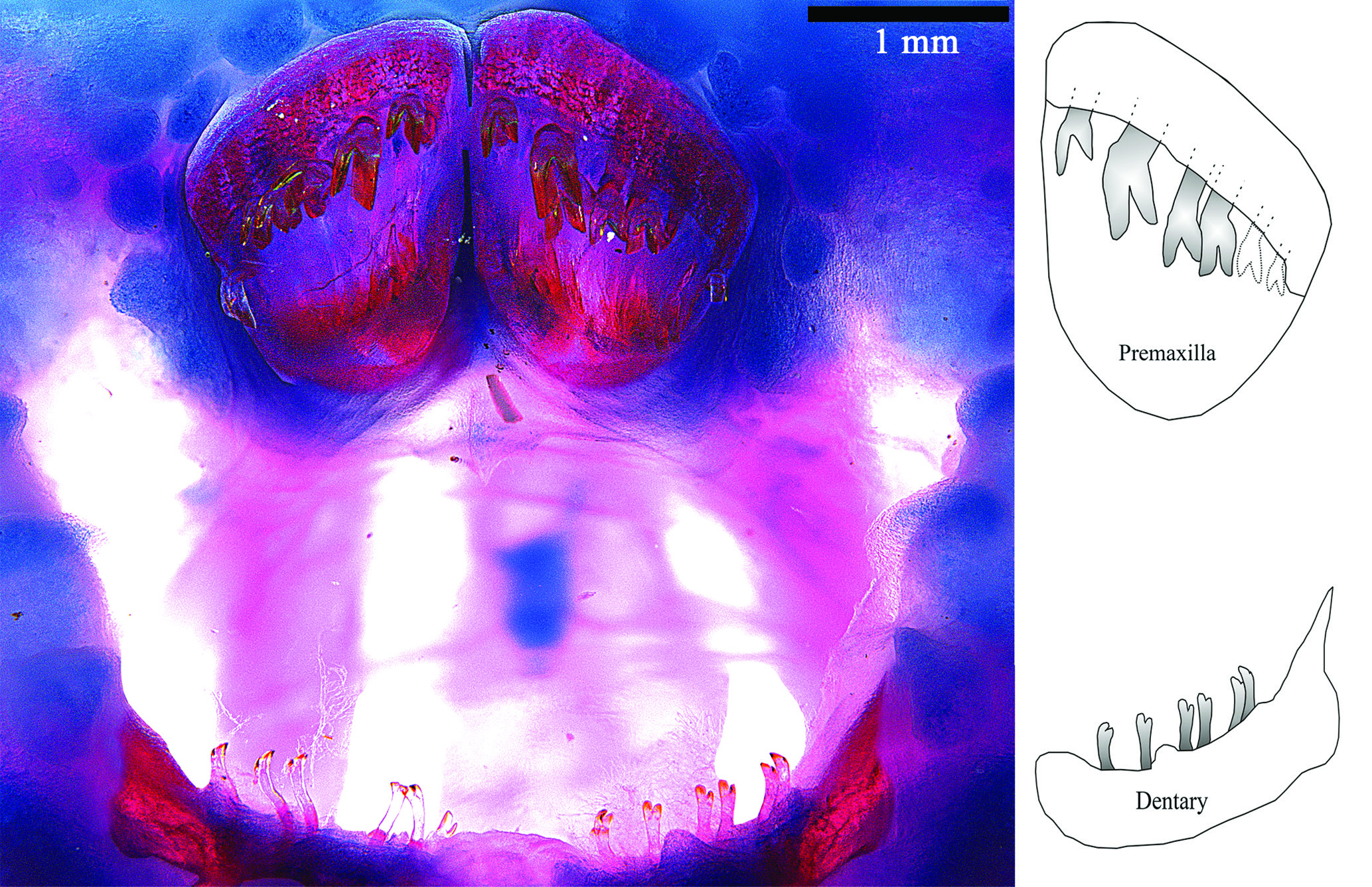
Oral disc large, nearly circular. Mouth opening U-shaped, wider than long. Oral internal papillae absent. Maxillary barbel very short, projected anteriorly from oral disc. Lips well developed. Whole border of oral disc covered with large round papillae. Surface of upper and lower lips covered by round papillae not densely packed. Lip surface with shallow notches mostly without papillae distributed at top of premaxillae, around dentaries and on midline on lower lip.
Teeth bicuspid; crown yellow and shaft translucent ( Fig. 4 View Fig ). Premaxillary teeth much larger than mandibular teeth, arranged in crescent from lateral to mesial. Cusps of premaxillary teeth asymmetrical; larger cusp with straight border, truncate; internal cusp slightly larger. Dentary teeth extremely delicate, bifurcate; cusps small, worn to similar size. Premaxilla with 5–6 and dentary with up to 8 teeth.
Dorsal fin II,7, branched rays decreasing in length posteriorly; last branched ray 1/3 shorter than dorsal-fin spine. Spinelet reduced, locking mechanism not functional and with small area exposed, covered by odontodes. Pectoral fin I,6; distal tip of branched rays not surpassing insertion of pelvic fin when adpressed. Pectoral-fin spine robust, flat and slightly curved inward; spine of equal size of first branched ray. Pectoral-fin spine distal region covered with hypertrophied odontodes, mainly in mature males. Adipose fin low but conspicuous, with small membranous extension posteriorly. Pelvic fin i,5, distal tip of branched rays surpassing insertion of anal fin when adpressed. First pelvic unbranched ray leaf-like, enlarged medially; whole ray flat and covered by odontodes ( Fig. 9b View Fig ). Pelvic fin larger than pectoral fin in majority of specimens; ratio between pectoral/ pelvic length with mean of 0.9 and standard deviation of 0.1. Anal fin ii,4, i,4 or i,5, branched rays coequal in length. Caudal fin i,14,i; seven branched rays on each lobe. Caudal fin almost truncate; principal rays mostly coequal in length, with lower rays slightly longer than upper lobe; all rays with small odontodes.
Color in alcohol. Body with dark olive-brown background with irregular light blotches; blotches slightly smaller and more densely organized on head than on trunk and fins. Four inconspicuous large transverse light bars on body: first on dorsal-fin origin, second large bar on posterior dorsalfin base, third anterior to adipose, and fourth on base of caudal fin. Ventral surface of body uniformly cream to beige colored. All fins with checkered pattern, alternating large dark blotches with clear ones.
Color in life. Very similar to preserved coloration, but with overall body color dark olive-brown on dorsum with light green spots on snout and cheek ( Fig. 2 View Fig ); thin creamy to light green vermiculations over interorbital area and posterior portion of head; clear marks on anterior portion of dorsum becoming larger and more irregular towards caudal peduncle. Ventral side of body uniformly beige.
Sexual dimorphism. Urogenital papilla small, but conspicuous on all specimens. The only externally mature male specimen was dissected and had a more melanic coloration, darkened on body and lips, losing bars and blotches along body and fins; dorsal, pectoral and pelvic fins with branched rays darkened showing light bar close to tip; caudal fin uniformly dark ( Fig. 3 View Fig ). Male with odontodes on cheek plates and pectoral fin more hypertrophied than on females. Mature male with fins more developed than females: pectoral spine large, strong, bearing more developed odontodes on distal half; first pelvic-fin unbranched ray strong, rounded for entire length and enlarged medially; branched rays on pelvic fin conspicuously longer than unbranched ray. Male has caudal peduncle covered with odontodes, except around anal fin, whereas females and juveniles present caudal peduncle ventrally smooth, without aligned odontodes.
Geographic distribution and habitat. Paralithoxus mocidade has only been recorded from an unnamed tributary of the Ajarani River, in the area of Serra da Mocidade, Branco River basin, Caracaraí Municipality, Roraima State, Brazil ( Fig. 5 View Fig ). All specimens were collected up and downstream of a 20-m high waterfall located at approximately 900 m elevation. The Ajarani River at the collection site is a small fast-flowing river (up to 15 m width and 2 m depth, as measured in dry season) with clear water, low temperature (about 21.7 ºC), acidic (pH 5.5), dissolved oxygen (DO) 20.7%, and smooth rocks on the bottom ( Fig. 6 View Fig ). During the day, juveniles were observed grazing in the open over smooth rocks, and adults were found in narrow cracks or under rock slabs during the night. Juveniles were captured with hand-nets and adults were collected by hand while snorkeling ( Fig. 7 View Fig ).
Etymology. The specific epithet “mocidade” is in allusion to the type locality, in Serra da Mocidade National Park. A noum in apposition.
Conservation status. Paralithoxus mocidade is known only from the type locality in a geographically isolated river system, which could generate concerns about its conservation status. However, the inaccessibility of the area and the fact that nearly two-thirds of the area of Serra da Mocidade are formally protected by the Serra da Mocidade National Park and Yanomami Indigenous Territory indicate that there are no imminent threats to the species, which can be categorized as Least Concern (LC) ( IUCN, 2017).
Taxonomic remarks. Paralithoxus mocidade and P. bovallii were recorded from the Branco River basin: P. bovallii from the Takutu (Branco)- Essequibo drainages and P. mocidade from the Ajarani River. Recent collections in the upper Ireng River by N. K. Lujan and J. W. Armbruster produced new specimens of P. bovallii from the type-locality, as listed in the redescription of the species by Lujan et al. (2018). Based on this and the original description ( Regan, 1906), we conducted a morphological comparison. Both P. bovallii and P. mocidade share a mottled color pattern on body and fins, a similar number of teeth on the jaws (6 to 8), same tooth shape (truncate) ( Fig. 4 View Fig ), and similar number of plates between dorsal and adipose fins. However, these species differ in eye size, with P. mocidade having a usually smaller eye, with orbit representing 9–12% (average 11%) vs. 11–19% (average 14.6%) in P. bovallii ( Lujan et al., 2018) . Based on Regan’s original ratios, the orbital diameter in P. bovallii was contained 6–7.5 times and interorbital distance 3 times in head length, whereas in P. mocidade , the orbit is contained 8–10 times and the interorbital 3–3.5 times in head length. Besides, P. bovallii has the pectoral fin larger than the pelvic whereas in P. mocidade pectoral and pelvic are equal or the pelvic is slightly larger: pectoral length 23.8–28.7% (mean of 25.5%) in SL in P. mocidade vs. 6.9–33.0% (26.2%) in P. bovallii ; pelvic fin 25.5–30.5% (27%) in SL in P. mocidade vs. 4.9–29% (22.8%) in P. bovallii ( Lujan et al., 2018) .
The other species described from the Essequibo River drainage is Lithoxus lithoides (also distributed in other drainages such as the Trombetas River, following Ferreira, 1993). Lithoxus lithoides is extremely different from Paralithoxus mocidade having larger eyes, larger pectoral fins (more developed in nuptial males), larger posterior membranous extension of the adipose fin, dorsal fin almost touching the adipose-fin plate, and three series of plates in the caudal peduncle. However, L. lithoides shares with P. mocidade the truncate teeth and membranous extension of the adipose (reduced in P. mocidade when compared to L. lithoides ). Some paratypes of L. lithoides were examined and measured, and differences in orbit and caudal peduncle were highly evident between the two species: orbit diameter ranged from 19–21% of head length in L. lithoides vs. 9.2–12.2% in P. mocidade ; caudal peduncle depth ranged from 18.8–23% of caudal peduncle length in L. lithoides vs. 27.6–33.5% in P. mocidade . Besides, L. lithoides has the premaxilla much smaller than the dentary, whereas in P. mocidade the toothed area in both jaws is of similar size, despite difference in the size of the teeth, reinforcing the general dissimilarity between both species.
Species of Paralithoxus recorded from French Guiana ( P. planquettei , P. boujardi and P. stocki ) share some features distinct from P. mocidade : teeth with acute cusps and caudal fin with distinct distal light bar vs. teeth truncate and caudal fin with alternating clear and dark blotches in P. mocidade . Besides, both P. boujardi and P. stocki have a larger pectoral fin (pectoral length 3.3–3.7 and 2.8–3.6 times in SL in P. boujardi and P. stocki respectively vs. 3.5–4.2 in P. mocidade ) and shorter caudal peduncle (caudal peduncle length 3–3.25 and 3.5 times in SL in P. boujardi and P. stocki respectively vs. 3.6–4.1 in P. mocidade ). Paralithoxus planquettei also has a shorter pectoral fin and a deeper caudal peduncle (caudal peduncle depth 9.8–11.75% in SL vs. 7.1–8.4% in P. mocidade ) and larger mandible (dentary length 2.5–2.8 times in interorbital distance vs. 3.6–4.9 in P. mocidade ).
Paralithoxus jariensis and P. raso were originally distinguished from other Paralithoxus species by a set of morphological features. Paralithoxus raso has a peculiar color pattern with light large blotches similar to P. pallidimaculatus ( Silva et al., 2017) . Both recently described species were recorded for the eastern part of the Amazon, in Pará and Amapá States. Paralithoxus jariensis is morphometrically very similar to P. mocidade but shows a reduction in number of plates: four plates between dorsal and adipose fin (vs. 7–8 in P. mocidade ) and 23–24 lateral median plates (vs. 24–26 in P. mocidade ). Paralithoxus raso differs from P. mocidade mainly by having a larger pectoral- fin spine (27–29% vs. 23–28% in SL in P. mocidade ), and smaller mandibles (premaxilla 3.3–4% and dentary 4.4–5.3% in head length in P. raso vs. 6.1–9.9% and 5.9–9.3% in head length in P. mocidade ). In their original description, the authors also conducted a molecular analysis using sequences of the COI mitochondrial gene for most available species of Paralithoxus ( P. planquettei , P. bovallii , P. pallidimaculatus , P. boujardi and P. stocki ). Paralithoxus jariensis and P. raso came out as sister group of P. planquettei , also from Eastern Guiana (Oyapock River).
Osteological remarks. Armbruster et al. (2018) presented a morphology-based phylogeny for Lithoxini in which synapomorphies were proposed for the genera Lithoxus , Paralithoxus and Avalithoxus as a way to compare their results with the molecular phylogeny presented in Lujan et al. (2018). Most of the synapomorphies listed for Paralithoxus were confirmed in this study with slight variation in P. mocidade , such as: first hypobranchial angled at 45° relative to the sagittal plane, maxilla distal end enlarged, and metapterygoid weakly sutured to lateral ethmoid.
Although cited as a synapomorphy for the tribe Lithoxini ( Armbruster et al., 2018), in P. mocidade the difference in size between the teeth on both mandibles and the distinct shape of the cusps are remarkable ( Fig. 4 View Fig ): cusps truncate and subequal in size on the premaxilla, and pointed and equally forked on the dentary. As cited above, truncate teeth were also observed in L. lithoides and P. bovallii . Lithoxus lithoides , however, has the mesial cusp of premaxillary teeth almost three times the size of the lateral cusp. Also, the size and enlargement of the distal end of the maxilla deserves attention. In P. mocidade , the maxilla is well developed, its length more than 50% the length of the palatine, and its distal enlargement is rather pronounced.
The suspensorium in P. mocidade is similar to that of other Lithoxini and comparable to P. bovallii , the closest species to P. mocidade . However, the metapterygoid anterior processes, both the digitiform and the “spoonshaped” ( Armbruster, 2004), are much more enlarged in P. mocidade than in P. bovallii and other Lithoxini. The anterior opercular process is also highly enlarged, similar to Avalithoxus jantjae ( Armbruster et al., 2018, fig. 4-A).
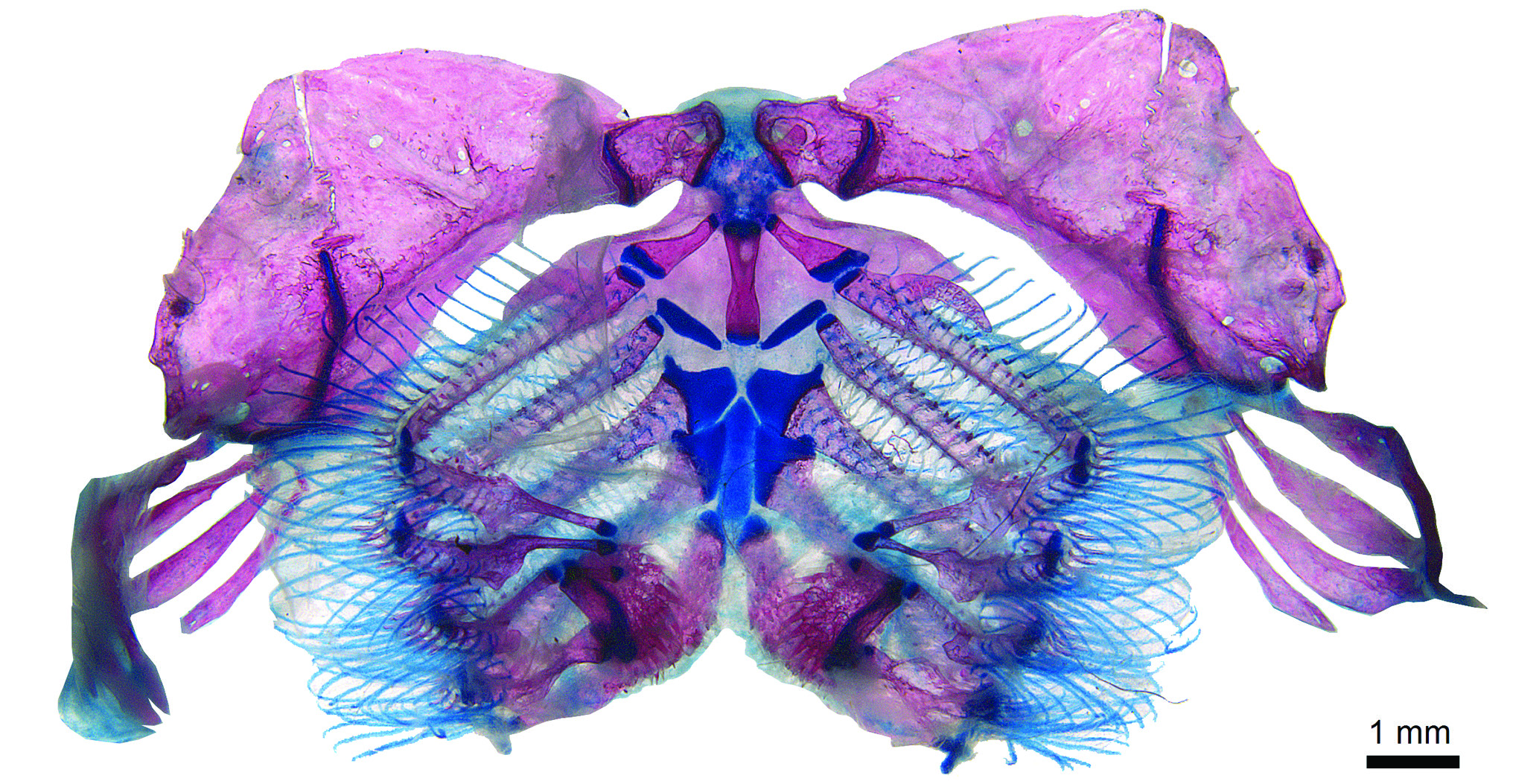
The branchial apparatus in P. mocidade has a strong hyoid arch, and strongly pointed, villiform teeth on both pharyngeal plates (upper and lower), a condition already registered by Armbruster (2004) for L. lithoides and P. bovallii . Paralithoxus mocidade has only one basibranchial (BB) ( Fig. 8 View Fig ), ossified and very elongate, almost filling the whole mesial extension of the branchial apparatus; the other basibranchial is a cartilaginous mass connected to the fourth and fifth ceratobranchials. The ordinary siluriform condition is the absence of the first basibranchial ( Arratia, 2003), second (BB2) ossified, and third (BB3) reduced, ossified or not ( Schaefer, 2003). Most loricariids show the generalized siluriform condition. In Paralithoxus mocidade , the long ossified BB can be explained by the fusion of BB2 and BB3 (very elongate BB) or BB2 lost ( Armbruster, 2004 – for L. lithoides and P. bovallii ). This condition was also observed in Exastilithoxus fimbriatus and other members of Lithoxini ( Armbruster et al., 2018).
Paralithoxus mocidade has a highly reduced accessory flange of the first ceratobranchial (CB1) ( Fig. 8 View Fig ) (approximately 1/5 of the ceratobranchial properly). Most Hypostominae have a well-developed CB1 accessory flange ( Schaefer, 1987; Armbruster, 2004), an extra sheet of thin bone originating on the same point of the ceratobranchial, of similar size and, usually, bearing a row of branchial filaments. Lithogenes and astroblepids lack this accessory flange. The condition in P. mocidade has been already cited for P. bovalli , L. lithoides , and some other non-hypostomines ( Armbruster, 2004) and it was also observed in Exastilithoxus fimbriatus .
Finally, the pectoral girdle in P. mocidade has a conspicuous small hornlike process ( Fig. 9a View Fig ) on the anterior surface of the cleithrum. Lithoxus lithoides and P. bovallii have a short and dense bony crest at the same point, but in P. mocidade this crest comes forward as a small, blunt, cylindrical process, inclined towards the lateral sides of the body, with an extra blue-stained tissue coming off its extremity that could be a tendon or a thin cartilage. This condition is quite unusual, and it was not observed in E. fimbriatus , but it has been observed in one specimen of Hypostomus gymnorhynchus .
Tab. 1. Morphometrics, meristics and ratios of Paralithoxus mocidade.
| Holotype | Mean (N=7) | SD | Min | Max | |
|---|---|---|---|---|---|
| Standard length | 50.49 | 38.35 | 55.2 | ||
| Percents in standard length | |||||
| Predorsal length | 44.6 | 44.9 | 2.0 | 42.0 | 48.3 |
| Head length | 34.5 | 34.1 | 1.4 | 32.1 | 36.2 |
| Head depth | 14.1 | 12.1 | 1.0 | 10.8 | 14.1 |
| Head width | 30.7 | 30.0 | 1.6 | 27.3 | 32.6 |
| Body depth at dorsal fin origin | 13.9 | 11.6 | 1.2 | 10.0 | 13.9 |
| Body width at dorsal origin | 23.0 | 20.6 | 1.7 | 19.3 | 23.0 |
| Body width at anal origin | 15.3 | 12.4 | 1.6 | 10.2 | 15.3 |
| Snout length | 17.9 | 18.4 | 1.0 | 17.2 | 20.6 |
| Orbit diameter | 3.6 | 3.8 | 0.3 | 3.2 | 4.4 |
| Interorbital width | 10.4 | 10.7 | 0.5 | 10.2 | 11.7 |
| Dorsal-fin base length | 20.0 | 18.8 | 1.4 | 15.8 | 21.1 |
| Interdorsal distance | 23.6 | 22.2 | 2.2 | 18.2 | 24.8 |
| Caudal peduncle length | 25.9 | 25.7 | 1.1 | 24.5 | 27.9 |
| Caudal peduncle depth | 7.5 | 7.9 | 0.4 | 7.1 | 8.4 |
| Dorsal-fin spine length | 21.5 | 22.0 | 1.6 | 20.6 | 24.5 |
| Pectoral-fin spine length | 27.9 | 25.5 | 1.9 | 23.8 | 28.7 |
| Pelvic-fin spine length | 27.0 | 27.8 | 1.8 | 25.5 | 30.5 |
| Anal-fin first ray length | 11.5 | 9.8 | 1.8 | 7.6 | 13.6 |
| Adipose-fin spine length | 7.3 | 6.8 | 1.1 | 4.9 | 8.1 |
| Lower lip width | 25.2 | 25.3 | 1.3 | 23.5 | 27.4 |
| Lower lip length | 10.2 | 10.3 | 0.7 | 9.5 | 11.4 |
| Dentary length | 2.6 | 2.6 | 0.3 | 2.1 | 3.2 |
| Premaxillary length | 2.7 | 2.6 | 0.4 | 2.2 | 3.2 |
| Percents in head length | |||||
| Head depth | 40.8 | 35.6 | 3.1 | 30.1 | 40.8 |
| Head width | 89.1 | 88.0 | 4.2 | 80.4 | 96.1 |
| Interorbital | 30.3 | 31.4 | 1.4 | 28.8 | 33.1 |
| Orbit diameter | 10.3 | 11.1 | 1.0 | 9.2 | 12.2 |
| Pectoral length | 80.9 | 74.4 | 5.6 | 66.9 | 84.9 |
| Pelvic length | 78.4 | 81.4 | 5.6 | 74.8 | 93.6 |
| Snout length | 52.1 | 53.8 | 1.8 | 51.6 | 56.8 |
| Premaxillary length | 7.8 | 7.7 | 1.1 | 6.1 | 9.9 |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |