Scinax strigilatus ( Spix, 1824 )
|
publication ID |
https://doi.org/ 10.5281/zenodo.175995 |
|
DOI |
https://doi.org/10.5281/zenodo.5663807 |
|
persistent identifier |
https://treatment.plazi.org/id/AB71878B-FFAC-FF8C-FF56-AFF9FA11FB97 |
|
treatment provided by |
Plazi |
|
scientific name |
Scinax strigilatus ( Spix, 1824 ) |
| status |
|
Scinax strigilatus ( Spix, 1824) View in CoL
( Figs. 2–4 View FIGURE 2 View FIGURE 3 View FIGURE 4 )
Hyla strigilata Spix, 1824 View in CoL .
Hyla strigillata - Werner, 1898 “1897”. Unjustified emendation or subsequent misspelling. Ololygon strigilata - Fouquette and Delahoussaye, 1977. First combination with Ololygon . Scinax strigilata View in CoL - Duellman and Wiens, 1992. First combination with Scinax View in CoL . Scinax strigilatus View in CoL - Köhler and Böhme, 1996. Change to masculine gender.
Neotype. MNRJ 38098, adult male, from Fazenda Pedra Formosa (13o57’S, 39o27’W), Municipality of Ibirapitanga, State of Bahia, Brazil, collected by B.V.S. Pimenta, R.T. Moura, and R.V. Lopes, between 27–30 May 2001.
Referred specimens. MNRJ 38091–38097, five adult males and two adult females, collected with the neotype; MNRJ 38099, adult male, from Fazenda Taquara (15o58’S, 39o22’W), Municipality of Belmonte, State of Bahia, Brazil, collected by B.V.S. Pimenta and P.H.C. Cordeiro, on 19 May 2001; MNRJ 38100, adult male, from Fazenda São João (13o42’S, 39o14’W), Municipality of Nilo Peçanha, State of Bahia, Brazil, collected by B.V.S. Pimenta and R.V. Lopes, on 0 6 September 2000; MNRJ 38101–38103, three adult females, from Fazenda Palmeira (15o56’S, 39o38’W), Municipality of Itapebi, State of Bahia, Brazil, collected by D.L. Silvano and B.V.S. Pimenta, on 19 and 22 January 2001; MNRJ 38980, adult male, form Reserva Particular do Patrimônio Natural Serra do Teimoso (15o09’S, 39o31’W), Municipality of Jussari, State of Bahia, Brazil, collected by B.V.S. Pimenta and P.H.C. Cordeiro, on April 2000.
Diagnosis. A species belonging to the Scinax catharinae group, characterized by: (1) medium size (males 23.8–28.6 mm SVL, females 36.9–38.3 mm SVL); (2) rounded snout in dorsal view; (3) well-marked canthus rostralis; (4) vomerine teeth in two transverse series; (5) inguinal gland not thickened; (6) greenish coloration on concealed parts of flanks and thighs.
Scinax strigilatus has the adult external morphological synapomorphy of the S. catharinae clade identified by Faivovich (2002): the “internal” vocal sac. It also has the muscular synapomorphies of the clade that can be observed with a superficial dissection (distal division of the middle branch of the m. extensor digitorum comunis longus, and insertion of the medial side of this branch on the tendon of the m. extensor brevis medius digiti IV). Scinax strigilata also lacks a pectoral fold.
Comparison with all species of the Scinax catharinae group. Scinax strigilatus differs from S. agilis , S. argyreornatus , S. berthae , S. centralis , S. machadoi , and S. ranki by its larger size (males combined SVL 13.5–23.3 mm; females combined SVL 19.5–28.7 mm in these species), and from S. catharinae by its smaller size (males SVL ~33.0 mm; females SVL 41.0–45.0 mm in S. catharinae ). It is distinguished from S. angrensis , S. carnevalli , S. humilis , S. jureia , and S. luizotavioi by its larger females (females combined SVL 27.5– 34.0 in these species), but males are smaller than in S. ariadne , S. brieni , and S. jureia (males combined SVL 29.0–35.0 mm in these species), and females smaller than in S. flavoguttatus (females SVL 40.0– 43.1 in S. flavoguttatus ). Scinax strigilatus differs from S. agilis , S. angrensis , S. argyreornatus , S. berthae , S. canastrensis , S. carnevalli , S. centralis , S. flavoguttatus , S. heyeri , S. kautskyi , S. longilineus , S. luizotavioi , S. machadoi , and S. rizibilis by its rounded snout in dorsal view (mucronate in S. kautskyi and S. littoralis ; subelliptical in S. angrensis , S. argyreornatus , S. berthae , S. canastrensis , S. centralis , S. longilineus , and S. luizotavioi ; subovoid in S. flavoguttatus , S. heyeri , and S. rizibilis ; truncate in S. agilis , S. carnevalli , and S. machadoi ). Due to its well marked canthus rostralis S. strigilatus is distinguished from S. brieni , S. catharinae , S. heyeri , S. jureia , S. machadoi , S. obtriangulatus , S. ranki , and S. trapicheiroi (canthus rostralis poorly marked in these species). It differs from S. agilis , S. angrensis , S. argyreornatus , S. ariadne , S. canastrensis , S. humilis , S. kautskyi , S. littoralis , and S. longilineus by presenting vomerine teeth in two transverse series (oblique in S. agilis , S. angrensis , S. argyreornatus , S. kautskyi , S. littoralis , and S. longilineus ; convex in S. ariadne , S. canastrensis , and S. humilis ). By the absence of an enlarged nuptial pad, S. strigilatus is distinguished from S. rizibilis (enlarged nuptial pad present). It is distinguished from S. angrensis and S. luizotavioi by the presence of supernumerary tubercles on feet (tubercles absent in these species), and from S. agilis , S. carnevalli , S. kautskyi , S. longilineus , and S. luizotavioi due to the scarcity and small size of tubercles on outer margins of forearm and tarsus (weakly crenulate in S. agilis , S. carnevalli , and S. longilineus ; crenulate in S. luizotavioi ; forearms tuberculated in S. canastrensis and S. kautskyi ; tarsus moderately tuberculated in S. agilis and S. machadoi and densely tuberculated in S. albicans and S. kautskyi ). Scinax strigilatus differs from S. luizotavioi due to its narrow toes (robust in S. luizotavioi ). Scinax centralis and S. hiemalis differ for having the inguinal glands particularly thick and enlarged. The greenish coloration on hidden areas of flanks and thighs differentiate S. strigilatus from S. agilis , S. aromothyella , S. berthae , S. canastrensis , S. centralis , S. flavoguttatus , S. heyeri , S. humilis , S. longilineus , S. machadoi , S. obtriangulatus , and S. trapicheiroi (orange or yellow spots in S. aromothyella , S. berthae , S. canastrensis , S. centralis , S. flavoguttatus , S. heyeri , S. longilineus , and S. machadoi ; blue coloration in S. humilis and S. trapicheiroi ; grayish violet in S. obtriangulatus , dark brown in S. agilis ).
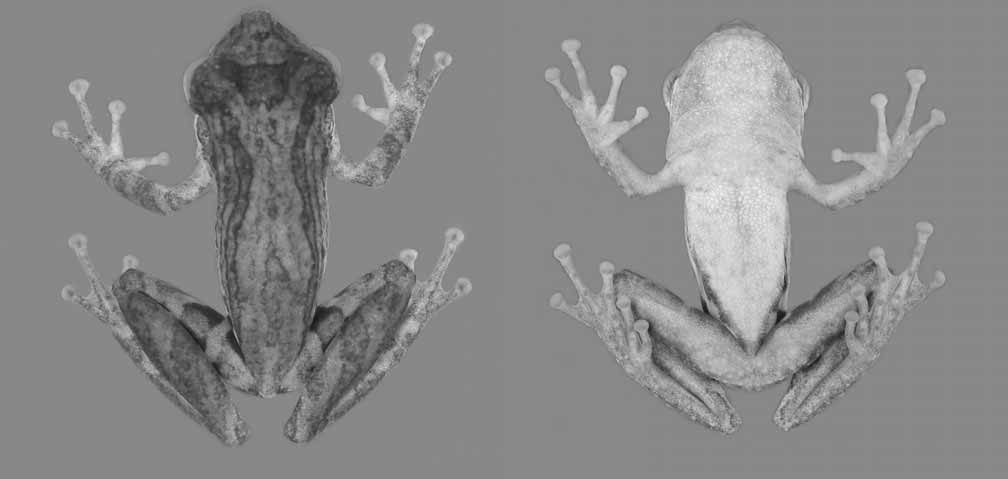
Description of the neotype. Body slender; head larger than 1/3 of the SVL, slightly wider than body; snout rounded in dorsal view, protruding in lateral view ( Fig. 3 View FIGURE 3 ); nostrils protuberant, located laterally, immediately before the tip of snout; canthus rostralis well marked, nearly straight; loreal region concave; eyes protuberant, large, its diameter almost 40% of HL; tympanum distinct, small, rounded, slightly smaller than the diameter of finger III disc; supratympanic fold weakly marked, extending from the posterior corner of eye to shoulder; vocal sac subgular, weakly expanded laterally; vocal slits present; tongue large, oval, free laterally and posteriorly, slightly notched behind; two transverse series of six vomerine teeth in between the large elliptical choanae.
Arms slender, forearms moderately more robust than arms; fingers narrow; finger lengths I<II~IV<III; subarticular tubercles nearly conical, more developed, bifid on finger IV; supernumerary tubercles abundant, small; outer metacarpal tubercle protruding, bifid; inner metacarpal tubercle weakly developed, nearly elliptical; fingers discs well developed, nearly elliptical, wider than large; disc of finger I smaller than the others; vestigial webbing between fingers II and III, and III and IV ( Fig. 3 View FIGURE 3 ); nuptial pad on the inner margin of thumb, barely noticeable.
Legs slender; tibia longer than thigh; sum of thigh and tibia lengths practically equal SVL; toes narrow; toe length I<II<III~V<IV; subarticular tubercles rounded, protruding; outer metatarsal tubercle rounded, much smaller than subarticular tubercles; inner metatarsal tubercle small, elongated; toe discs well developed, approximately elliptical, wider than large; supernumerary tubercles abundant, small; discs of toes II and III smaller than the others; webbing formula II 2 - – 3- III 1 % – 3- IV 3 - - 1½ V ( Fig. 3 View FIGURE 3 ). Pectoral fold absent. Dorsal surfaces and flanks smooth with scattered granules, mainly on sacral region and head, where they are larger; a tubercle between eye and nostril, on canthus rostralis; gular region and chest finely granular; belly and ventral surfaces of thighs aureolate; inguinal glands not thick or enlarged. Measurements (mm): SVL 27.1; HL 10.7; HW 9.3; ED 4.2; IOD 3.3; END 3.3; IND 0.5; TD 1.6; TL 12.4; TBL 14.4; FL 18.7.
Color in life. General pattern light brown, with a dark brown stripe from nostril to the anterior corner of eye; a white stripe from eye to maxilla and another one from eye to the shoulder; a dark brown, ‘W’-shaped mark between the eyes; a pair of wide dark brown stripes from the upper eyelids to the inguinal region, and a pair of light brown stripes from the posterior corner of the eye, covering the tympanum, and extending to the inguinal region; a narrow, dark brown line between them; granules on dorsum and flanks light brown; scattered lines and blotches on dorsum midbody and sacral region; flanks with white background. Arms with transverse light brown bars emarginated by dark brown lines over a light brown background; hands with many dark brown dots. Thighs and tibiae with four dark brown transverse bars on the anterior surface and dorsum over a light brown background; posterior surfaces of thigh almost completely dark brown. Tarsus and feet with many dark brown dots over a light brown background. Concealed surfaces of flanks and thighs greenish. Ventral surfaces white with scattered, dark brown dots on gular region and forelimbs, densely dotted on legs.
Variation. Some measurements are shown in Table 1 View TABLE 1 . Females are much larger than males, and their snouts much less protruding, almost vertical. The number of vomerine teeth varies from five to eight among specimens analyzed, also varying between vomerine ridges of the same specimen. Some specimens show larger supernumerary tubercles on hands and feet, fingers I and II also webbed at base, discs of toes II and III as developed as the others, and webbing formula varying as II 1 % – 3- III 1 % – 2½ IV 2 ½ – 1½ V or II 2 – 3 + III 1 % – 3+ IV 3 - – 1% V. Colors in preservative may be more or less faded; gular region, chest, and belly of most specimens is dotted and/or marbled of dark brown over cream background.
Distribution. Currently known from five locations within the Atlantic Rain Forest Domain in the southern region of the State of Bahia, Brazil ( Fig. 5 View FIGURE 5 ). The Fazenda Pedra Formosa, locality where the neotype was collected, is only 35 km airline away from the Municipality of Camamu, one of the three localities visited by Spix in the Atlantic Rainforest Domain of the State of Bahia (for the detailed itinerary of Spix and Martius in Brazil, see Vanzolini 1981).
Natural history. Most specimens of Scinax strigilatus were captured on the marginal vegetation of streams inside secondary or undisturbed forest patches. Some were caught near temporary ponds near streams formed after flooding, and MNRJ 38099 was found on the marginal vegetation of a small temporary pond in a road inside a disturbed forest fragment, far from streams. Males call perched on the vegetation, 90-150 cm above the ground.
Males Females Remarks. While Scinax strigilatus has been computed on previous species counts of Scinax ( Duellman & Wiens 1992; Faivovich et al. 2005; Wiens et al. 2005), or even of the S. catharinae group (Faivovich et al. 2005), it has been done exclusively on the basis of previous literature records, with the knowledge that there were problems about its status, effectively turning it into a sort of “ghost” species. Moreover, no taxonomist working with the S. catharinae group during the last 25 years has ever tried to differentiate any species of this group from S. strigilatus (e.g., Peixoto & Weygoldt 1987; Caramaschi & Kisteumacher 1988; Pombal and Bastos 1996; Faivovich 2005).
Reasons for the association of the name Hyla strigilata Spix with the new material from southern Bahia were discussed earlier in this paper. Considering the chaos that surrounded this name in the past, also reviewed here, naming a new species for these specimens is an alternative that had been considered during this project. However, we maintain that the designation of a neotype for Hyla strigilata puts a definitive end to all the problems of the past, and eliminates all ambiguities associated with the available name Ololygon Fitzinger, 1843 , whose type species is Hyla strigilata . We consider these two reasons valuable, as the Scinax catharinae clade is quite difficult to work with by itself. Any reasonable solution to its outstanding nomenclatural problems will allow researchers to focus on the many taxonomic and biological problems of the group, without the burden of past confusions. The designation of a neotype for Hyla strigilata Spix, 1824 , and the consequent association of this name with known populations, turns Scinax strigilatus into a full, valid species of Scinax .
Note added in proof: During recent field work at the Municipality of Arataca, Bahia, near RPPN Serra do Teimoso, additional specimens of Scinax strigilatus were collected, among which there is a female with a snout-vent length of 45.6 mm (MNRJ 44988). A notable sexual dimorphism in size is common in the S. catharinae group (compare with a snout-vent length of 28.6 mm in the largest male reported in this paper); this large female eliminates the most significant difference found between the populations to which we are applying the name S. strigilatus and the description provided by Peters (1872a) of the holotype of Hyla strigilata .
TABLE 1. Mean, standard deviation (SD), and range of some measurements from seven males and five females of Scinax strigilatus.
| Mean | SD | Range | Mean | SD | Range |
|---|---|---|---|---|---|
| SVL 26.6 | 1.91 | 23.8–28.6 | 37.3 | 0.57 | 36.9–38.3 |
| HL 10.0 | 0.71 | 9.2–11.1 | 13.6 | 0.50 | 13.2–14.3 |
| HW 8.8 | 0.75 | 7.9–10.1 | 12.5 | 0.29 | 12.2–12.9 |
| ED 4.0 | 0.28 | 3.5–4.2 | 5.2 | 0.31 | 4.8–5.6 |
| IOD 3.2 | 0.46 | 2.7–3.9 | 5.4 | 0.51 | 4.6–5.9 |
| END 2.8 | 0.31 | 2.5–3.3 | 4.1 | 0.47 | 3.5–4.8 |
| IND 0.5 | 0.12 | 0.4–0.8 | 0.8 | 0.07 | 0.8–0.9 |
| TD 1.5 | 0.18 | 1.2–1.7 | 1.6 | 0.27 | 1.3–2.0 |
| THL 13.8 | 1.15 | 12.4–15.4 | 19.4 | 0.71 | 18.6–20.4 |
| TBL 14.9 | 0.96 | 14.0–16.7 | 20.6 | 0.71 | 19.9–21.7 |
| FL 19.1 | 1.23 | 17.1–20.6 | 26.3 | 1.16 | 25.4–28.3 |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.