Veromessor (Brown, 1949)
|
publication ID |
https://doi.org/10.11646/zootaxa.5206.1.1 |
|
publication LSID |
lsid:zoobank.org:pub:CE749F6C-5832-4152-AB4B-6D89ACCDD560 |
|
DOI |
https://doi.org/10.5281/zenodo.7327852 |
|
persistent identifier |
https://treatment.plazi.org/id/039387FD-FF87-FFA4-7FC5-FE1E5DECFBBD |
|
treatment provided by |
Plazi |
|
scientific name |
Veromessor |
| status |
|
Biology of Veromessor
Veromessor is an interesting genus because it contains relatively few species, yet notable interspecific variation occurs in several morphological and ecological traits. Morphological variation occurs in degree of psammophore development, degree of worker polymorphism and worker body size, size of the propodeal spines, and eye size and structure; ecological variation occurs in colony size and associated foraging method, seasonality of mating flights, and queen size and colony founding strategy. We also discuss mating frequency given that queens of V. pergandei mate with multiple males. Interspecific variation in each character is discussed below, while a companion paper maps these traits onto a molecular phylogeny (M.L. Borowiec, unpub. data). Moreover, species of Veromessor are used in ecological, sociobiological, and behavioral studies such that it is important to understand if trait evolution results from convergent evolution or common ancestry. Distribution patterns also are discussed because some species display parapatric distributions, while other species are broadly sympatric but may or may not occur at the same locales or in the same habitats (see also Bennett, 2000; Johnson, 2000b).
Psammophore development
The psammophore consists of numerous long hairs that extend from the lateroventral margin of the head. The function of these hairs is unknown, but it has been suggested that they are used to carry sand or soil ( Porter & Jorgensen, 1990; Spangler & Rettenmeyer, 1966; Wheeler, 1907). Psammophores can also be used to carry seeds. Johnson (1991) observed Pogonomyrmex rugosus workers carry multiple seeds in their psammophore when presented with a patch of novel seeds, whereas V. pergandei did not use their psammophore to carry multiple seeds. Psammophores are rare in ants, but they are common in species that inhabit arid areas, with up to one-third of the species in some desert areas possessing psammophores ( Bernard, 1948).
The psammophore ranges from poorly to well developed across species of Veromessor , with development diagnosed by number and distribution of J-shaped hairs on the ventral surface of the head capsule (=hypostomal region). As discussed by Bennett (2000), the psammophore in species of Veromessor can be placed into four categories of increasing development: (1) J-shaped hairs mostly absent, with scattered straight or evenly curved hairs, (2) J-shaped hairs present but not arranged in a distinct row, usually mixed with straight or evenly curved hairs, (3) J-shaped hairs arranged in a V-shaped row which does not reach the posterior part of the lateroventral margin of the head capsule, usually mixed with straight or evenly curved hairs, and (4) J-shaped hairs arranged in a distinct row around the lateroventral margin of the the head capsule ( Table 1 View TABLE 1 , Figure 6 View FIGURE 6 ).
Across species of Veromessor , psammophore development loosely corresponds with habitat. The three species with the most poorly developed psammophores ( V. andrei , V. chicoensis , V. stoddardi ) are restricted to the more mesic California coastal ranges, the California Central Valley, and Sierra Nevada foothills. Veromessor chamberlini has a moderately well developed psammophore, and it is also restricted to more mesic southern California coastal habitats. The two other species with a moderately well developed psammophore ( V. lobognathus , V. smithi ) are widespread and inhabit cooler, higher elevation areas such as pinyon pine-juniper habitats. Two species with well developed psammophores ( V. julianus , V. pergandei ) inhabit low elevation, hot desert environs, while the other two species ( V. lariversi , V. pseudolariversi ) inhabit both low elevation, hot desert and mid-elevation, cool desert environs; all four species often occur in sandy habitats (see also Wheeler, 1907). This interspecific variation suggests that degree of psammophore development is subject to environmental pressures.
Propodeal spines
The function of propodeal spines is unclear, but the two primary hypotheses are that these spines function for defense or structural support ( Blanchard & Moreau, 2017; Sarnat, Friedman, Fischer, Lecroq-Bennet, & Economo, 2017; R. Keller, pers. comm.). Interestingly, size of the propodeal spines varies greatly across the few species of Veromessor . The spines are short (length less than to similar to distance between their bases) in V. chicoensis , V. lariversi , V. pergandei , V. pseudolariversi , V. smith , and V. stoddardi , medium length (length about 1.0–1.5× as long as the distance between their bases) in V. lobognathus , long (length> 2× as long as the distance between their bases) in V. julianus , and very long (length> 3× as long as the distance between their bases) in V. andrei and V. chamberlini ( Table 1 View TABLE 1 ).
Eye size and structure
The eyes of insects possess an incredible array of adaptations to enhance vision across the gamut of light levels that they experience. One commonly studied contrast relates to differences in eye structure between nocturnal and diurnal species, with nocturnal species typically having larger eyes and larger eye facets. Cuticular pigment level also often is correlated with level of ambient light, with species having little or no cuticular pigment typically living in dim light to lightless environments.
Eye structure and pigment level varied between nocturnal and diurnal species of Veromessor , as well as in three other ant genera ( Myrmecocystus , Aphaenogaster , Temnothorax ) ( Johnson & Rutowski, 2022). Veromessor contains two species ( V. lariversi and V. pseudolariversi ) that are pale in color, i.e., light yellowish to yellowishorange, while all other congeners are darker ( Johnson & Rutowski, 2022). Differences in pigment level correlated with foraging time—pale species mostly are strict nocturnal foragers whereas dark species usually forage diurnally, or foraging activity is temperature dependent (diurnal during cool seasons, nocturnal during hottest seasons), and several species are largely matinal-crepuscular-nocturnal foragers ( V. julianus , V. smithi , and V. stoddardi ).
As predicted, both pale species had large eyes, but they displayed a different eye structure. Veromessor pseudolariversi had larger eyes and more eye facets than V. lariversi , but mean facet diameter was similar for the two species ( Figures 30 View FIGURE 30 & 53 View FIGURE 53 ), despite their being sister species.All three variables (eye size, facet number, facet diameter) were larger for these two species than for all other dark congeners except for V. smithi . Compared to V.pseudolariversi , the eyes of V. smithi were smaller and had fewer facets, but facet diameter was similar. Compared to V. lariversi , the eyes of V. smithi were larger and facet diameter was larger, but facet number was similar ( Figure 53 View FIGURE 53 ). Moreover, eyes of the two pale species of Veromessor evolved to maximize sensitivity over resolution, which is the pattern typical for most nocturnal insects with apposition eyes ( Greiner, 2006; Stöckl, Smolka, O’Carroll, & Warrant, 2017).
Worker polymorphism and worker body size
The genus Veromessor is interesting because workers range from monomorphic to polymorphic ( Table 1 View TABLE 1 ). Most species are monomorphic ( V. andrei , V. chamberlini , V. lariversi , V. lobognathus , V. pseudolariversi , V. smithi ), but two species are weakly polymorphic ( V. julianus , V. pergandei ) and two species are polymorphic ( V. chicoensis , V. stoddardi ). All species of Veromessor that display some degree of polymorphism show monophasic allometry, which is the most primitive form of polymorphism in which growth is nonisometric (Wills, Powell, Rivera, & Suarez, 2018; Wilson, 1953). Worker polymorphism has been examined in detail only in the weakly polymorphic V. pergandei , where workers display an annual cycle of mean body size with the largest workers present during winter months ( Gordon, 1978; Rissing, 1987). Additionally, some tasks are related to worker size in V. pergandei . For example, Steatoda and Asagena spiders prey on workers of V. pergandei , but only the largest 30% of workers remove webs of these species from the foraging column, and only the largest workers retrieve nestmates that are ensnared in webs ( Kwapich & Hölldobler, 2019). Groups of polymorphic workers also dig longer nests with more branching than do groups of monomorphic workers, and larger workers build tunnels with a larger diameter ( Kwapich et al., 2018). Further studies should examine distribution of body sizes within and across colonies for all species. These studies should include seasonal and geographic variation in worker body size given the annual cycle displayed by colonies of V. pergandei (Rissing, 1987) , along with the size-frequency distribution for foragers within colonies, as graphed by Rissing and Pollock (1984).
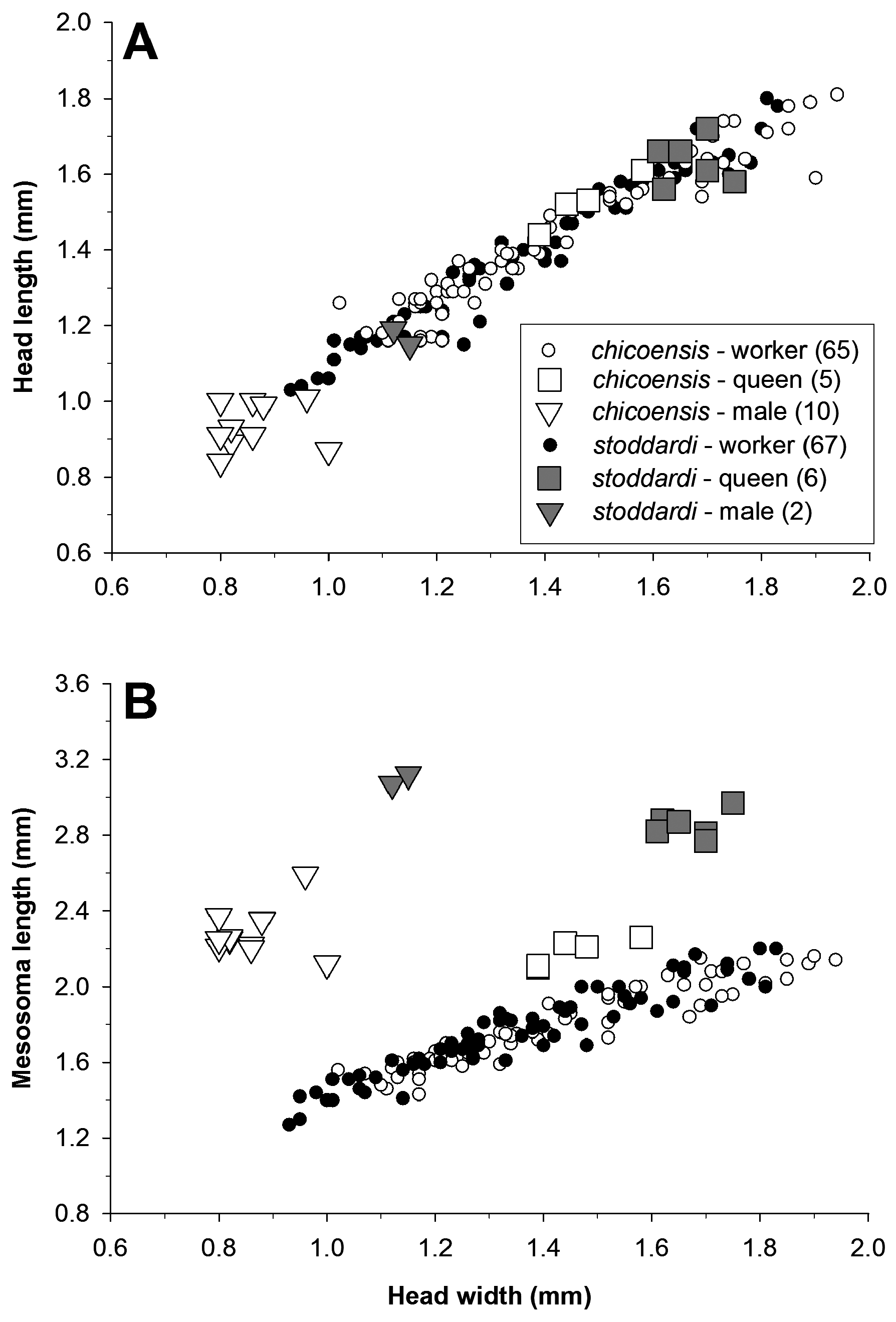
Species of Veromessor also show morphological similarities and differences within groups of ecologically similar species. The three large-colony species ( V. andrei , V. julianus , V. pergandei ) form one group of ecologically similar species, while small-colony species form three groups of ecologically similar species: (1) V. lariversi and V. pseudolariversi , (2) V. lobognathus and V. smithi , and (3) V. chicoensis and V. stoddardi . Two of the large-colony species, V. julianus and V. pergandei , have a similar allometry for head width versus head length and mesosoma length versus pronotum width. For V. andrei , the allometry for mesosoma length versus pronotum width is similar to that of V. julianus and V. pergandei , but head length in V. andrei is greater than head width for any given worker size in the other two species. Workers of V. andrei are also less variable in size than those of V. julianus and V. pergandei ( Figure 54 View FIGURE 54 ).
The ecologically similar pairs of small-colony species display two patterns. First, body size varies across groups with there being a group of smaller species ( V. lariversi , V. pseudolariversi ), a group of larger species ( V. lobognathus , V. smithi ), and a group of polymorphic species ( V. chicoensis , V. stoddardi ). The former two groups also have one species that has greater variation in body size ( Figure 55 View FIGURE 55 ). Veromessor chamberlini does not readily fit into any of these species groups.
Colony size and foraging method
Species of Veromessor display a dimorphism in colony size with three species ( V. andrei , V. julianus , V. pergandei ) having colonies that probably greatly exceed 50,000 workers, while the other seven species have colonies that typically range from several hundred to 1,000 –2,000 or more workers. All three large-colony species forage in columns that extend 10–40 m from the nest, with workers dispersing into a fan to forage individually near the end of the column (for a review see Plowes, Johnson, & Hölldobler, 2012). In contrast, all small-colony species use solitary foraging, although V. chamberlini , V. chicoensis , and V. stoddardi sometimes forage in short columns ( Table 1 View TABLE 1 ; M. Bennett, pers. comm.; R.A. Johnson, pers. obs.).
Colony size and foraging strategy also correlate with development of the pygidial gland. The three large-colony, column foraging species possess a well developed pygidial gland that consists of large paired clusters of glandular cells, large paired reservoir sacks, and a pair of distinct textured cuticle structures associated with the gland. In contrast, the pygidial gland reservoir and textured tergal cuticle structures are lacking or rudimentary in six of the seven small-colony species (not examined in V. lariversi ) ( Hölldobler et al., 2013). These two groups of species should be examined for additional differences in glands and gland chemistry.
Seasonality of mating flights
Most desert ants have summer mating flights that are triggered by rain ( Johnson, 2000b, 2001). In contrast, all species of Veromessor have mating flights that appear to be triggered by photoperiod, i.e., flights do not occur in response to rains. All species of Veromessor appear to release few sexuals per day over a period of up to several weeks. Photoperiod-triggered mating flights appear to be a derived trait given that species of Novomessor , the mostly closely related genus ( Ward et al., 2015), have mating flights triggered by summer rains ( Johnson, 2000b). Most species of Veromessor have summer mating flights (June–August), whereas those of V. julianus and V. pergandei occur during late winter–early spring ( Johnson, 2000a). The late winter–spring flights for these two species correlate with their being the only two species of Veromessor that are restricted to hot desert habitats ( Wheeler & Wheeler, 1973). Queens of both species have poor heat tolerance compared to species of Novomessor and Pogonomyrmex (see Johnson, 2000a), suggesting that Veromessor queens could not survive hot summer soil temperatures (see also Rissing, Johnson, & Pollock, 1986). Moreover, it seems probable that shifting mating flights from summer to late winter–early spring was prerequisite to these two species invading hot desert habitats ( Johnson, 2000 a, 2001). Based on these patterns, we predict that mating flights of V. sp. cf. pergandei (sexuals are unknown) occur during late winter or spring ( Table 1 View TABLE 1 ).
Mating frequency
Obligate multiple mating (effective queen mating frequency> 2) is rare in ants (but see Denny, Franks, Powell, & Edwards, 2004; Overson, Fewell, & Gadau, 2016; Villesen, Murakami, Schultz, & Boomsma, 2002; Wiernasz, Perroni, & Cole, 2004). Little is known about mating frequency in species of Veromessor , but we include this topic to highlight the need for data so as to determine if species variation correlates with life history traits such as colony size. Mating frequency is known only for V. pergandei , in which queens mate with multiple males; patriline number ranges from one to seven (mean = 3.80, n = 9 colonies) with an effective mating frequency ( M e) of 2.56 ( Kwapich et al., 2017; Ode & Rissing, 2002). Additionally, the most productive colonies of V. pergandei had significantly fewer patrilines, a larger peak forager population, and a larger annual foraging range ( Kwapich et al., 2017).
Queen size and colony founding strategy
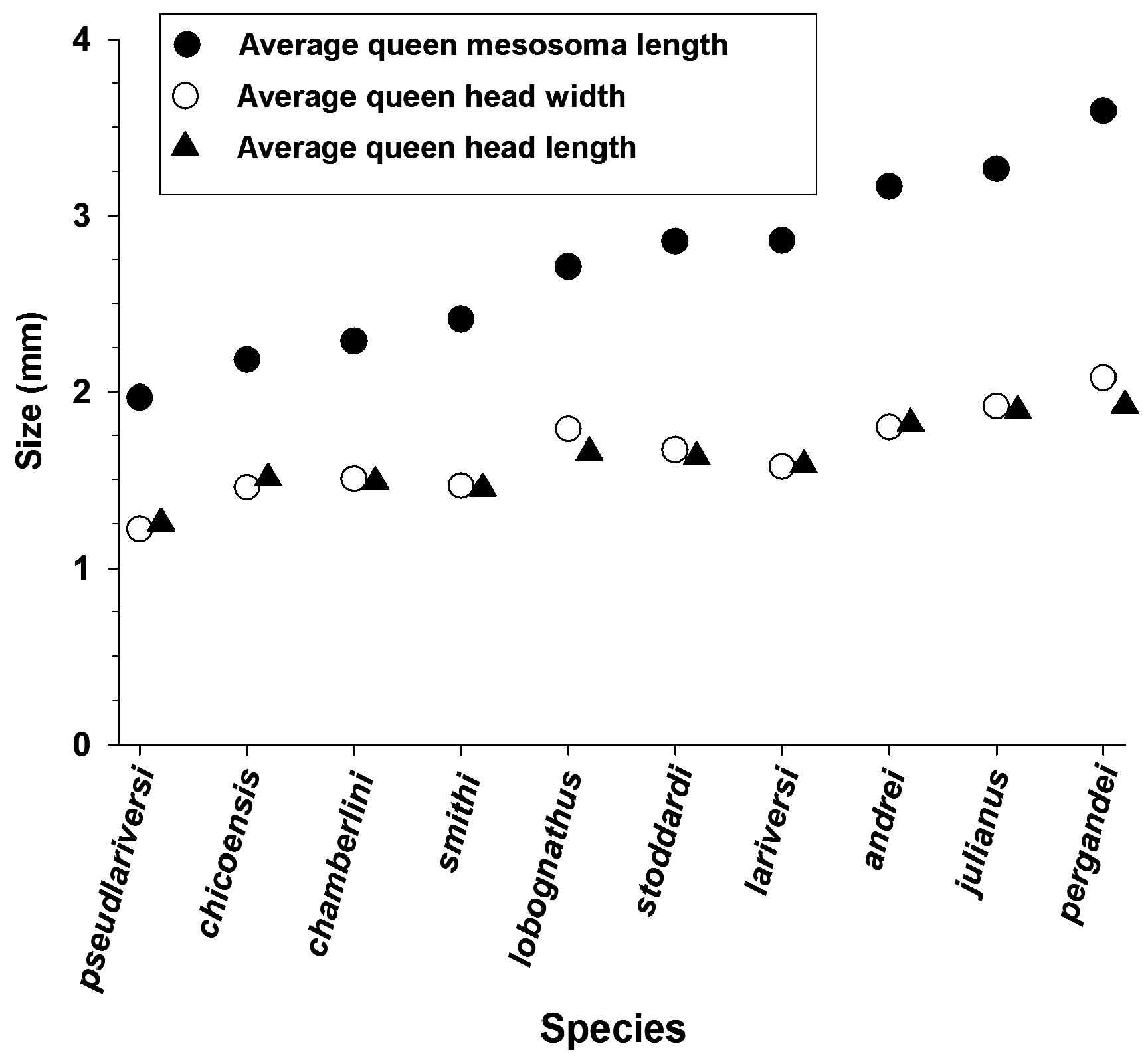
The evolution of queen size and method of colony founding is a promising research area given the wide variation in known queen founding strategies combined with the large variation in queen body size across species of Veromessor ( Figure 56 View FIGURE 56 ). The three large-colony, column foraging species have the largest queens in the genus, with an average dry mass of 20.5 mg for V. pergandei (see also Cahan & Rissing, 2005), 11.1 mg for V. julianus , and 7.5 mg for V. andrei (see above). The queens of V. julianus and V. pergandei are the largest queens in the genus, and these also are the only two species that are restricted to hot desert habitats ( Wheeler & Wheeler, 1973), where increased body size can increase longevity under desiccating conditions ( Johnson, 1998, 2021; Wiernasz & Cole, 2003). At the low end of the scale, size of V. chicoensis queens is similar to that of their workers ( Figure 19 View FIGURE 19 ), and undoubtedly this species has the lowest queen to worker dimorphism in the genus. The queens of V. smithi are also relatively small, with a size similar to or only slightly larger than that of workers (workers: HW = 1.33–1.61 mm, HL = 1.33–1.58 mm, ML = 1.76–2.07 mm; queens: HL = 1.40–1.63 mm, HW = 1.40–1.63 mm, ML = 2.06–2.38 mm). A similar range of queen body size also occurs in Pogonomyrmex , where queens of P. schmitti are similar in size to their workers (Johnson, 2015; Johnson & Cover, 2015).
Little is known about colony founding strategies for species of Veromessor . All species of Veromessor probably have independent colony founding, but this has only been documented in V. andrei , V. julianus , and V. pergandei . Veromessor julianus is haplometrotic ( Johnson, 2000a), whereas V. pergandei displays geographic variation in founding strategy (see above). The larger queens of V. pergandei are fully claustral, while the smaller queens of V. andrei appear to be semi-claustral with obligate queen foraging based on their absence of storage proteins ( Brown, 1999b). Nothing is known about colony founding for queens of small-colony species. However, if body size correlates with colony founding strategy, then queens for most to all small-colony species are predicted to be semi-claustral obligate foragers. For example, the very small size for queens of V. chicoensis , especially relative to worker size, suggests that this species is semi-claustral with obligate queen foraging. We also note that queens of V. pseudolariversi are very small ( Figure 56 View FIGURE 56 ), but they are much larger than their workers compared to the queens of V. lobognathus and V. smithi . Interestingly, V. lariversi and V. pseudolariversi have similar geographic ranges and are sympatric at several sites, but the queens differ in size with those of V. pseudolariversi being the smallest in the genus, while those of V. lariversi are the largest among small-colony species ( Figure 56 View FIGURE 56 ). Additional studies should examine colony founding strategies and variation in queen body size. For most of these species, it is doubtful that sufficient numbers of founding queens could be located to experimentally test founding strategy (e.g., Johnson, 2002, 2006). However, founding strategy might be inferred by comparing storage protein levels for alate queens across species (see Hahn, Johnson, Buck, & Wheeler, 2004).
Distribution patterns
Veromessor is mostly restricted to western North America with species inhabiting arid and semi-arid habitats, grasslands, sclerophyll woodlands, and desert scrublands. Distribution patterns of Veromessor are discussed in two groups that correspond to colony size—species that have large colonies (>50,000 workers) and species that have small colonies (usually < 1,000 –2,000 workers) (see also Johnson, 2000b). The three large-colony species ( V. andrei , V. julianus , V. pergandei ) have mostly parapatric distribution patterns with their ranges showing little to no overlap across the narrow contact zones that separate the species (herein we define parapatric as species that occupy separate but adjoining areas, such that only a small fraction of individuals in each species encounter the other species) (see Figure 1 View FIGURE 1 , pg 83 in Johnson, 2000b). Veromessor julianus is restricted to central and southern parts of the Baja California peninsula in Mexico, as well as a narrow band along cool Pacific coastal areas north to Bahía San Quintin ( Johnson, 2000 a, 2000b) ( Figure 27A View FIGURE 27 ). Alternatively, V. pergandei occurs in the more xeric desert areas along the east coast of the peninsula south to northern Baja California Sur. Where these two species come in contact, V. pergandei occurs in more xeric microhabitats, i.e., in drier soils and/or at lower elevations ( Johnson, 2000a). The different micro- and macrohabitats occupied by V. julianus and V. pergandei correlate with queen body size as queens of V. pergandei are larger than those of V. julianus (see also Wiernasz & Cole, 2003).
Veromessor julianus and V. andrei contact each other only near Bahía San Quintin, Baja California, Mexico ( Figures 10A View FIGURE 10 , 27A View FIGURE 27 ). Veromessor pergandei and V. andrei rarely coexist as the latter species inhabits the cool foothills of the California coastal range and inland valleys from northwestern Baja California to southern Oregon. In contrast, V. pergandei inhabits hot desert areas to the east and only occasionally penetrates to the adjacent coastal valleys, but rarely above elevations of 1,100 –1,200 m (see also Johnson, 2000 a, 2000b); there are also several records for V. pergandei in hot coastal areas of California such as in the Central Valley ( Figure 41A View FIGURE 41 ).
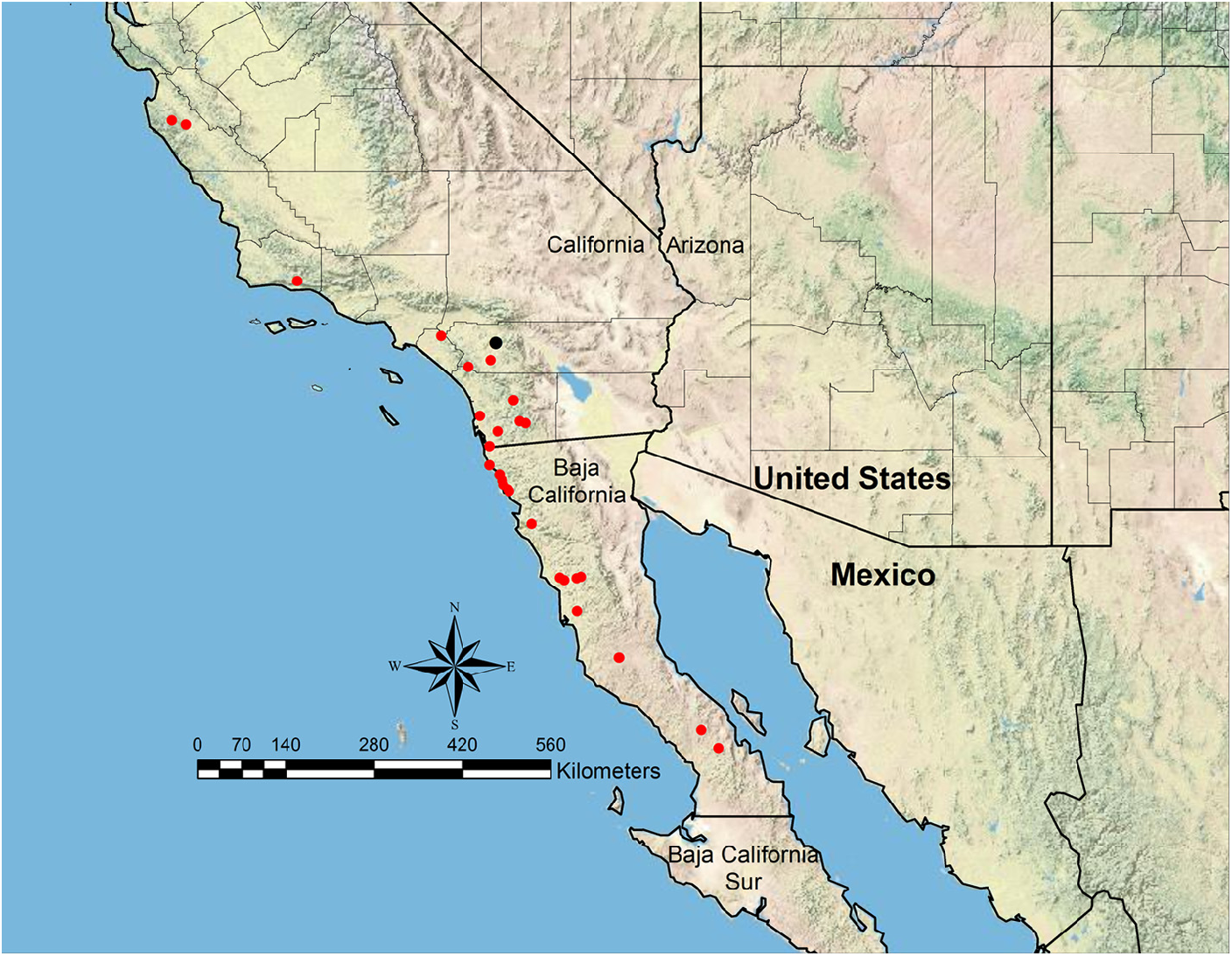
The other seven species of Veromessor have relatively small colonies with number of workers ranging from several hundred to 1,000 –2,000 or more. Distribution patterns of these species can be divided into three groups: (1) three California species ( V. chamberlini , V. chicoensis , V. stoddardi ) that occur in non-desert areas of the California coastal range west of the Sierra Nevada from northern to southern California, with one species extending into central Baja California, (2) four broadly sympatric species ( V. lariversi , V. lobognathus , V. pseudolariversi , V. smithi ) that occur east of the Sierra Nevada in California and are most common in Nevada, with ranges sometimes extending into Arizona and adjacent states, and (3) one species ( V. sp. cf. pergandei ) that appears to be restricted to sand dunes of hot desert habitats in southwestern Arizona and probably northwestern Sonora, Mexico.
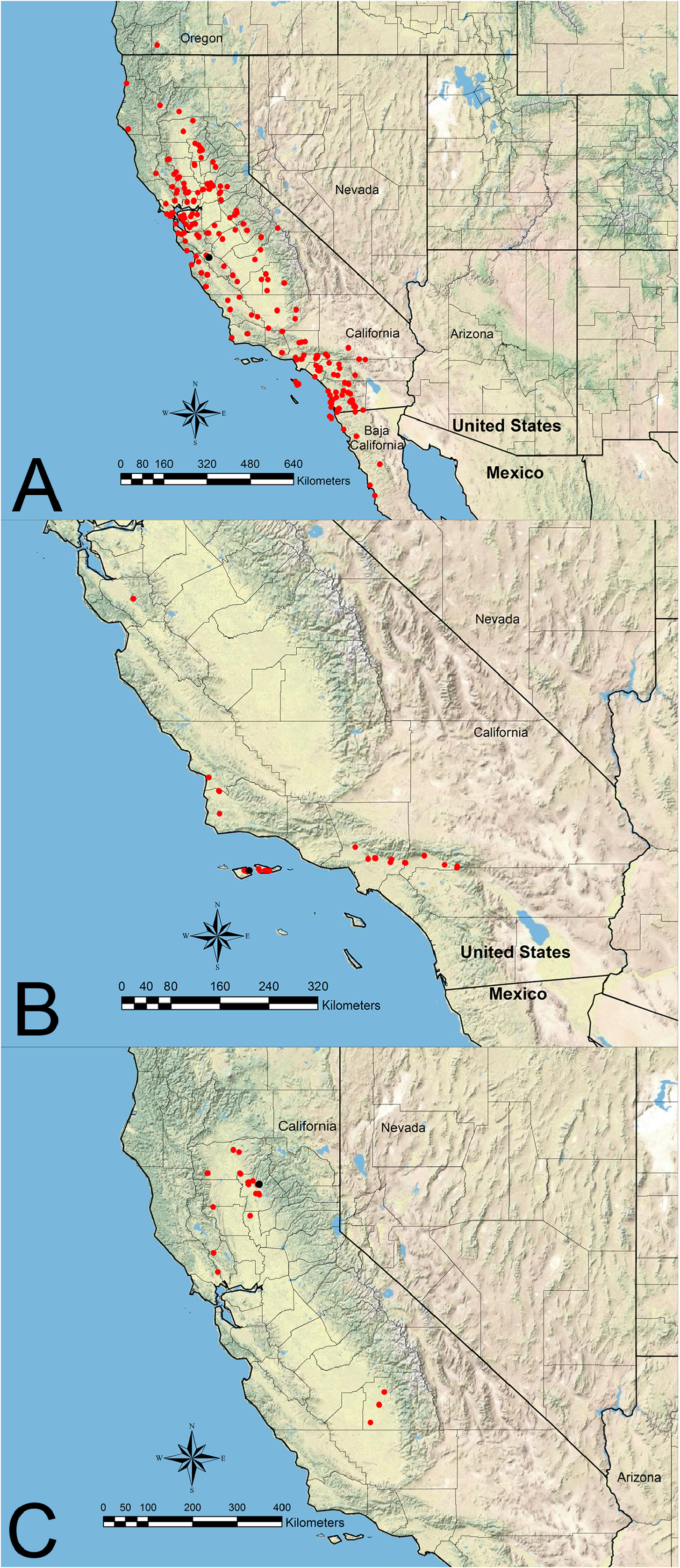
The first group consists of three species ( V. chamberlini , V. chicoensis , V. stoddardi ) that are restricted to California and Baja California. Veromessor chicoensis is geographically isolated from the other two species as it is restricted to the Sacramento and San Joaquin Valleys of northern and central California ( Hamm & Kamansky, 2009) ( Figure 10C View FIGURE 10 ). Veromessor stoddardi is most closely related to V. chicoensis (M.L. Borowiec, unpub. data), and these two species have a mostly contiguous distribution with V. chicoensis in northern coastal range areas, while V. stoddardi occurs in southern coastal range areas, with the nearest locales being about 170 km apart ( Figures 10C View FIGURE 10 , 52 View FIGURE 52 ). Veromessor chamberlini occurs on Santa Cruz and Santa Rosa Islands and across locales along the southern base of the San Gabriel Mountains and in coastal areas north to near the San Francisco Bay area ( Figure 10B View FIGURE 10 ).
The four species in the second group occur in California east of the Sierra Nevada and have broadly sympatric distributions. These species form two groups based on habitat of occurrence. Veromessor lobognathus and V. smithi typically inhabit mid-elevation, pinyon pine–juniper habitats, rocky bajadas, and buttes. Both species are sympatric at some locales ( Figures 27C View FIGURE 27 , 41C View FIGURE 41 ), but it is not known if these two species have different microhabitat preferences. Alternatively, V. lariversi and V. pseudolariversi typically inhabit high desert habitats, often in sandy soils. Both species have a similar geographic distribution and both species are sympatric at some sites ( Figures 27B View FIGURE 27 , 41B View FIGURE 41 ). It is not known if these two species have different microhabitat preferences.
The final species, V. sp. cf. pergandei , is only known from one hot desert habitat locale in sand dunes of southwestern Arizona.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
SubFamily |
Myrmicinae |