Alpheus malleator Dana, 1852
|
publication ID |
https://doi.org/ 10.11646/zootaxa.3637.4.2 |
|
publication LSID |
lsid:zoobank.org:pub:54055A0B-A1CD-4C7D-AAA9-F46AFD0B1D5F |
|
DOI |
https://doi.org/10.5281/zenodo.6163793 |
|
persistent identifier |
https://treatment.plazi.org/id/BB778E34-FFAB-FFA0-2CD0-D527FBF0FBE7 |
|
treatment provided by |
Plazi |
|
scientific name |
Alpheus malleator Dana, 1852 |
| status |
|
Alpheus malleator Dana, 1852 View in CoL
( Figs. 1–6 View FIGURE 1 View FIGURE 2 View FIGURE 3 View FIGURE 4 View FIGURE 5 View FIGURE 6 , 11 View FIGURE 11 A–H)
Alpheus malleator Dana 1852a: 23 ; Dana 1852b: 557; Dana 1855: 23, pl. 31, fig. 9a-h; Kingsley 1883: 112; Sharp 1893: 113; Coutière 1899 (see Chace & Forest 1970 for all page numbers); Moreira 1901: 9; Luederwaldt 1919: 430; Oliveira 1940: 142; Holthuis 1951: 73; Sourie 1954: 112, 253; Crosnier & Forest 1965: 605; Crosnier & Forest 1966: 240, fig. 10; Evans 1967: 408; Chace 1972: 68; Coelho & Ramos 1972: 148; Abele 1976: 273; Christoffersen 1980a: 74; Christoffersen 1980b: 39; Abele & Kim 1986: 197, 206-207, figs. i, j; Martínez-Iglesias et al. 1997: 425; Christoffersen 1998: 359; McClure 2005: 145 (partim, not fig. 16); Hurt et al. 2008: 516 et seq.; De Grave & Fransen 2011: 386.
Crangon malleator — Schmitt 1924a: 64, fig. 8; Schmitt 1936: 367; Schmitt 1939: 24.
Alpheus pugilator A. Milne-Edwards 1878: 229 .
(?) Alpheus tuberculosus Osorio 1892: 201 ; Osorio 1898: 194; Rathbun 1900: 313; Balss 1914: 98, figs. 1-5; Balss 1916: 21; Vilela 1949: 56; Holthuis 1951: 73; Crosnier & Forest 1965: 605; Crosnier & Forest 1966: 242.
(?) Alpheus Belli Coutière, 1898: 149 , fig. 1.
(?) Alpheus belli — Coutière 1899 (see Chace & Forest 1970 for all page numbers); Chace 1972: 63; Pequegnat & Ray 1974: 246; Christoffersen 1998: 357.
(?) Alpheus malleator var. edentatus Zimmer 1913: 387 , figs. G-M; Balss 1916: 22; Schmitt 1924b: 71.
Not Alpheus malleator — Nobili 1901: 2; Rathbun 1902: 288; Sivertsen 1933: 4; Abele 1975: 72; Rodríguez de la Cruz 1977: 28; Brusca 1980: 252; Wicksten 1983: 43; Banner & Banner 1984: 42; Hendrickx & Wicksten 1987: 16; Kim & Abele 1988: 31, fig. 12; Wicksten & Hendrickx 1992: 5; Lemaitre & Alvarez-Leon 1992: 42; Wicksten 1993: 152; Hickman & Zimmermann 2000: 40; Wicksten & Hendrickx 2003: 64; Hendrickx & Hermoso-Salazar 2005: 433, fig. 1D (map); Lazarus-Agudelo & Cantera-Kintz 2007: 228 (= Alpheus wonkimi sp. nov., see below).
Not Crangon malleator — Rathbun 1910: 607 (= Alpheus wonkimi sp. nov., see below).
Material examined. Brazil: 1 male (cl 16.1 mm), MZUSP 0 0 138, São Paulo, Ilha da Vitória (northeast of Ilhabela), leg. Gunther, 1906 [det. M.J. Rathbun, 1915]; 2 males (cl 9.5, 11.3 mm), MZUSP 22296, São Paulo, Ubatuba, Praia do Lázaro, leg. E.J. de Paula, 13 July 1976; 2 males (cl 10.6, 15.4 mm), MZUSP 13094, São Paulo, Ubatuba, north of Praia Grande, leg. M.A. Pinheiro, 18 October 1997; 1 male (cl 9.9 mm), MZUSP 28270, São Paulo, Ilha da Vitória (northeast of Ilhabela), leg. V. Cobo, December 2006. Panama: 1 male (cl 11.1 mm), OUMNH.ZC. 2012-07-0146, Caribbean coast, Isla Grande, intertidal basaltic rocks exposed to high-energy waves, leg. N. Knowlton et al., 6 June 1995 [fcn B-424]; 1 male (cl 8.3 mm), USNM, same collection data as for the previous specimen [fcn B-423]. Puerto Rico: 1 male (cl 10.8 mm), 2 ov. females (cl 10.5, 10.8 mm), MNHN-IU- 2010-4199, locality not specified, leg. P. Serre, 1907; 1 male (cl 12.4 mm), 1 ov. female (cl 14.0 mm), MNHN-IU- 2010-4194, locality not specified, bases of hard corals, leg. P. Serre, 1907. Trinidad & Tobago: 1 ov. female (cl 7.9 mm), OUMNH.ZC. 2010-01-0071, Tobago, locality not specified, collector and year unknown. Cape Verde: 1 male (cl 7.5 mm), 1 female (cl 6.0 mm), USNM, Santiago, Cidade Velha, leg. N. Knowlton et al., 3 April 1998. Senegal: 1 male (cl 7.2 mm), MNHN-Na 3109, north of Dakar, C24, leg. Postel, 1949. Guinea: 1 male (cl 13.0 mm), 2 ov. females (cl 11.0 mm, 12.1 mm), MNHN-IU-2010-4192, Tamara, Îles de Los (Loos Islands), west coast, low tide, leg. M. Serrand, 6 July 1913; 1 male (cl 11.8 mm), MNHN-IU-2010-4193, same collection data as for the previous specimens. Equatorial Guinea: 4 males, 4 females (cl 3.7–12.3 mm, male largest), MNHN-IU-2010-4196, Annobon, Punta del Palmar, intertidal, 29 July 1965, leg. A. Crosnier; 5 males, 1 ov. female, 4 females (cl 5.0– 12.6 mm, ov. female largest), MNHN-IU-2010-4198, Annobon, near San Pedro, intertidal, 30 July 1965, leg. A. Crosnier. Congo: 1 male (cl 12.8 mm), MNHN-IU-2010-4197, Djeno Rocks near Pointe Noire, intertidal, small tide pools among rocky outcrops, 4 April 1965, leg. A. Crosnier.
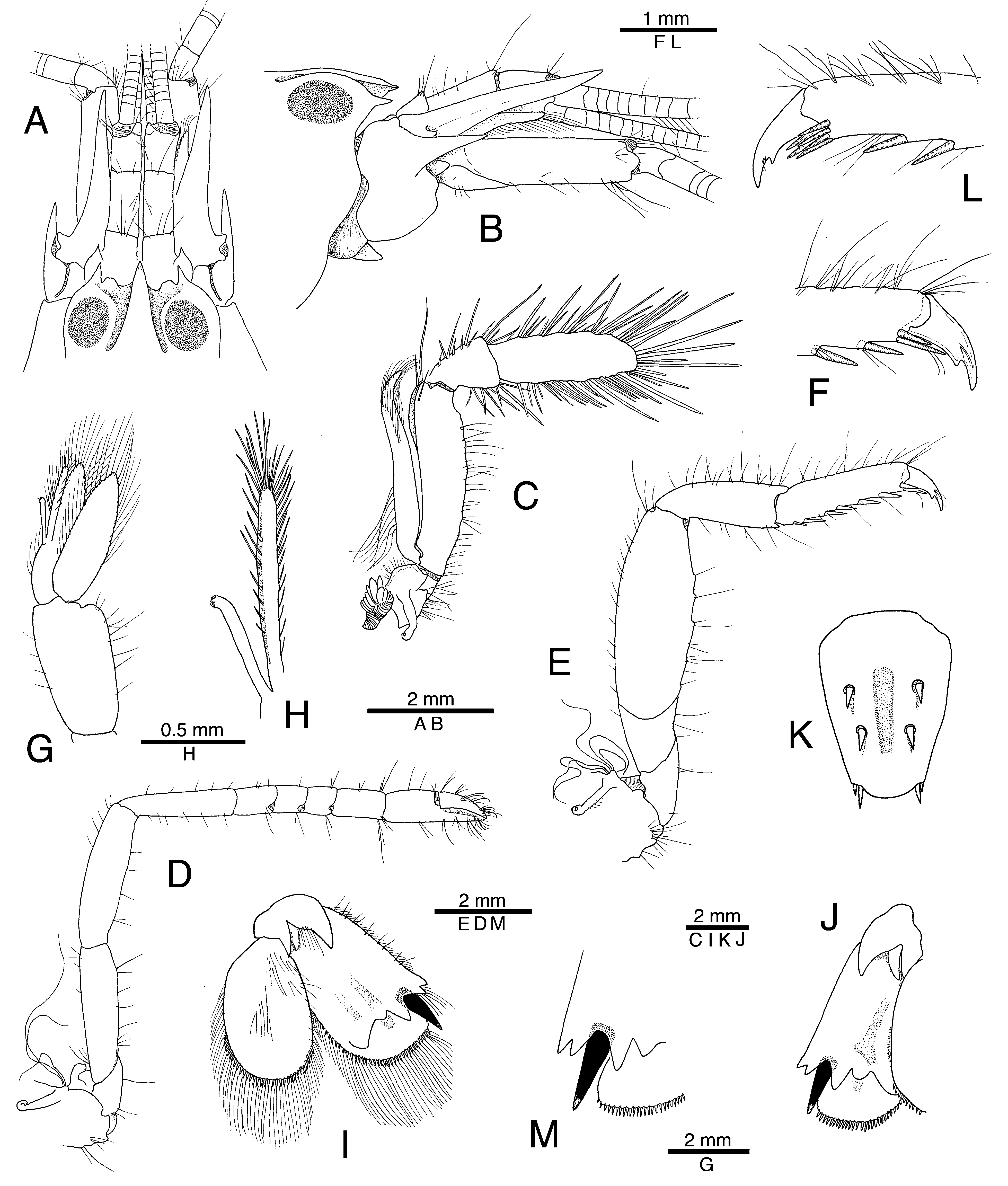
Description. Body stout, moderately large (maximum carapace length 16.1 mm, maximum total length about 50 mm). Rostrum short to moderately long, typically not reaching mid-length of first article of antennular peduncle, dorsally flattened, more or less widening posteriorly; lateral margins abruptly delimited by deep adrostral furrows; tip subacute or blunt, often descendent ( Figs. 1 View FIGURE 1 A, B, 3A, E). Orbital hoods swollen, with acute or subacute teeth; anterior margin between each orbital tooth and rostrum with variously developed additional (= adrostral) tooth ( Figs. 1 View FIGURE 1 A, B, 3A, E). Post-rostral area without median tubercle; pterygostomial angle rounded; cardiac notch well developed.
Abdomen without specific features on sternum. Telson broad, stout, tapering posteriorly, slightly depressed along mid-dorsal line, with two stout dorsal spiniform setae and two pairs of posterolateral spiniform setae, mesial much longer than lateral ( Figs. 1 View FIGURE 1 K, 3J); anal tubercles well developed.
Antennular peduncles moderately stout, with second article longest, about 1.8 times as long as wide; stylocerite short, almost reaching distal margin of first article, with acute tip ( Figs. 1 View FIGURE 1 A, 3A, E). Antenna with enlarged, stout basicerite bearing strong, sharp distolateral tooth reaching to or slightly beyond mid-length of second antennular article; scaphocerite large, stout; lateral margin shallowly concave, proximally with distinctly projecting, blunt tooth; blade narrow separated from strong distolateral tooth by deep cleft; distolateral tooth reaching well beyond distal end of antennular peduncle, almost reaching distal margin of carpocerite ( Figs. 1 View FIGURE 1 A, B, 3A, E).
Third maxilliped stout; coxa with blunt lateral plate; antepenultimate article not operculate, somewhat compressed and twisted, with numerous slender setae; penultimate and ultimate articles strongly compressed, very setose; penultimate article short, distally widening; ultimate article about three times as long as wide, broad, densely covered with long, very thick setae (Fig, 1C).
Major cheliped very stout; ischium very short; merus subtriangular in cross-section, stout; carpus short, cupshaped; chela broadly ovoid; lateral surface with longitudinal groove running from about mid-length of palm to end of pollex and a low crest delimiting shallow groove running obliquely from linea impressa to distodorsal third of palm; ventrolateral and dorsolateral surfaces with shallow, rounded bumps; distolateral margin of palm with two rounded bumps; mesial surface more or less rugose, with numerous setose tubercles, row of much larger tubercles present close to dorsal margin; distomesial margin of palm with very large, bluntly projecting, dorsal bump and two smaller, blunt or subacute bumps closer to propodo-dactylar articulation; pollex much shorter than dactylus, distally truncate; dactylus hammer-shaped, with short, truncate plunger, latter furnished with stamen-shaped sensillae ( Figs. 2 View FIGURE 2 A–D, 4B, C, F, G, 5A–D).
Minor cheliped stout; ischium short; merus subtriangular in cross-section, stout; carpus short, cup-shaped; chela with palm and fingers subequal in length; lateral and mesial surfaces of palm covered with groups setae, many arising from small bumps, ventral surface somewhat rugose, dorsal margin with larger, rounded bumps; distomesial margin with large, projecting, subacute tooth; fingers moderately curved, stout, setose, with sharp cutting edges, pollex somewhat excavated laterally, finger tips crossing ( Figs. 2 View FIGURE 2 E–G, 4D, E).
Second pereiopod with ischium and merus subequal in length; first carpal article shorter than merus, about the same length as sum of second, third and fourth articles; chela comparatively large, simple ( Figs. 1 View FIGURE 1 D, 3B, F). Third and fourth pereiopods generally similar, stout; ischium unarmed; merus somewhat swollen, length about three times greatest width, distoventral margin unarmed; carpus with small distoventral tooth; propodus with six strong spiniform setae along ventral margin and two (occasionally more) spiniform setae adjacent to dactylus; dactylus stout, strongly curved, biunguiculate ( Figs. 1 View FIGURE 1 E, F, L, 3C, G). Fifth pereiopod shorter and much more slender than third and fourth ( Fig. 6 View FIGURE 6 A); propodal brush well-developed.
Second male pleopod with appendix masculina densely setose, about twice as long as appendix interna, almost reaching distal margin of endopod ( Figs. 1 View FIGURE 1 G, H, 3H). Uropod with large subacute lobe on protopod; distolateral spiniform seta black in both sexes, stout, long, reaching to or slightly beyond distal margin of exopod, flanked by large mesial tooth and smaller lateral tooth, latter similar in size and shape to distolateral tooth of exopod; distal margin of exopod and endopod with row of short, stout, spiniform setae ( Figs. 1 View FIGURE 1 I, J, M, 3D, I).
For additional illustrations see Dana (1855), Coutière (1899), Zimmer (1913), Crosnier & Forest (1966).
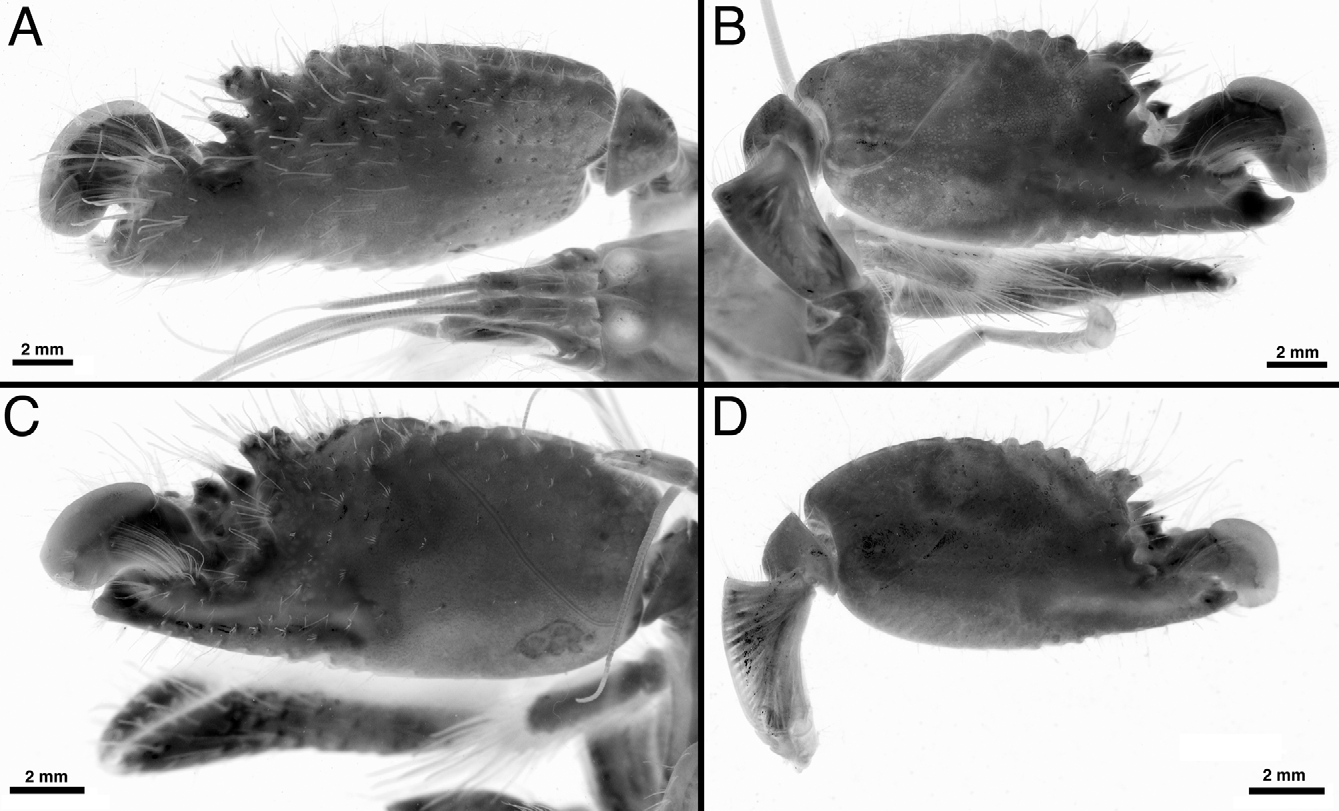
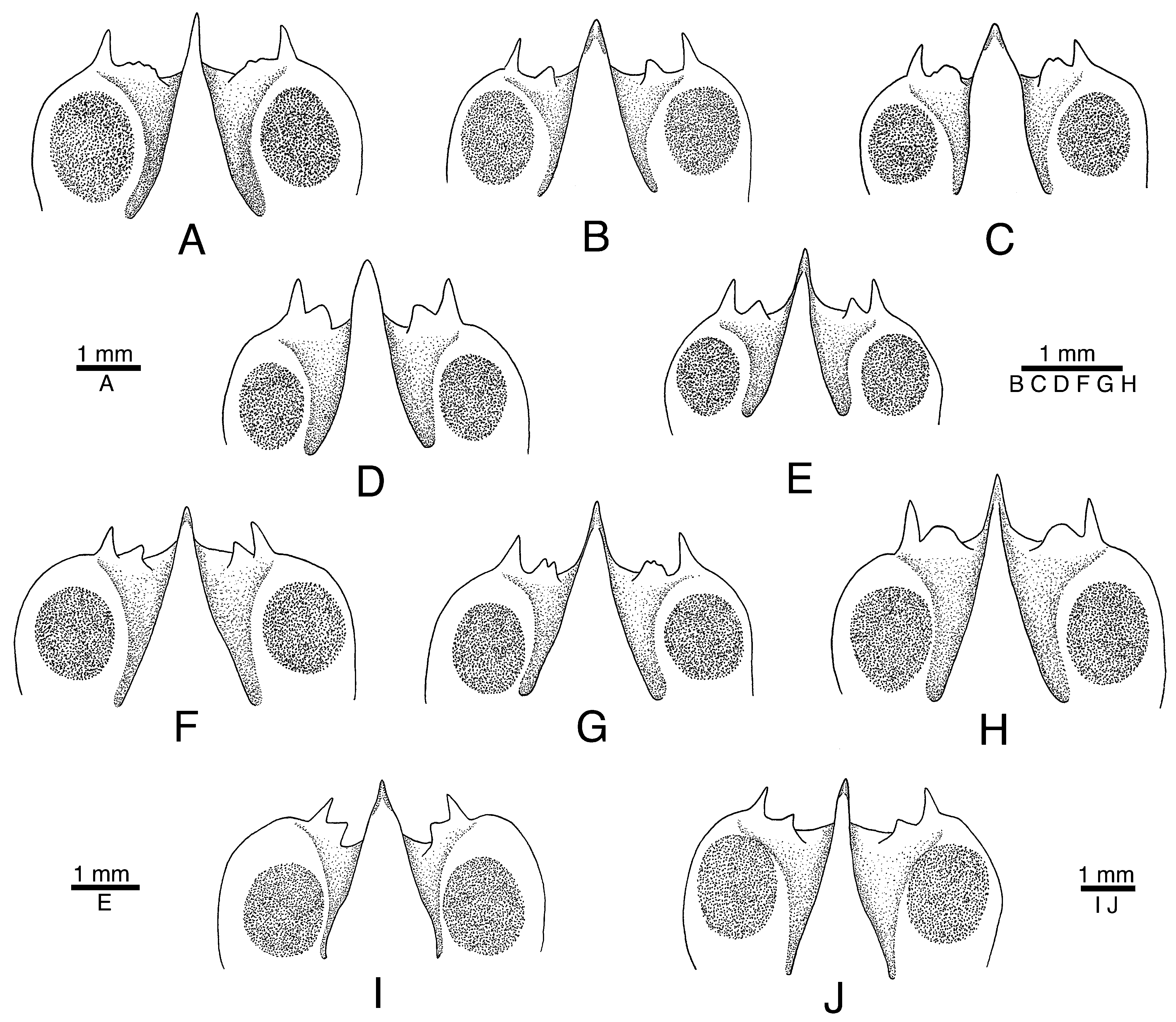
Variation. Alpheus malleator is remarkably variable in the configuration of the frontal margin of the carapace, especially in the width and shape of the post-rostral plate and the size (large, small, reduced to barely noticeable bumps), shape (rounded, subacute, with several distal points), direction (anterior, anteromesial) and position (marginal, submarginal) of adrostral teeth between the rostrum and orbital teeth ( Figs. 1 View FIGURE 1 A, 3A, 4A, E, 11A–H). This variation was first reported in West African specimens by Crosnier & Forest (1966: fig. 10, a, g), but is also marked in the Caribbean and Brazilian populations ( Fig. 11 View FIGURE 11 A–H). The chelipeds, especially the major cheliped, also show some degree of variation. Adult males have larger chelae, stronger major chelae, with more pronounced distal bumps on the palm ( Figs. 4 View FIGURE 4 B, C, 5A, B). In females and younger males, the major chelae are smaller in size, shorter, more rounded, with shorter distal bumps on the palm ( Figs. 4 View FIGURE 4 F, G, 5C, D; see also 6B, D). The ratio of the first carpal article of the second pereiopods is also somewhat variable ( Fig. 3 View FIGURE 3 B, F; see also Crosnier & Forest 1966: fig. 10, e, i). The thickness of the spiniform seta on the uropodal exopod is also somewhat variable, being generally wider in males ( Figs. 1 View FIGURE 1 I, J, M), especially in the Brazilian and West African specimens ( Fig. 3 View FIGURE 3 D, I). However, the colour of this spiniform seta appears to be black in both males and females, contrary to the statement of Abele & Kim (1986) that the “distolateral spine on uropod of exopod [is] dark-colored in male only”.
Colour pattern. Body ground colour variable from dark chocolate-brown to reddish-brown; carapace with more or less marked, whitish, transverse band and numerous irregular patches and spots, many interconnected; rostrum dark-brown; orbital hoods, flanks and anterolateral areas of carapace mostly colourless; abdomen usually with irregular, white, transverse bands on each somite, some bands broken into rhomboid-shaped patches, bands usually continuing on pleura and splitting into two large white areas (usually two per pleuron: anterior and posterior), bands sometimes very narrow or absent (especially in larger individuals); major and minor chelipeds brown to red-brown, with some paler areas mesially, some tubercles dark brown, others marked by pale or whitish spots; dactylus of major chela red-brown centrally, pinkish dorsally and distally; pollex of major chela mostly dark brown or red-brown; adhesive disks contrasting whitish; fingers of minor chela dark or red-brown; second pereiopods and walking legs red or reddish; antennular and antennal flagella pale brown to green-yellowish; tail fan brown to red-brown, without spots, diaeresis darker brown, spiniform seta of uropodal exopod black; pleopods reddish ( Fig. 6 View FIGURE 6 ).
Type locality. Rio de Janeiro? (“in portu Rio Janeiro?” in Dana 1852a).
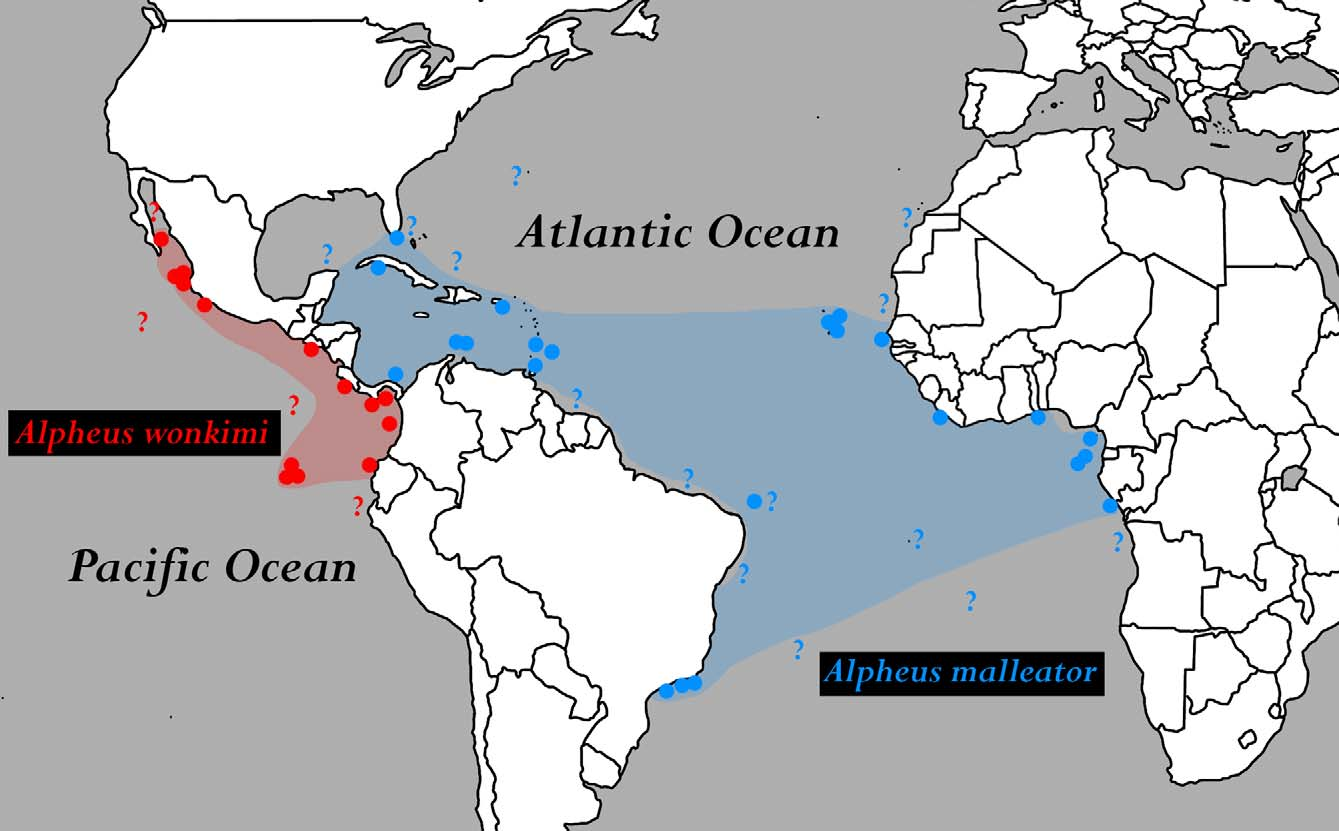
Distribution. Western and eastern Atlantic: Gulf of Mexico; Florida; Caribbean Sea ( Panama, Cuba, Puerto Rico, Trinidad & Tobago, Barbados); Brazil (Fernando de Noronha, Rio de Janeiro, São Paulo); West Africa ( Cape Verde, Senegal, Guinea, São Tomé, Annobon, Congo) (e.g., Coutière 1898, 1899; Holthuis 1951; Crosnier & Forest 1966; Chace 1972; Abele & Kim 1986; Christoffersen 1998; McClure 2005; present study). See remarks below.
Ecology. Rocky and mixed sand-rock shores and coral reef substrates (e.g., fossilised coral terraces, bases of hard corals), deep inside crevices, possibly rock-boring or at least capable of enlarging natural cavities.
Remarks. Although the geographic range of A. malleator spans almost the entire tropical western and eastern Atlantic ( Fig. 12 View FIGURE 12 ), the species appears to be uncommon or at least to have a patchy occurrence. This is certainly due to its special microhabitat requirements, partly endolithic life style and the resulting difficulties of sampling. Alpheus malleator seems to prefer exposed shores and bioeroded hard substrates, such as porous basaltic rocks, fossilised coral terraces, bases of living corals etc., usually covered with algae and with deep crevices lined with various invertebrates (sponges, ascidians). In addition, most records of A. malleator are from islands situated at some distance from the mainland (e.g., Isla Grande, Ilha Vitória, Loos Islands) or offshore islands ( Puerto Rico, Tobago, Cape Verde, São Tomé, Annobon). Thus, the beach profile, exposure and availability of suitable substrates certainly influence the distribution of A. malleator at a local scale. However, due to its cryptic habits, relative uncommonness and preference for exposed, often wave-battered rocks, A. malleator may be present in many localities where it has not yet been collected (marked with “?” in Fig. 12 View FIGURE 12 ). For instance, despite intensive sampling efforts in presumably suitable microhabitat in São Tomé in 2006, no specimens of A. malletor were found, showing that even a targeted collecting may be unsuccessful.
The presence of A. malleator in Florida requires confirmation. The only Florida record of A. malleator is based on keys in Abele & Kim (1986) who did not provide a specific locality. However, it is quite possible that A. malleator occurs in southern Florida, e.g., in the Florida Keys and/or Dry Tortugas, both offering the type of microhabitat required by this species.
The presence of A. malleator in Fernando de Noronha off northeastern Brazil is based on the tentative synonymy of A. belli . Currently, A. malleator is not known from the mainland coast of northeastern Brazil. Noteworthy, the authors’ search for A. malleator on exposed rocky shores of Ceará, Rio Grande do Norte and Alagoas in 2011–2012 did not yield any specimens. Most Brazilian records of A. malleator are from much further south, in particular from the near-continental islands of Rio de Janeiro and São Paulo. Similarly, A. malleator has not (yet) been found around oceanic islands with abundance of exposed rocky shores, such as Bermuda and Ascension Island, which are marginal to the species’ geographic range ( Fig. 12 View FIGURE 12 ).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |