Centrolene sabini, Catenazzi, Alessandro, May, Rudolf Von, Lehr, Edgar, Gagliardi-Urrutia, Giussepe & Guayasamin, Juan M., 2012
|
publication ID |
https://doi.org/ 10.5281/zenodo.215385 |
|
DOI |
https://doi.org/10.5281/zenodo.5685479 |
|
persistent identifier |
https://treatment.plazi.org/id/03935706-AD00-9F1E-1CC5-FBBCFE79F2A1 |
|
treatment provided by |
Plazi |
|
scientific name |
Centrolene sabini |
| status |
sp. nov. |
Centrolene sabini View in CoL new species
( Figures 1–4 View FIGURE 1 View FIGURE 2 View FIGURE 3 View FIGURE 4 )
Centrolene sp.: Catenazzi et al. 2009: 84–87.
Holotype. MUSM 27941, an adult male from Esperanza (13°10’37” S, 71°36’31” W, 2800 m), Distrito Kosñipata, Provincia Paucartambo, Región (Departamento) Cusco, Perú, collected on 9 February 2009 by A. Catenazzi, J. C. Jahuanchi and R. Sotelo.
Paratopotypes. MUSM 28017, an adult male from the type locality, collected on 4 January 2010 by A. Catenazzi. MUSM 28018–28019, two adult males ~ 100 m S of the type locality (13°10’41” S, 71°36’31” W, 2750 m), collected on 6 January 2010 by A. Catenazzi and J. C. Jahuanchi.
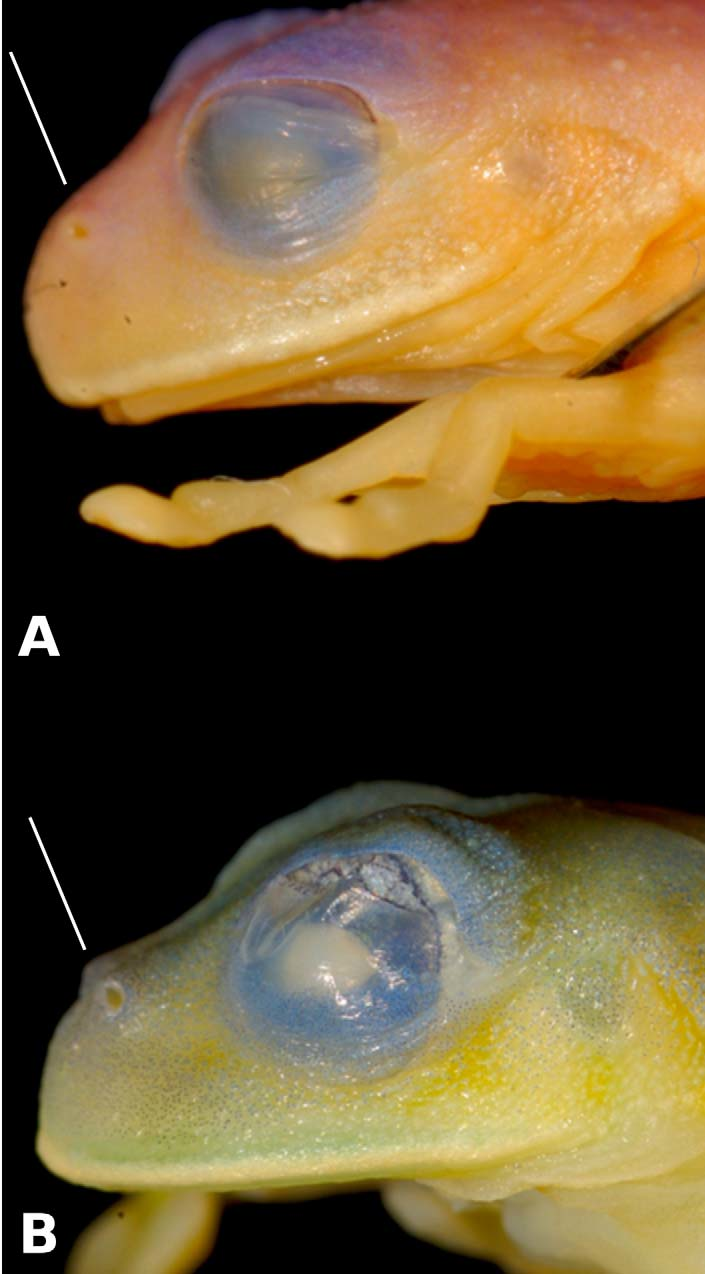
Diagnosis. Centrolene sabini is distinguished from species in other centrolenid genera in Peru by having a white ventral parietal peritoneum (transparent in Hyalinobatrachium ) and by having humeral spines in males ( Fig 1 View FIGURE 1 ; absent in Cochranella , Hyalinobatrachium , Nymphargus and Rulyrana ). Morphologically, the new species is most similar to C. lemniscatum from the eastern slopes of the Andes in Región (Departamento) San Martín, 1050 km north of the Kosñipata Valley ( Duellman & Schulte 1993). Both species have humeral spines that are evident and lack vomerine teeth. Centrolene sabini differs from C. lemniscatum ( Figs. 1–2 View FIGURE 1 View FIGURE 2 ; characters of the latter in parentheses) by having distinct yellowish white labial stripes that extend into continuous lateral stripes (white labial stripes not continuous with thin lateral stripes), larger SVL ranging between 29.6–31.2 mm (n = 5) with the smallest male being larger than an adult female of C. lemniscatum (27.2 mm in a female; 27.0 mm in the male holotype), strongly protruding nostrils (directed anterolaterally, but not strongly protruding; Fig. 2 View FIGURE 2 ), internarial area depressed (barely depressed), snout profile obtuse or inclined anteroventrally (bluntly rounded; Fig. 2 View FIGURE 2 ), and by its long advertisement call composed of 8–14 peaked notes (short call with a single tonal note in C. lemniscatum , G. Gagliardi- Urrutia, pers. obs.). Additionally, C. sabini lay eggs on the upper side of leaves (Type A of Guayasamin et al. 2009), whereas C. lemniscatum lay eggs on the tip of upper sides of leaves (Type B; G. Gagliardi-Urrutia, pers. obs.). Moreover, the new species has green-yellowish coloration (bright green in C. lemniscatum ), large dorsal spicules with yellowish-green tips (smaller, white spicules), and iris silvery-bronze with fine black reticulations (iris white with fewer reticulations). Centrolene sabini could also be confused with the morphologically similar C. buckleyi , which has a call with 1–5 notes at fundamental frequencies of 3520–4139 Hz in Yanayacu, Amazonian slopes of the Andes in Ecuador ( Guayasamin et al. 2006); in Colombia, advertisement calls of C. buckleyi are short and composed of a single note ( Bernal et al. 2004) with a fundamental frequency of up to 5200 Hz ( Bolívar et al. 1999). In contrast, the advertisement call of C. sabini is long and includes 8–14 peaked notes with fundamental frequencies ranging between 2597 and 3919 Hz (Catenazzi et al. 2009; as Centrolene sp.). Morphological characters found in species of Peruvian Centrolene but not in C. sabini include: Finger I longer than Finger II in C. fernandoi , and C. muelleri ; dermal fringes on limbs in C. hersperium and C. muelleri ; and vomerine teeth in C. azulae and C. fernandoi . Additionally, analysis of the mitochondrial genes 16S and ND1 clearly differentiate C. sabini from morphologically similar species found in the Andes (i.e., C. altitudinale , C. buckleyi , C. heloderma , C. hesperium , C. notostictum , C. muelleri , and C. venezuelense ; no sequences of C. lemniscatum are available). Genetically, with the current taxon sampling, the most closely related species to C. sabini is C. lynchi (Duellman 1980) , an endemic to Colombia and Ecuador; C. lynchi differs from C. sabini by having both dark and light spots on the dorsum and an advertisement call that consists of a tonal note followed by 1–3 peaked notes with higher dominant frequencies ( Dautel et al. 2011). Centrolene lynchi further differs from C. sabini in lacking a yellowish white lateral stripe (Duellman 1980), and in having smaller SVL averaging 24.7 mm in males and 24.9 mm in a single female, snout truncate in dorsal view and rounded in profile, dorsal skin shagreened with fine spicules, and different webbing formulas between finger and toes (Duellman 1980). Centrolenids not found in Peru that are morphologically similar to C. sabini include C. altitudinale (endemic to Cordillera de Mérida, Venezuela), C. bacatum , C. ballux (endemic to the Andes of Colombia and Ecuador), C. notostictum , C. quindianum (endemic to the Colombian Andes), and C. venezuelense (endemic to Cordillera de Mérida, Venezuela). Centrolene altitudinale differs from C. sabini by lacking a yellowish white line dividing the upper and lower flanks. Centrolene bacatum is distinguished from C. sabini mainly by having white warts below the eye and being considerable smaller (SVL <21.8 mm). Centrolene ballux is recognized by having, in life, small greenish white to yellowish white warts and an SVL <23.3 mm (tips of spicules yellowish-green, male SVL 29.6–31.2 mm in C. sabini ). Centrolene notostictum is also considerable smaller than C. sabini (male SVL <25.0 mm in C. nostostictum ) and lacks the line between the upper and lower flanks that characterizes C. sabini . Centrolene quindianum is easily differentiated by having small black spots on the dorsum (absent in C. sabini ). Finally, C. venezuelense is distinguished by its call consisting of 2–4 notes and a dominant frequency at 3900–4400 Hz ( Señaris & Ayarzagüena, 2005), whereas C. sabini has a call with 8–14 notes and a frequency of 2597–3919 Hz (Catenazzi et al. 2009).
Characterization. (1) Males with humeral spines; (2) liver tetralobed, covered by clear peritoneum; (3) parietal peritoneum white; visceral peritoneum translucent; (4) in life, dorsum green with yellowish-green spots and patches, iris silvery-cream with fine black reticulations, yellowish white labial stripe continuous with distinct stripe on flanks, except for area between the edge of the mouth and point of arm insertion, where stripe becomes thin ( Fig. 1 View FIGURE 1 A–C); bones green; (5) in preservative, pale lavender with white lateral stripe; (6) dorsal surfaces of males spiculate, with spicules more numerous near orbits; (7) snout obtuse or inclined anteroventrally in lateral profile and rounded in dorsal profile ( Fig. 2 View FIGURE 2 A); (8) tympanum small (tympanum diameter 16–23% of eye diameter), oriented posterolaterally; tympanic annulus indistinct; tympanic membrane pigmented, similarly to surrounding skin; (9) no webbing between Fingers I and II, webbing between Fingers II and III vestigial ( Fig. 3 View FIGURE 3 A), webbing formula III 2 ½––2½IV; (10) prepollex not separated from Finger I; in males, nuptial pad Type I ( Fig. 1 View FIGURE 1 B, 3A); (11) Finger I shorter than Finger II (Finger II 110–117% of length of Finger I); (12) ulnar and inner tarsal folds low; outer tarsal fold absent; (13) webbing on foot moderate ( Fig. 3 View FIGURE 3 B), webbing formula: webbing on foot I 1 ½– 2 II 1– 2III 2– 2 ½ IV3 – 2 ½V; (14) one pair of enlarged tubercles below vent; (15) disc of Finger III of large size, about 56–65% of eye diameter; (16) vomerine teeth absent; (17) males call from upper sides of leaves; females deposit eggs on upper sides of leaves ( Fig. 4 View FIGURE 4 ); fighting behavior unknown; (18) in adult males, SVL 30.5 ± 0.3 (range 29.6–31.2 mm; n = 5); females unknown.
Description of holotype. Adult male, SVL 31.2 mm. Head width 118% of head length, 32% of SVL; head length 27% of SVL; snout rounded in dorsal view and obtuse or inclined anteroventrally in lateral profile; canthus rostralis indistinct, straight; loreal region slightly concave; nostril ovoid, directed dorsolaterally on protuberance, closer to tip of snout than to eye; internarial area slightly depressed. Eye moderately large, ED 40% of HL, directed anterolaterally; greatest diagonal EW 82% of IOD; transverse diameter of disc of Finger III 65 % eye diameter. Tympanum small, oriented laterally with slight posterior inclination; tympanic annulus not evident; supratympanic fold moderate, obscuring dorsal portion of tympanic annulus; tympanic membrane pigmented similarly to surrounding skin. Vomerine teeth absent; choanae widely separated and quadrangular; tongue ovoid, with ventral posterior third not attached to mouth floor and posterior margin notched. Vocal sac single, median, subgular; vocal slits extending posterolaterally from the posterolateral base of tongue to angle of jaws.
Humeral spine present and visible externally ( Fig. 1 View FIGURE 1 A). Hand length 132% of head length; digits broad, bearing narrow lateral fringes, including outer edge of Finger III and inner edge of Finger I; low ulnar fold visible. Relative lengths of fingers: III> IV> II> I; webbing absent between Fingers I and II and vestigial between fingers II and III; webbing formula for outer fingers: III 2 ½–2 ½IV; terminal discs subtruncate; width of disc on Finger III 61.5% of eye diameter; width of disc on Finger I 56.9% of that of Finger II. Subarticular tubercles broad, nearly as wide as digits, round, elevated; supernumerary tubercles prominent on proximal segments; palmar tubercle moderately large, ovoid with diagonal orientation, elevated, its length 12% of HnL. Prepollex slightly enlarged; nuptial pad present (Type I of Flores 1985), covering the proximal third of Finger I; prepollical spine absent.
Hind limbs long, slender, length of tibia 55% of SVL, length of foot 49% of SVL; dermal fringes and tarsal tubercles absent; inner tarsal fold low, outer tarsal fold absent; inner metatarsal tubercle elongate and elliptical, low, measuring 10% of foot length; outer metatarsal tubercle absent. Toes moderately slender, bearing lateral fringes, including outer edge of Toe V and inner edge of Toe I; length of toes IV> V> III> II> I; toes about three-fifths webbed; webbing formula 1½– 2 II 1– 2III 2– 2 ½ IV3 – 2 ½V. Terminal discs subtruncate; width of digit on Toe I 55 % of that on Toe IV, which is 116% of that on Finger III; width of disc on Toe IV 86 % of width of disc on Finger IV. Subarticular tubercles as wide as digits, round, elevated.
Skin on dorsal surfaces of head and body spiculate; skin on dorsal surfaces of limbs smooth. Flanks smooth dorsally, coarsely granular ventrally. Skin on belly and ventral surfaces of thighs coarsely granular; other ventral surfaces (including throat) smooth. Cloacal opening directed posteroventrally at upper level of thighs; cloacal sheath short and unmodified; cloacal tubercles small, fleshy, located immediately posterior to cloacal slit; para- and subclocal folds absent; one pair of moderately large, round, elevated tubercles on ventral surfaces of thighs below vent.
MUSM MUSM MUSM MUSM Character 27941 28017 28018 28019 Color in life. Dorsum green with posterior elongate patches of yellowish-green; tips of spicules yellowishgreen; yellowish-white labial stripe continuous with distinct lateral stripe above insertion of arm extending to groin; flanks below lateral stripe yellow; narrow white stripe on outer edge of forearm and foot; digits pale yellowish-green; iridophores covering between half to two-thirds of the parietal (condition P3 of Cisneros-Heredia & McDiarmid 2007); heart not visible, iridophores covering the pericardium (condition V1 of Cisneros-Heredia & McDiarmid 2007); bones green; iris silvery-bronze with fine black reticulations.
Color in preservative. Dorsal surfaces of head and body pale lavender, limbs and venter cream; edge of upper lip and white lateral stripe distinct. Dense melanophores in skin of dorsal surfaces of head (including upper eyelid) and body; tips of spicules unpigmented; less dense melanophores in loreal and tympanic regions, on posterior side of dorsum and dorsal surfaces of forelimbs, thighs, shanks, tarsi, and fourth and fifth toes. Flanks, anterior and posterior surfaces of thighs, fingers and inert three toes, and all ventral surfaces are cream and lack melanophores.
Measurements (in mm) of the holotype. SVL = 31.2; tibia length = 17.5; foot length = 15.3; head length = 8.4; head width = 9.9; interorbital distance = 3.0; upper eyelid width = 2.5; internarial distance = 2.2; eye-to-nostril distance = 1.8; eye-to-nostril distance = 1.8; eye diameter = 3.4; tympanum diameter = 0.8; eye-tympanum distance = 1.6; radioulna length = 7.0; hand length = 11.0; Finger-I length = 4.7; Finger-II length = 5.5; and disc of Finger III = 2.2 ( Table 1 View TABLE 1 ).
Variation. Measurements of the paratypes are given in Table 1 View TABLE 1 . Only males are known. There is little variation in coloration among these specimens. In MUSM 28017 and MUSM 28018 the spicules are green and cannot easily be distinguished from the background dorsal coloration. Furthermore, in MUSM 28019 the lateral stripe is thin between the edge of the mouth and point of arm insertion.
Reproduction and advertisement call. The advertisement call consists of 8–14 peaked notes with emphasized frequencies between 2597–3919 Hz (Catenazzi et al. 2009; as Centrolene sp.). All recorded calls of C. sabini displayed consistent patterns of amplitude modulation that are unusual among centrolenid frogs. Length of calls recorded at 12°C varied between 3089–7563 ms when the call included only a series of 6–11 notes, and 8537–10111 ms when the first series of notes was followed by a second series of 3 notes. These two series of notes were separated by intervals of 1459–1652 ms. Advertisement calls were separated by intervals of 258 ms on average. A complete description of the advertisement call on the basis of vocalizations produced by two specimens (MUSM 27941, holotype and MUSM 17966) was provided by Catenazzi et al. (2009). Epiphyllous egg clutches contained between 35 and 45 eggs (n = 5; Fig. 4 View FIGURE 4 ).
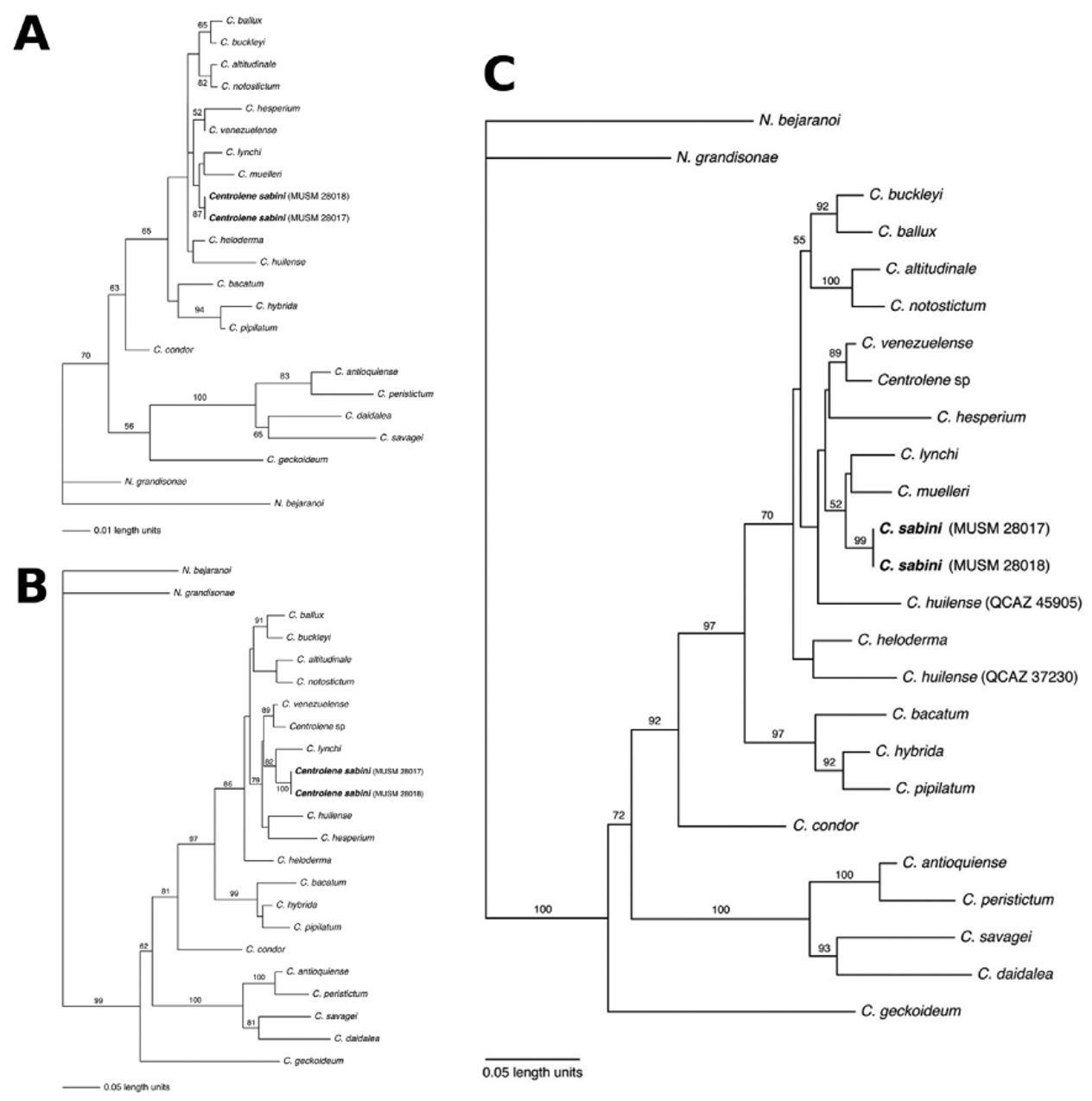
Phylogenetic relationships. The phylogenetic analysis of both genes and the combined dataset resulted in similar topologies ( Fig. 5 View FIGURE 5 ).
The combined mitochondrial tree and the 16S topology show that Centrolene sabini is closely related to C. lynchi and C. muelleri ( Fig. 5 View FIGURE 5 A, C). The phylogeny based on ND1 places C. lynchi as the sister taxon of C. sabini ( Fig. 5 View FIGURE 5 B; ND1 sequences were not available for C. muelleri ). Our phylogenetic analyses confirm that C. buckleyi is not closely related to the new species, despite similarities in morphology ( Bolívar et al. 1999; Bernal et al. 2004; Guayasamin et al. 2006; Catenazzi et al. 2009). Additionally, six species that until now were placed in the genus Centrolene (sensu Guayasamin et al., 2009) based on morphological traits (i.e., ballux , condor , heloderma , huilense , lynchi , and muelleri ) show a close phylogenetic affinity with Centrolene species, although taxon sampling only included species of one additional genus ( Nymphargus ). Therefore, it is likely that placement of these six species in the genus Centrolene is correct.
Distribution and ecology. Centrolene sabini is only known from elevations between 2750 and 2800 m in the Kosñipata valley, upper Manu National Park. The species was first collected and recorded on 28 January 1999 ( Catenazzi & Rodríguez 2001). Males were active and calling at night temperatures ranging between 11.0–12.7°C. Sympatric frog species include Bryophryne nubilosus , Gastrotheca antoniiochoai , G. nebulanastes , Noblella pygmaea , Oreobates lehri , Pristimantis pharangobates , Psychrophrynella usurpator , Telmatobius sp. ( Lehr & Catenazzi 2008, 2009; Duellman et al. 2011). Catenazzi et al. (2011) reported the presence of the fungus Batrachochytrium dendrobatidis in several species of frogs from the Kosñipata Valley, but in none of the specimens of C. sabini collected in 2009 (n = 1) and 2010 (n = 4).
Etymology. The specific name is dedicated to Andrew Sabin, in recognition of his support for herpetology. We propose the following common names: Sabin's glassfrog in English and Rana de cristal de Sabin in Spanish.
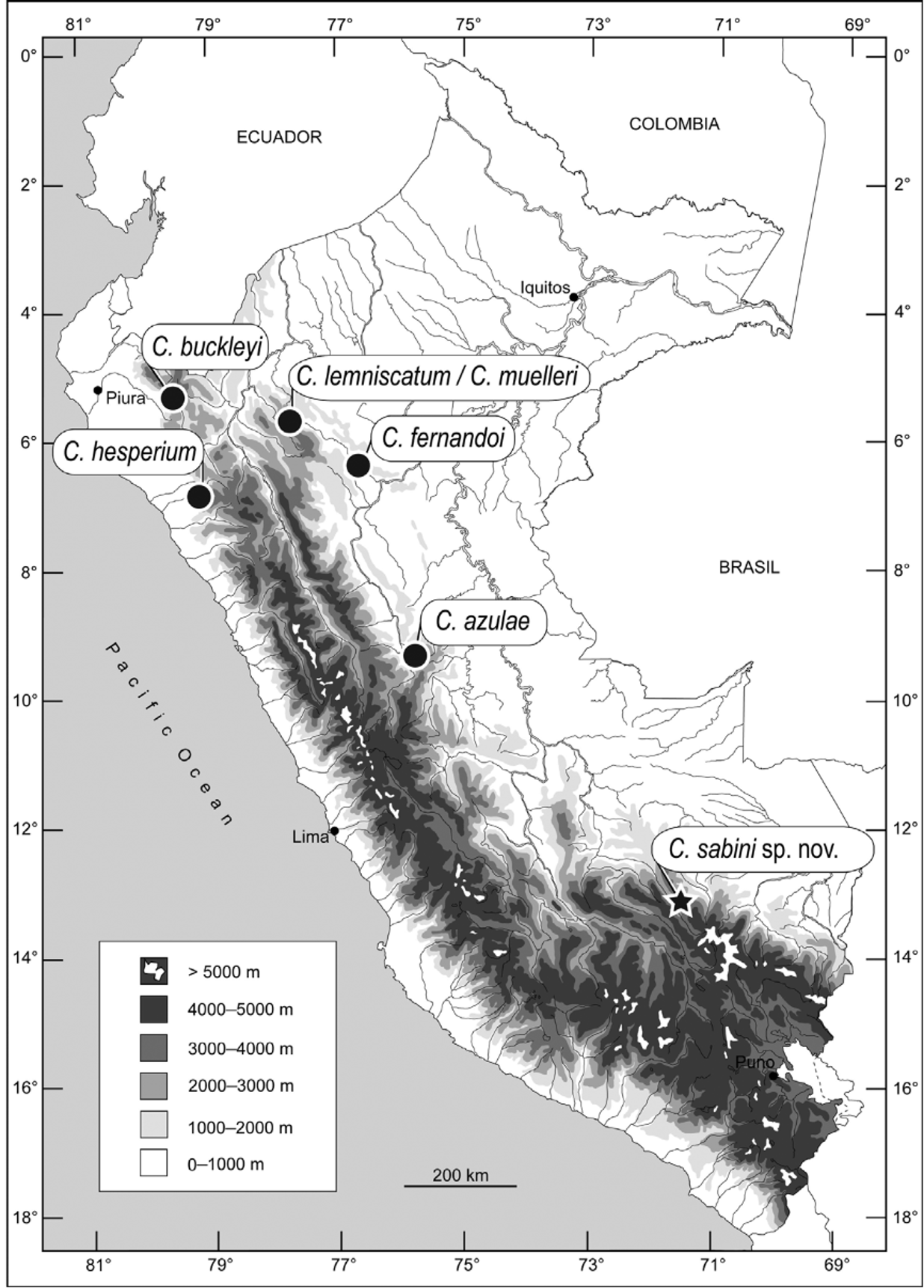
Discussion. Seven species of Centrolene have been reported from Peru ( Fig. 6 View FIGURE 6 ), but the generic placement of half of these taxa is uncertain. Guayasamin et al. (2009) listed C. azulae and C. fernandoi as “ incertae sedis within Centroleninae because molecular data are not available, and morphological and behavioral characters do not provide unambiguous evidence on their generic placement”. These two species differ from most other species of Centrolene by having vomerine teeth. A third species, C. lemniscatum , was tentatively assigned to Centrolene by Guayasamin et al. (2009) on the basis of morphology.
The new species presents a combination of morphological and behavioral characteristics that is typical of the genus Centrolene ( Guayasamin et al. 2009) . These traits include the presence of humeral spines and nuptial pads in males, a tetralobed liver covered by a translucent hepatic peritoneum, a white ventral peritoneum anteriorly (concealing the heart) and translucent posteriorly, green bones, melanophores giving a lavender coloration in preservative, males calling from the upper side of leaves, and females depositing eggs on the upper side of leaves along forest streams. On the basis of these characteristics, and the phylogenetic trees presented in Fig. 5 View FIGURE 5 , we assign the new species to Centrolene .
The discovery of Centrolene sabini considerably extends the geographic range of the genus from the type locality of C. azulae in the Cordillera Central in northern Peru ( Flores & McDiarmid 1989) to the Cordillera de Paucartambo in southern Peru. However, another species of Centrolene is known from photographic records taken in a stream at 1750 m in Santuario Nacional Megantoni (12°11’12” S, 72°28’12” W; 145 km NW of the Kosñipata Valley; Rodríguez & Catenazzi 2005). We are unable to assess whether the geographic ranges of C. sabini and the species from Santuario Nacional Megantoni are disjunct from that of other congeneric species, because the montane forests on the eastern slopes of the Peruvian Andes have been poorly explored. However, along the elevational gradient of the Kosñipata Valley, intensive sampling of amphibian communities over the past decade revealed the presence of C. sabini in only two adjacent high-elevation streams (Catenazzi et al. 2011). The elevational range does not overlap with that of other centrolenid species known to occur in this valley ( Duellman 1976; Cannatella & Duellman 1982; Catenazzi et al. 2009), or with the elevational range of any other species of Centrolene in Peru (Table 2). Therefore, C. sabini seems to occupy a narrow elevational and geographical range in southern Peru.
1 Flores & McDiarmid (1989) 2 Duellman & Wild (1993)
3 Duellman and Schulte (1993) 4 Cadle and McDiarmid (1990)
TABLE 1. Measurements (in mm) of the types of Centrolene sabini.
| Sex | male | male | male | male |
|---|---|---|---|---|
| SVL | 31.2 | 29.6 | 30.4 | 30.9 |
| TL | 17.5 | 16.0 | 16.1 | 17.0 |
| FL | 15.3 | 14.4 | 15.3 | 15.3 |
| HL | 8.4 | 8.0 | 8.2 | 8.3 |
| HW | 9.9 | 9.6 | 9.6 | 9.8 |
| IOD | 2.5 | 3.0 | 2.9 | 3.0 |
| EW | 2.2 | 2.4 | 2.2 | 2.5 |
| IND | 2.0 | 2.2 | 2.0 | 2.2 |
| E–N | 1.6 | 1.6 | 1.8 | 1.8 |
| Eye D | 3.2 | 3.2 | 3.2 | 3.4 |
| Tymp D | 0.5 | 0.6 | 0.6 | 0.8 |
| E–T | 1.1 | 1.2 | 1.5 | 1.6 |
| RL | 6.7 | 6.8 | 6.9 | 7.0 |
| Hand L | 10.5 | 10.8 | 11.0 | 11.0 |
| Finger I | 4.2 | 4.8 | 5.0 | 4.7 |
| Finger II | 4.8 | 5.3 | 5.6 | 5.5 |
| Disc F I | 1.0 | 1.2 | 1.1 | 1.2 |
| Disc F III | 2.0 | 2.0 | 1.8 | 2.2 |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |