Geoplana quagga Marcus, 1951, 2017
|
publication ID |
https://doi.org/10.11646/zootaxa.4362.1.5 |
|
publication LSID |
lsid:zoobank.org:pub:F72F750D-563E-4960-BAB3-CBEE139A288C |
|
DOI |
https://doi.org/10.5281/zenodo.6042422 |
|
persistent identifier |
https://treatment.plazi.org/id/0397BB6C-FFF3-FFD2-BDBE-972DC8EAFC08 |
|
treatment provided by |
Plazi |
|
scientific name |
Geoplana quagga Marcus, 1951 |
| status |
incertae sedis |
Geoplana quagga Marcus, 1951 incertae sedis
( Figs. 7–8 View FIGURE 7 View FIGURE 8 , Tables 3, 4)
Material examined. MLP – He 7412, San Antonio town, 8 May 2008; cephalic and anterior region at the level of the ovaries: sagittal sections on 37 slides; pre-pharyngeal region: transverse sections on 11 slides; pharynx and copulatory apparatus: sagittal sections on 31 slides . MLP – He 7411-1, San Antonio town, 7 May 2008; cephalic and anterior region at the level of the ovaries: sagittal sections on 13 slides; pharynx and copulatory apparatus: sagittal sections on 15 slides . MLP – He 7411-2, San Antonio town, 7 May 2008; preserved in ethanol.
Locality. San Antonio town ( 26°03’17.25’’S, 53°43’50.29’’W), Misiones province, Argentina ( Fig. 1 View FIGURE 1 ). GoogleMaps
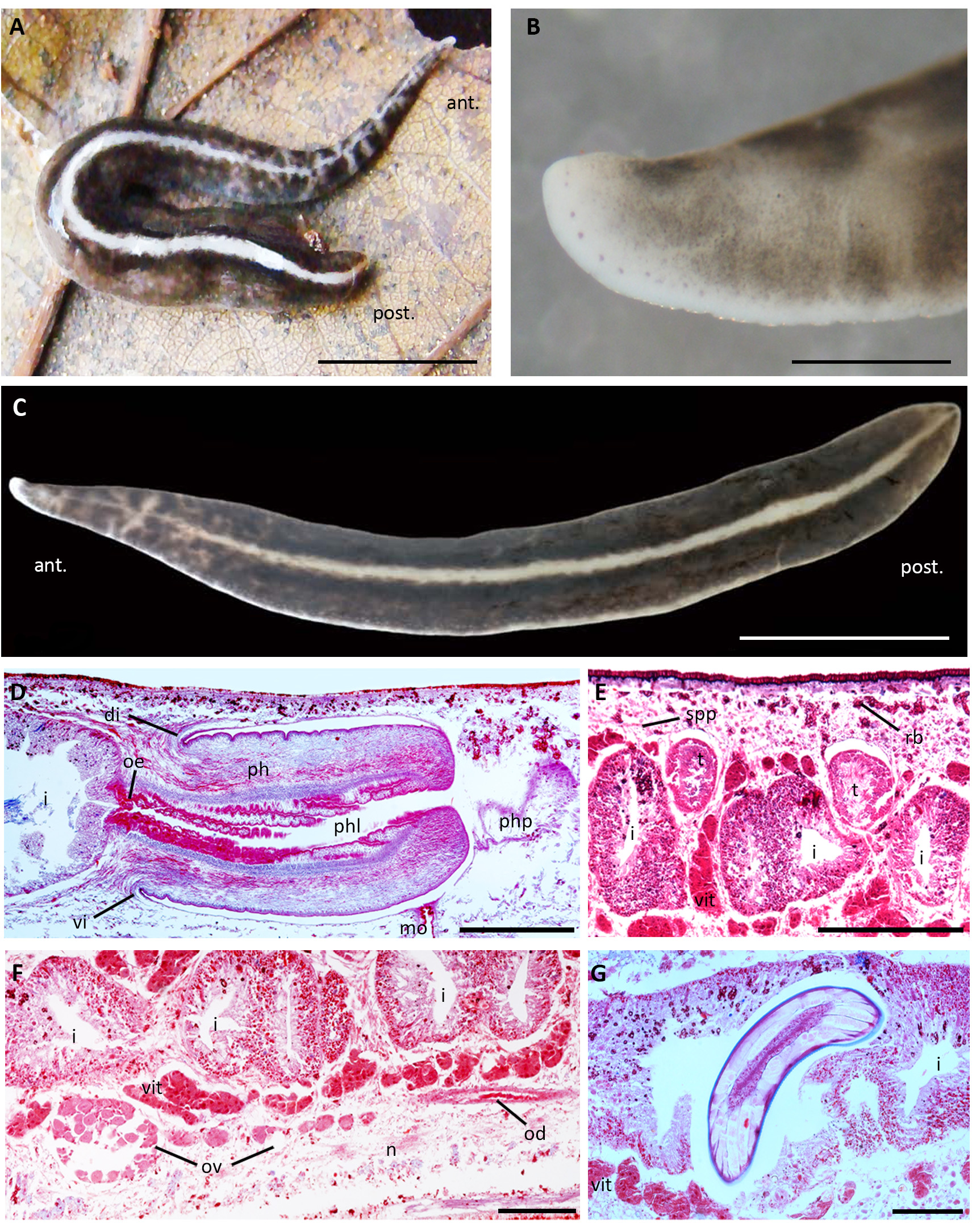
Description. External morphology. Body elongate, with parallel margins. When crawling, the first quarter of the body is elongated and much thinner than the rest of the body, with the anterior tip blunt and the posterior region ending abruptly ( Fig. 7A, B View FIGURE 7 ). The dorsal surface is black grey with a pure white median stripe flanked by jet black para-median stripes formed by very dense pigment on each side of the median stripe ( Fig. 7A, C View FIGURE 7 ). Along the first quarter of the body, the median band is very thin and traversed by narrow pure white irregular stripes ( Fig. 7A, C View FIGURE 7 ). The ventral surface and body margins are light grey. The eyes, with small clear halos, are exclusively marginal. They are uniserial throughout the body, except in the middle third, where they are irregularly bi-serial ( Fig. 7B, C View FIGURE 7 ). When crawling, maximum length was ~ 30mm, whereas after fixation was 20.5–22.6mm. Maximum width was 3– 3.4mm. The mouth and gonopore distances from the anterior tip are 65–74% and 78–83% respectively relative to body length.
Internal morphology. Rhammites cover the entire dorsal epidermis (20µm high), which receives fine granular erythrophil and cyanophil secretions. The ventral epidermis (25µm high), which is ciliated on the creeping sole (~95% of body width), receives the same types of secretions, being cyanophil secretions more abundant than erythrophil ones. Small rhabdites are present along the creeping sole. Glandular margin with coarse granular erythrophil secretion and abundant rhammites. Sensory pits (50µm deep), as simple invaginations of ventral epidermis, contour the cephalic region.
Cutaneous musculature composed of a subepidermal circular layer (2.5–5µm thick), an intermediate diagonal layer (5–10µm thick), and an inner longitudinal layer (25–50µm thick). The cutaneous musculature occupies 5–9% of the body height (CMI). Parenchymal musculature constituted by a dorsal layer, with decussate fibres, followed by a transverse supra-intestinal and a sub-intestinal muscle layer. Also, scarce dorso-ventral fibres intermingle among intestinal branches. The parenchymal musculature occupies 7–12% of the body height.
The cylindrical pharynx, 1.3–1.5mm in length (6–7% of body length), occupies most of the pharyngeal pouch ( 1.6–1.7mm in length), with the mouth located at the distal third of the latter ( Fig. 7D View FIGURE 7 ). Pharynx lined by ciliated cuboidal epithelium, followed by a longitudinal subepithelial muscle layer (5µm thick) and a subjacent circular layer (5–10µm thick). Pharyngeal lumen lined by ciliated columnar epithelium, followed by a single muscle layer composed mainly of circular fibres with some longitudinal fibres intermingled (40–70µm thick). Pharyngeal stroma pierced by highly abundant fine granular erythrophil and cyanophil secretions which open in the pharyngeal tip ( Fig. 7D View FIGURE 7 ). A short oesophagus (~100µm in length) connects the pharyngeal lumen with the intestine.
(-) not measured. (*) distal portion of prostatic vesicle. (+) eversible penis.
Dorsal testes located below the supra-intestinal parenchymal muscle layer ( Fig. 7E View FIGURE 7 ), constituting two irregular rows on each side of the body. They are posterior to the ovaries, extending until the proximity of the pharyngeal root (19–25% and 55–64% of body length from the anterior end, respectively). At pre-pharyngeal level, sperm ducts are dorsal and laterally displaced to the ovovitelline ducts, located among fibres of the sub-intestinal parenchymal muscle layer. Behind the pharynx, they are expanded and full of spermatozoa, forming spermiducal vesicles. Their distal portions bend medially to open laterally into the extrabulbar prostatic vesicle, which is Cshaped in sagittal view ( Fig. 8A–C View FIGURE 8 ). Then, the prostatic vesicle penetrates the antero-dorsal face of the penis bulb and communicates with the ejaculatory duct, which runs obliquely to reach the centre of the bulb ( Fig. 8A, B View FIGURE 8 ). The ejaculatory duct opens in a sinuous duct of the male atrium, which is formed by their folded walls ( Fig. 8A, B, D View FIGURE 8 ). These folds of the atrium project backward forming a kind of papilla (eversible penis) (450–550µm long) ( Fig. 8A, B View FIGURE 8 ). The male atrium (500–600µm long) houses this papilla in its entire cavity, which is well separated from the female atrium by a dorsal fold located at the level of the gonopore ( Fig. 8A, B View FIGURE 8 ). Sperm ducts lined by ciliated cuboidal epithelium, without underlying musculature at the pre-pharyngeal level. The extrabulbar prostatic vesicle is lined with a ciliated, columnar epithelium, pierced by erythrophil fine granules very densely arranged and intensely stained ( Fig. 8B, C View FIGURE 8 ), and surrounded by a thick muscle layer mainly composed of circular fibres with interspersed oblique and longitudinal fibres (65–80µm thick). The ejaculatory duct, whose lumen is more dilated than the prostatic vesicle, is also lined with a ciliated, columnar epithelium, which receives fine granular erythrophil secretion, in less quantity compared with the prostatic vesicle ( Fig. 8B–D View FIGURE 8 ). The ejaculatory duct is wrapped by a muscle layer arranged as the prostatic vesicle but thinner (25–30µm thick). The cell bodies of the glands which pierce the ejaculatory duct are in the surrounding parenchyma, outside the penis bulb. The male atrium is lined by columnar epithelium, followed by a circular and a longitudinal muscle layer, being thicker dorsally (25–30µm thick) than ventrally (10µm thick). The atrial epithelium is filled with scarce fine granular erythrophil secretion discharged by cell glands which lie below its muscularis. The lining epithelium of the male atrial folds is columnar, non-ciliated and strongly stained because it is crossed by abundant erythrophil fine granules ( Fig. 8B, D, E View FIGURE 8 ), whose cell bodies lie immediately below the circular muscle layer (5–10µm thick) and a subjacent longitudinal layer (25–30µm thick). Also, abundant cell necks with fine erythrophil granules traverse the atrial folds and open in the ectal region of the papilla ( Fig. 8B, E View FIGURE 8 ). The cell bodies of these glands are located outside the bulb ( Fig. 8A, B View FIGURE 8 ).
Ovaries proximally ovoid and distally elongate, drop-shaped, ventral to the intestine, located just above the nervous plate ( Fig. 7F View FIGURE 7 ). They appear at 3.4–5mm from the anterior tip (15–24% of body length). Ovovitelline ducts originate dorsally from the ovoid part of the ovaries and run backwards. Numerous vitelline follicles, which discharge throughout the entire length of the ovovitelline ducts, accommodate among intestine branches ( Fig. 7E– G View FIGURE 7 ). At the level of the gonopore, the ovovitelline ducts gradually ascend and turn towards the sagittal body plane and join in a short common glandular ovovitelline duct (~100µm long) located above the posterior region of the female atrium ( Fig. 8A, B View FIGURE 8 ). The female genital canal (250–300µm in length), projecting dorso-anteriorly, connects the common ovovitelline duct with the female atrium, whose proximal part is funnel-shaped ( Fig. 8A View FIGURE 8 ). The rest of the atrium is a poorly folded ample cavity (450–500µm long) ( Fig. 8A, B View FIGURE 8 ). The ovovitelline ducts are lined with cuboidal ciliated epithelium surrounded by a thin longitudinal muscle layer (2.5µm thick). Their distal ascending portions are pierced by amorphous cyanophil secretion from the shell glands, which also discharge into the common ovovitelline duct. This common duct is lined with columnar ciliated epithelium continuous with the female canal, but in the latter is non-ciliated. Both the common duct and female canal are mainly wrapped by circular muscle fibres with some oblique ones (10–15µm thick). The epithelium of the female atrium is pseudostratified, with nuclei at different heights, pierced by abundant erythrophil fine granules ( Fig. 8B View FIGURE 8 ). Its muscularis is composed of circular fibres with some interspersed longitudinal and oblique fibres, thicker dorsally (120–150µm thick) than ventrally (40–75µm thick). The gonopore canal is lined with a columnar ciliated epithelium, filled with erythrophil fine granules densely grouped apically ( Fig. 8B View FIGURE 8 ).
Parasitism. Nematode larvae were found in the specimen MLP-He 7412 inside the parenchyma, near the copulatory apparatus ( Fig. 7G View FIGURE 7 ).
Remarks and comparative discussion. The anatomical features of specimens from San Antonio ( Argentina) are coincident with the original description of Geoplana quagga provided by Marcus (1951) from material collected in São Paulo ( Brazil). This species is easily distinguished from other geoplaninids due to its characteristic colour pattern, consisting of white transversal stripes irregularly arranged on both sides of an also white median band, and a black background. These transversal stripes occupy the anterior region, approximately one-fifth of the body length in live specimens. The eyes pattern of Argentinean specimens also agrees with that of the Brazilian specimens, being mostly uniserial along the body and marginally arranged ( Marcus 1951). Besides, the body size and relative position of the mouth and gonopore regarding body length are quite similar between Argentinean and Brazilian flatworms.
Geoplana quagga is now considered as a species incertae sedis because the diagnosis of the genus Geoplana View in CoL was emended ( Carbayo et al. 2013). Some features of G. quagga , mainly regarding the reproductive system, are incongruent with the re-definition of Geoplana View in CoL , namely: prostatic vesicle intrabulbar, penis papilla protrusible extending even along the female atrium, male atrium not folded, ascending portion of the ovovitelline ducts lateral to female atrium ( Carbayo et al. 2013). In G. quagga , the reproductive system exhibit, among other features, extrabulbar prostatic vesicle, eversible penis (no permanent penis papilla) formed by folds of the male atrium, and ovovitelline ducts ascending at the level of gonopore. Although G. quagga does not belong to the genus Geoplana View in CoL , we decided to maintain it in this genus until new evidence supports their inclusion in an appropriate one.
Although the description of the reproductive system and the reconstruction of the copulatory apparatus of G. quagga sketched by Marcus (1951) fit well with our specimens, we found some small differences from the histological point of view. In this regard, Marcus pointed out the presence of a long ejaculatory duct which starts outside the penis bulb with a narrow lumen, and follows inside the bulb proximally as a dilated canal (obliquely oriented) and distally with a narrow lumen and highly sinuous, before opening on the tip of the penis papilla. Based on our specimens, we have interpreted that the ejaculatory duct corresponds only with the dilated, obliquely oriented, part mentioned by Marcus. Besides, the distal sinuous and narrow portion corresponds with the narrow lumen left by the folds of the male atrium, which take the form of a papilla (eversible penis).
Regarding the female reproductive system, according to Marcus (1951), the female atrium is an ample cavity with folded walls, but in our specimens, it is poorly folded.
The finding of Geoplana quagga in Argentina ( Misiones province) is the first report of this species outside Brazil, thus expanding its distribution range. Previous reports place this species in the states of São Paulo, Rio de Janeiro and Santa Catarina ( Marcus 1951; Froehlich 1956a, 1959) ( Fig. 1 View FIGURE 1 ). However, because the Argentinean specimens were found in proximity to the frontier with the state of Paraná ( Brazil), the presence of G. quagga in this state is highly expected. Froehlich (1955a) catalogued G. quagga as a ‘man-follower species’ because it has always been found in anthropized areas, both in cities (gardens, backyards, vacant lots) and peri-urban areas (near houses) ( Marcus 1951; Froehlich 1956a, 1959). Similarly, the specimens from San Antonio were collected under fallen logs in a man-disturbed riparian native forest, near human settlements.
Although Marcus (1951) did not give an indication or explanation concerning the origin of the species name, we believe that it could have derived from an extinct subspecies of plains zebra, Equus quagga quagga , an equid that lived in South Africa until the 19th century. This equid was distinguished from other zebras by its limited pattern of white stripes mainly on the anterior part of the body ( Skinner & Chimimba 2005). This striped pattern may have reminded Marcus (1951) of this extinct animal, thereby giving the species name to this land planarian.
| MLP |
Museo de La Plata |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Geoplana quagga Marcus, 1951
| Negrete, Lisandro & Brusa, Francisco 2017 |
Geoplana quagga
| Negrete & Brusa 2017 |
G. quagga
| Negrete & Brusa 2017 |
Geoplana quagga
| Negrete & Brusa 2017 |
G. quagga
| Negrete & Brusa 2017 |
Geoplana
| Stimpson 1857 |
Geoplana
| Stimpson 1857 |
Geoplana
| Stimpson 1857 |