Marcusenius krameri, Maake, Pholoshi A., Gon, Ofer & Swartz, Ernst R., 2014
|
publication ID |
https://doi.org/ 10.11646/zootaxa.3780.3.2 |
|
publication LSID |
lsid:zoobank.org:pub:AFB77705-1519-413D-96CD-25EDE68A6C73 |
|
DOI |
https://doi.org/10.5281/zenodo.4658254 |
|
persistent identifier |
https://treatment.plazi.org/id/B573AEDB-207C-45ED-84E0-AF74B81D9A66 |
|
taxon LSID |
lsid:zoobank.org:act:B573AEDB-207C-45ED-84E0-AF74B81D9A66 |
|
treatment provided by |
Plazi |
|
scientific name |
Marcusenius krameri |
| status |
sp. nov. |
Marcusenius krameri View in CoL , sp. nov.
Figure 2 View FIGURE 2 b; Table 8 View TABLE 8
Marcusenius View in CoL Limpopo population: Kramer et al. 2007: 661.
Holotype. SAIAB 188295, 116.21 mm SL, male, Mogalakwena River (also known as the Nyl), Modimolle town, Pretoria Street, above a drift, 24°42'04.2" S, 28°24'40.8" E, Limpopo River system, South Africa, P. Maake and O. Gon, 14 October 2010. Hologenetype COI (GenBank KJ174307 View Materials ), Hologenetype cyt b (GenBank KJ174297 View Materials ).
Paratypes. SAIAB 191224, 5: 92–117 mm SL, Paragenetype COI (GenBank KJ174305 View Materials and KJ174306 View Materials ), Paragenetypes cyt b (GenBank KJ174295 View Materials and KJ174296 View Materials ), same collection data as holotype. MRAC B3-16-P-1-2, 2: 100–107.77 mm SL, Paragenetype COI (GenBank KJ174308 View Materials ), Paragenetype cyt b (GenBank KJ174298 View Materials ), same collection data as holotype. SAIAB 188294, 11: 50–147 mm SL, Mokolo River, above dam wall (opposite bank) of dam at intersection of roads R518 and R510, 23°22'01.7" S, 27°41'09.6" E, P. Maake and O. Gon, 12 October 2010. USNM 410774, 2: 119.73–130.8 mm SL, same collection data as previous lot. SAIAB 188290, 20: 57–197 mm SL, Tshinane River, R523 below bridge at Ngudza, 22°53'56.3" S, 30°31'28.7" E, P. Maake and O. Gon, 10 October 2010. BMNH 2013.9.4.72-73, 2: 110–142 mm SL, same collection data as previous lot.
Non-type specimens. SAIAB 79147, 6: 37.2–122 mm SL, Blyde River. SAIAB 188301, 2: 62.2–140.92 mm SL, Cross River, Danda village, below bridge, tributary from Nwanedi Dam. SAIAB 84510: 22 mm SL, Damani Dam. SAIAB 84485, 5: 35–48mm SL, Damani Dam. SAIAB 84439, 2: 52–79 mm SL, Sambandou Junction. SAIAB 84527, 7: 32–56 mm SL, Mbwedi Pumphouse.
Diagnosis. Middle body depth 21.9–28.1% SL; distance from dorsal fin origin to end of caudal peduncle 39.9– 41.7% SL, distance from anal fin origin to end of caudal peduncle 42.8–44.9% SL; dorsal fin length 15.5%–20.0% SL; anal fin length 23.2–25.5% SL; depth of caudal peduncle 26.0–40.6% of its length; scales around caudal peduncle 16; total vertebrae 44; anterior GR (5–6) + (5) = (10–11); posterior GR (8) + (9) = 17; dorsal fin rays 22– 25; anal fin 28–31 rays; scales along lateral line 69–72. Conical teeth on upper/lower jaws 4–6.
Description. Measurements and meristic counts are given in Tables 8 View TABLE 8 . Head with terminal mouth well in front of eye; mental lobe on lower jaw protruding in front of upper jaw; head and body compressed; snout rounded and blunt; pre-anal distance shorter than pre-dorsal distance; distance from origin of anal fin to origin of dorsal fin slightly greater or equal to middle body depth; pre-pelvic distance twice as long as the distance between pelvic and anal fins; dorsal and anal fins set well back on the body, situated about two thirds of standard length from the snout and opposite each other; dorsal fin shorter, originating on vertical at 4th or 5th anal-fin ray and ending before anal-fin base; distal margin of dorsal and anal fins obliquely orientated, with rays becoming gradually shorter posteriorly; dorsal fin rays 23 (22–25), its anterior rays highest; anal fin rays 29 (28–31); males at sexual maturity have kink in anal fin base, distinctly curving inward; anterior anal fin rays of sexually mature males strong and sometimes darker, 3rd–5th rays longer than first two rays, crescentic and rounded or curved backwards, but anteriorly sharp or pointy in females and juveniles; pectoral fin rays 10, fin length 53.7–82.3% of head length; body depth 26.6% (21.9–28.1%) SL; caudal peduncle slender, subcylindrical across its entire length, 19.2% (17.6–24.5%) SL; 16 scales (N = 35) around the caudal peduncle; caudal peduncle depth 34.9% (26.0–40.6%) of its length; lateral line scales 70 (69–72), cycloid and with reticulate striae; jaws with 4–6 (usually 4–5, N = 27) conical teeth; 44 vertebrae (excluding urostyle); gill rakers on anterior side more developed (longer and smooth, i.e. no spines), anterior GRt = 10 (10–11) on the first gill arch ( Table 6), gill raker of anterior side conicals with thin tips; posterior GRt 17 on the posterior side of first gill arch; gill rakers on posterior side shorter, thicker and with flattened tips.
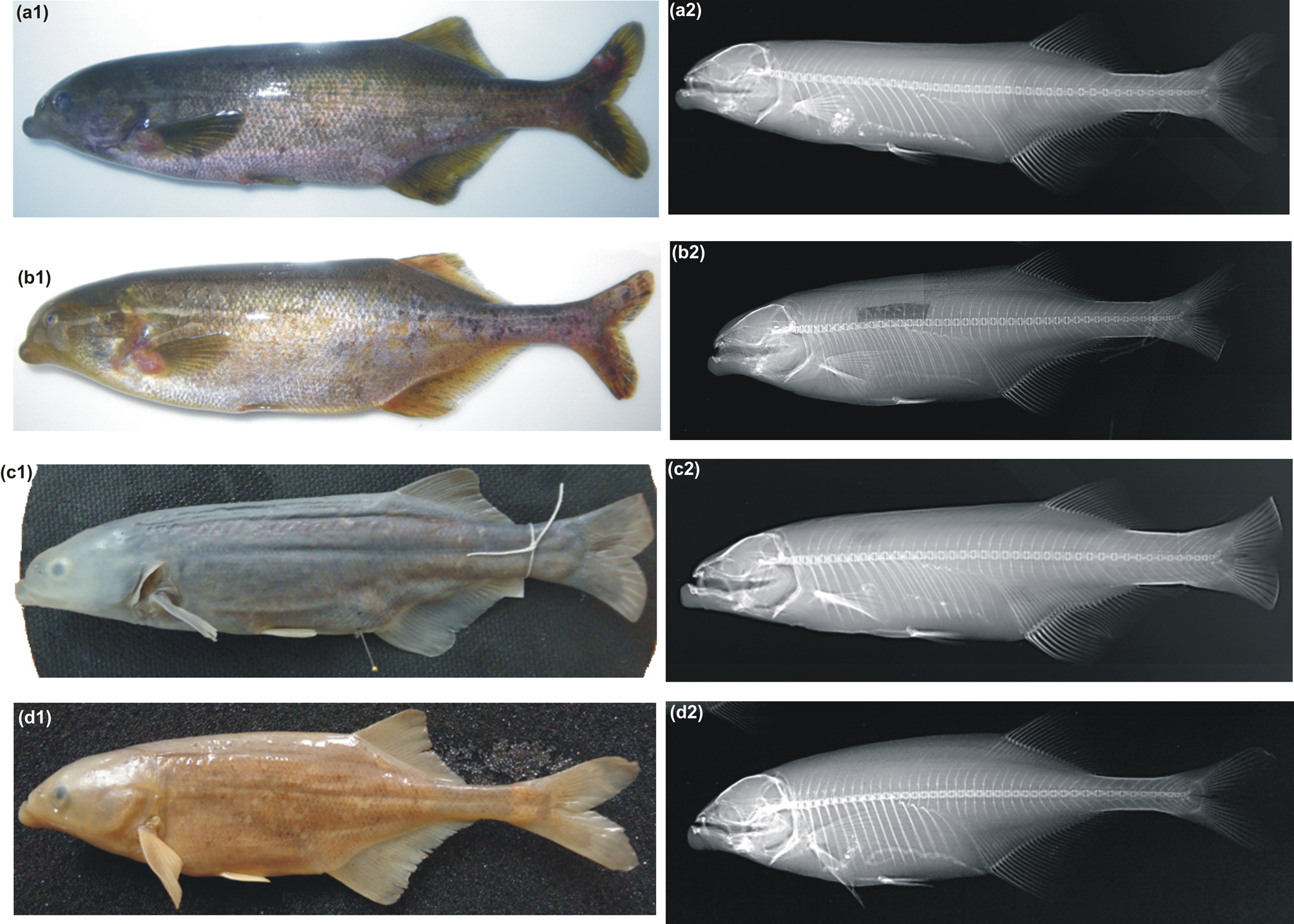
Coloration. Immediately after death specimens are medium brown, speckled with darker, irregular blotches on the sides, fins yellowish ( Fig. 2 View FIGURE 2 b). In 70% alcohol, the head (including eye), fins and upper back sometimes covered by milky-grey mucus layer. Specimens from Tshinane River are similar to those from type locality, but clearly darker from belly to back, including the fins.
Electric organ discharge: According to Kramer et al. (2007), the EODs of ten individuals (of which six were examined morphologically in the present study: SAIAB 79147) of this species (referred to as Marcusenius Limpopo population) were distinct from those of M. pongolensis , M. macrolepidotus , M. altisambesi and M.
devosi View in CoL . Kramer et al. (2007) concluded that, when comparing EODs from South African systems: (1) the durations of the N phase or Ndur variable of female and juvenile EODs from M. krameri View in CoL increased with size, while not a single EOD variable correlated with size in M. pongolensis View in CoL , (2) the M. krameri View in CoL EODs differed significantly from those of M. pongolensis View in CoL in three variables, i.e. Ndur, Parea and Narea (see table VIII in Kramer et al. 2007).
Character Holotype N Mean/Median Min Max Std. Dev SL, mm 116.21 mm 35 113.59 48.2 207 34.65 NB: MP = M. pongolensis View in CoL , ML = M. lucombesi View in CoL , MC = M. caudisquamatus View in CoL .
Remarks. Compared with M. pongolensis , M. krameri specimens are best distinguished by their moderately short, laterally compressed deep body. Marcusenius krameri also has the greater distance from dorsal fin origin to end of caudal peduncle (39.9–41.7% vs 37.1–39.8% SL) and from anal fin origin to end of caudal peduncle (42.8– 44.9% vs 40.8–42.1% SL), and its anal-fin base is longer (23.2–25.5% vs 19.5–21.9 % of SL), all of which do not overlap with M. pongolensis . Marcusenius krameri differed clearly from M. macrolepidotus with the dorsal and anal fin length, caudal peduncle depth and body depth, all of which are less and caudal peduncle length and number of scales around caudal peduncle which are more in the latter species. Marcusenius krameri also has the highest number of posterior gill rakers and anal-fin rays among the South African species ( Tables 6 and 7, respectively). Caudal peduncle depth of M. krameri increased significantly with size as compared to other species, resulting in the unusually large range of this character. Marcusenius krameri is closely related to M. caudisquamatus , but the latter species has a deeper caudal peduncle into its length (40.4–51.7% vs 26.0–40.5) and 19 circumpeduncular scales (see also Tables 6, 7). Marcusenius krameri species can also be distinguished based on the phylogenetic relationships forming a sister species to M. caudisquamatus and its genetic divergence in the in the cyt b dataset (0.9–2.4%) ( Fig. 4 View FIGURE 4 ).
Distribution and ecology. Marcusenius krameri occurs in the mainstem of the Limpopo River and several of its tributaries, ranging from the Matlabas River in the west to the Olifants River in the east. Individuals of this species were found in groups. During the day they were commonly encountered below undercut river banks, especially in dense networks of tree roots or reed beds along the stream margins. Places with shallow water and slow-flowing reaches of rivers and streams are especially attractive to them. Large numbers of small juveniles were found in bushes of aquatic reeds and floating grass on the periphery of the river or in side channels. The conductivity in most rivers where it occurred ranged from 55 to 106 µS/cm (type locality), and a pH from 7.41 to 8.5 units in October 2010 ( pH = 7.86, in type locality). In summer, water temperature was 24–27 °C between 10 am to 6 pm. Kramer et al. (2007) reported a higher water conductivity of 154µS/cm and a much colder temperature, 16.7 °C, between the 25th and 26th September 1998.The turbidity where specimens were collected was moderate to high at a depth not exceeding 1.5 m.
The biology of the species is unknown, but males mature from an estimated length of 84 mm SL (the characteristic notched anal fin of males was observed at this size), but females probably mature at a larger size. The largest specimens were a 207 mm SL female and a 192 mm SL male.
Etymology. Marcusenius krameri is named for Prof. Bernd Kramer from the Zoological Institute of the University of Regensburg, Germany, in recognition of his contribution to the systematics of southern African mormyrids.
TABLE 8. Basic statistics for morphological characters of Marcusenius krameri sp. nov. See Table 1 for characters definitions. Asterisks indicate non-overlapping characters with other Marcusenius species.
| HL, mm | 24.07 mm | 35 | 23.37 | 11.63 | 42.07 | 6.44 |
|---|---|---|---|---|---|---|
| Ratio of SL | ||||||
| HL | 0.2071 | 35 | 0.2083 | 0.1860 | 0.2427 | 0.0129 |
| vBD | 0.2573 | 35 | 0.2434 | 0.2188 | 0.2711 | 0.0136 |
| mBD | 0.2662 | 35 | 0.2511 | 0.2190 | 0.2808 | 0.0145 |
| dAD | 0.2730 | 35 | 0.2586 | 0.2432 | 0.2802 | 0.0089 |
| CPL | 0.1915 | 35 | 0.2053 | 0.1762 | 0.2448 | 0.0139 |
| PDL | 0.6298 | 35 | 0.6343 | 0.6178 | 0.6538 | 0.0109 |
| PAL | 0.5935 | 35 | 0.5971 | 0.5581 | 0.6172 | 0.0129 |
| pD | 0.3989 | 35 | 0.4047 | 0.3989 | 0.4168 | 0.0038 *MP |
| pA | 0.4319 | 35 | 0.4360 | 0.4280 | 0.4490 | 0.0051 *MP |
| pPL | 0.2311 | 35 | 0.2317 | 0.2123 | 0.2467 | 0.0080 |
| pVL | 0.3848 | 35 | 0.3951 | 0.3805 | 0.4140 | 0.0077 |
| dVA | 0.2193 | 35 | 0.2090 | 0.1846 | 0.2350 | 0.0133 |
| dPA | 0.3823 | 35 | 0.3786 | 0.3450 | 0.3994 | 0.0150 |
| dPV | 0.1740 | 35 | 0.1840 | 0.1580 | 0.2074 | 0.0104 |
| LD | 0.1867 | 35 | 0.1813 | 0.1554 | 0.2002 | 0.0115 |
| LA | 0.2472 | 35 | 0.2404 | 0.2319 | 0.2547 | 0.0063 * MP |
| CPD/CPL | 0.3486 | 35 | 0.3393 | 0.2602 | 0.4058 | 0.0327 * ML, MC |
| Ratio of HL | ||||||
| HW | 0.5991 | 35 | 0.5893 | 0.4985 | 0.6362 | 0.0267 |
| LSN | 0.4936 | 35 | 0.4765 | 0.4383 | 0.5197 | 0.0223 |
| IOW | 0.4591 | 35 | 0.4475 | 0.3483 | 0.5094 | 0.0310 |
| ED | 0.1683 | 35 | 0.1897 | 0.1368 | 0.2267 | 0.0183 |
| LP | 35 | 0.7029 | 0.5370 | 0.8227 | 0.0657 | |
| Counts | Median | |||||
| nD | 23 | 35 | 23 | 22 | 25 | 0.6585 |
| nA | 29 | 35 | 28 | 28 | 31 | 0.7778 * MP |
| SPc | 16 | 35 | 16 | 16 | 16 | 0 * MC |
| nTL | 6 | 35 | 5 | 4 | 6 | 0.5314 |
| nTU | 5 | 35 | 5 | 4 | 5 | 0.4710 |
| TV | 44 | 22 | 44 | 44 | 44 | 0 * MP |
| LSS | 70 | 23 | 70 | 69 | 72 | 0.9639 |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Marcusenius krameri
| Maake, Pholoshi A., Gon, Ofer & Swartz, Ernst R. 2014 |
Marcusenius
| Kramer 2007: 661 |