Imbira negrita, Negrete & Brusa, 2017
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4362.1.5 |
|
publication LSID |
lsid:zoobank.org:pub:F72F750D-563E-4960-BAB3-CBEE139A288C |
|
DOI |
https://doi.org/10.5281/zenodo.6042420 |
|
persistent identifier |
https://treatment.plazi.org/id/0397BB6C-FFF9-FFCD-BDBE-92CAC8E5FC08 |
|
treatment provided by |
Plazi |
|
scientific name |
Imbira negrita |
| status |
sp. nov. |
Imbira negrita sp. nov.
( Figs. 4–6 View FIGURE 4 View FIGURE 5 View FIGURE 6 , Tables 1, 2)
Type material. Holotype. MLP–He 7415, Moconá Provincial Park, 6 November 2015; cephalic region: transversal sections on 21 slides; anterior region at the level of the ovaries: sagittal sections on 46 slides; pre-pharyngeal region: transverse sections on 6 slides; pharyngeal region: sagittal sections on 30 slides; post-pharyngeal region: sagittal sections on 17 slides; copulatory apparatus: sagittal sections on 25 slides. Paratype 1, MLP–He 7416-1, Moconá Provincial Park, 6 November 2015; cephalic region: transversal sections on 17 slides; anterior region at the level of the ovaries: sagittal sections on 24 slides; pre-pharyngeal region: transverse sections on 6 slides; pharyngeal region: sagittal sections on 13 slides; copulatory apparatus: sagittal sections on 12 slides. Paratype 2, MLP–He 7416-2, Moconá Provincial Park, 6 November 2015; cephalic region: transversal sections on 15 slides; anterior region at the level of the ovaries: sagittal sections on 21 slides; pre-pharyngeal region: transverse sections on 6 slides; pharynx and copulatory apparatus: sagittal sections on 16 slides. Paratype 3, MLP–He 7417, Moconá Provincial Park, 8 October 2016; preserved in 70% ethanol.
Diagnosis. Elongated body with parallel margins; 50–110mm in length in maximum extension; dorsal surface jet black and ventral surface ivory; eyes marginal along the body; short female canal opening in the postero-dorsal portion of the female atrium; epithelial lining of female atrium with stratified appearance.
Type locality. Moconá Provincial Park (27°08’56’’S, 53°53’54’’W), Misiones province, Argentina ( Fig. 1 View FIGURE 1 ). GoogleMaps
Etymology. The species name alludes to the black pigmentation of the dorsum (black=negra/o in Spanish), since in Argentina and other south American Spanish-speaking countries, negrita is a colloquial way to call someone or something black or very obscure.
Description. External morphology. Body elongate, with parallel margins, and flattened in cross section ( Fig. 4A–C View FIGURE 4 ). Anterior tip blunt, and posterior body region gradually sharpening, with pointed end ( Fig. 4A View FIGURE 4 ). Dorsal surface jet black and ventral surface ivory, only whitish around the mouth and gonopore ( Fig. 4A View FIGURE 4 ). After fixation, dorsal ground colour black grey and ventral surface oyster white. Eyes, with small clear halos, exclusively marginal, being uniserial throughout the body, except in the middle third, where they are irregularly bi-serial. When crawling, the maximum length of the largest specimen was 110mm, whereas that after fixation was 90.5mm. The maximum length of the smallest specimen was 36.4mm after fixation ( Table 1). Maximum width ranged between 2.9mm and 5.3mm, whereas maximum height ranged between 1 and 1.6mm. The mouth and the gonopore are located at 71–81% (mean: 76%) and 88–93% (mean: 90%) from the anterior tip, respectively ( Table 1).
Internal morphology. Sensory pits (30–50µm deep), as simple invaginations of ventral epidermis, contour the anterior tip and extend along the body margins in a single irregular row until ~ 5mm from the anterior end ( Fig. 4B View FIGURE 4 ). Sensory pits at intervals of 30–40µm near anterior tip, posteriorly becoming more gradually spaced. At the prepharyngeal region, the ventral epidermis (25–40µm high), which is ciliated on the creeping sole (~100% of body width at the pre-pharyngeal region), receives fine granular cyanophil secretion (more abundant in the ventro-lateral margins of the body), fine and coarse granular erythrophil secretions, and coarse granular xanthophil secretion. Cell bodies of these secretions located in the subjacent parenchyma, below cutaneous musculature ( Fig. 4C, E View FIGURE 4 ). Additionally, small rhabdites are highly abundant. The same types of secretory cells discharge through the dorsal epidermis (20–25µm high), with rhammites covering the whole dorsal epidermis ( Fig. 4C, D View FIGURE 4 ). There is no glandular margin. The dorsal and ventral epidermis of the cephalic region receives the same types of secretory cells as the pre-pharyngeal region, except xanthophil secretion. The creeping sole of the cephalic region occupies ~85% of the body width.
Cutaneous musculature composed of a subepidermal circular layer (2.5–5µm thick), an intermediate diagonal layer (5–15µm thick), and an inner longitudinal layer (50–60µm thick) ( Fig. 4D, E View FIGURE 4 ). The thickness of the cutaneous musculature occupies 9–12% of the body height ( Table 1). Parenchymal musculature constituted by a dorsal, not well organized, layer with oblique and some longitudinal fibres, followed by a transverse supra-intestinal and a sub-intestinal muscle layer ( Fig. 4C–E View FIGURE 4 ). Longitudinal fibres (20–30µm thick) accommodate immediately above the sub-intestinal layer ( Fig. 4E View FIGURE 4 ). Also, dorso-ventral fibres are arranged among intestinal branches ( Fig. 4D View FIGURE 4 ). The parenchymal musculature occupies 5–7% of the body height ( Table 1). In the cephalic region, and even at the level of the ovaries, parenchymal muscles exhibit the same arrangement as the pre-pharyngeal region, but longitudinal fibres are more abundant. They form discrete bundles (30–45µm thick) above the sub-intestinal muscle layer and below it mainly towards the body margins ( Figs. 4B View FIGURE 4 , 6F, G View FIGURE 6 ). Numerous parenchymal muscle fibres variously oriented traverse the nervous plate along the cephalic region ( Fig. 4B View FIGURE 4 ).
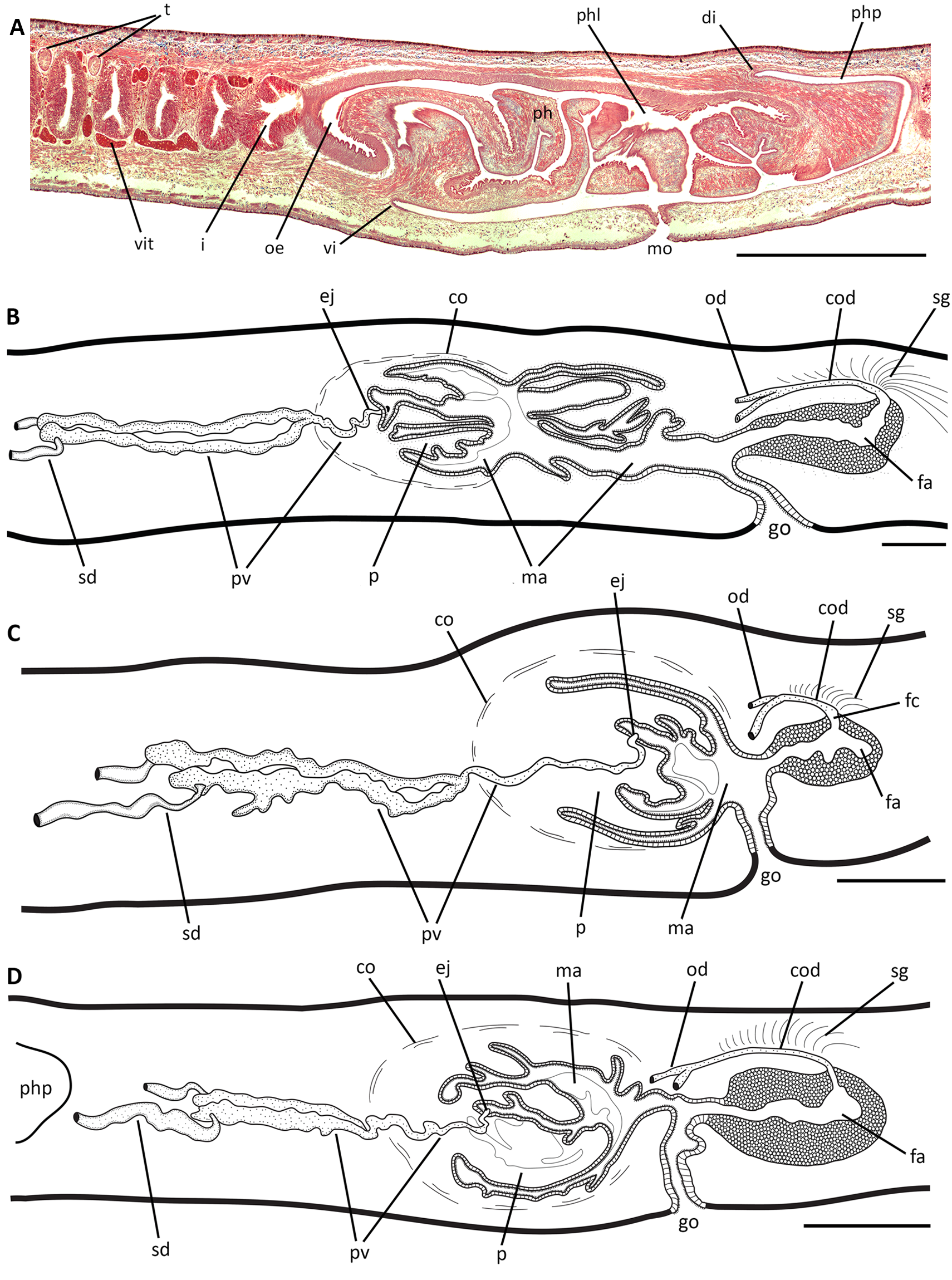
The pharynx is collar-type, 2.6–5.1mm in length (6–7% of body length), occupying most of the pharyngeal pouch (2.3–6.5mm in length) ( Fig. 5A View FIGURE 5 ). Dorsal insertion is 1.4–3.5mm posteriorly displaced ( Fig. 5A View FIGURE 5 ). The pharynx is lined by ciliated cuboidal epithelium, followed by a longitudinal muscle layer (2.5–5µm thick) and a subjacent circular muscle layer (5–10µm thick). Pharyngeal lumen lined with ciliated columnar epithelium, followed by a circular subepithelial muscle layer (50–75µm thick) and a subjacent longitudinal muscle layer (5– 10µm thick). Two types of secretory cells, whose cell bodies are located in the surrounding parenchyma anterior and lateral to the pharynx, discharge through the pharyngeal epithelium: highly abundant cells with fine granular erythrophil secretion and abundant fine granular cyanophil secretion ( Fig. 5A View FIGURE 5 ). Oesophagus (0.4–1mm in length) lined with ciliated columnar epithelium, followed by a subepithelial circular muscle layer (100–130µm thick) and a thin subjacent longitudinal muscle layer (5–15µm thick). The oesophagus:pharynx ratio is 15–21%.
The dorsal mature testes are ovoid in shape, arranged in one or two irregular rows on each side of the body. They are located below the supra-intestinal parenchymal muscle layer ( Fig. 4C, D View FIGURE 4 ), extending from behind the ovaries to near the pharynx (17–33% and 67–76% of body length from the anterior tip, respectively) ( Table 2). They occupy 18–20% of the body height at the pre-pharyngeal region. Sperm ducts, dorsally and laterally displaced from the ovovitelline ducts, located among fibres of the sub-intestinal parenchymal muscle layer ( Fig. 4C, E View FIGURE 4 ). Laterally and behind the pharynx, sperm ducts are slightly dilated and full of spermatozoa. Their distal courses turn to the sagittal plane to open ventro-laterally into the forked portion of the extrabulbar prostatic vesicle ( Figs. 5B–D View FIGURE 5 , 6A View FIGURE 6 ). These long tubular paired portions (1–2.2mm long) run laterally sinuous and continue as an unpaired intrabulbar portion whose course is also tortuous (0.4–0.7mm long) ( Figs. 5B–D View FIGURE 5 , 6B–E View FIGURE 6 ). Very close before opening into the male atrium, the prostatic vesicle connects with the ejaculatory duct, which is a short canal with a narrow lumen (150–300µm long) ( Figs. 5B–D View FIGURE 5 , 6C, E View FIGURE 6 ). Male atrium (0.7–2.5mm long), with highly folded walls, leaving a reduced lumen inside the cavity ( Figs. 5B–D View FIGURE 5 , 6B–E View FIGURE 6 ). The folded walls of its proximal part, where the ejaculatory duct opens, conform a kind of papilla: the eversible penis ( Figs. 5B–D View FIGURE 5 , 6B–E View FIGURE 6 ). Only the male atrium of the holotype exhibits dorsal folds posteriorly to the eversible penis ( Figs. 5B View FIGURE 5 , 6B, C View FIGURE 6 ).
Sperm ducts lined with ciliated cuboidal epithelium, without underlying musculature at pre-pharyngeal level. Epithelial lining of the prostatic vesicle, both paired and unpaired portions, columnar and ciliated, filled with abundant erythrophil fine granular secretion. Cell bodies of prostatic glands located in the surrounding parenchyma between the pharyngeal pouch and the prostatic vesicle ( Fig. 6A View FIGURE 6 ). Musculature of the prostatic vesicle mainly composed of longitudinal fibres with some interspersed oblique fibres (10–15µm thick). Ejaculatory duct lined with cuboidal to columnar ciliated epithelium, which is pierced by scarce erythrophil fine granules, surrounded by a thin longitudinal muscle layer (2.5µm thick). Epithelial lining of the male atrium columnar and densely ciliated, strongly erythrophil, receiving abundant fine granular secretion ( Fig. 6B–E View FIGURE 6 ). Its muscularis, composed of a subepithelial circular layer and a subjacent longitudinal layer, is thicker dorsally (20–30µm thick and 10–25µm thick, respectively) than ventrally (5–10µm thick and 5–10µm thick, respectively). Common muscle coat formed by loose longitudinal fibres (10–15µm thick). Male atrium connected with the female atrium by a narrow lumen ( Figs. 5B–D View FIGURE 5 , 6C–E View FIGURE 6 ).
Ovaries (150–220µm long and 130–210µm diameter) almost spherical in shape, located immediately beneath the intestine branches and among muscle fibres of the sub-intestinal parenchymal layer ( Fig. 6F, G View FIGURE 6 ). They are at 9.5–12.7mm from the anterior end (14–30% of body length) ( Table 2). Ovovitelline ducts emerge from the lateroventral face of ovaries ( Fig. 6F View FIGURE 6 ). In the pre-pharyngeal region, they are located between the sub-intestinal muscle fibres and nervous plate ( Fig. 4C, E View FIGURE 4 ). Before the gonopore, ovovitelline ducts ascend in their course to the median plane of the body. At the level of the gonopore, they join each other in a long common glandular ovovitelline duct (250–800µm long). This duct runs dorsally to the female atrium with a convex course and curves distally to open into the postero-dorsal region of the female atrium through a very short female canal (~50µm in length) ( Figs. 5B– D View FIGURE 5 , 6H View FIGURE 6 ). Female atrium (0.5–1.6mm in length) without folded walls, but with a narrow lumen because it is lined with a tall epithelium (see below) ( Figs. 5B–D View FIGURE 5 , 6C–E, H View FIGURE 6 ). Vitellaria, well developed (except paratype 1), located among intestinal branches ( Figs. 4C–E View FIGURE 4 , 6F, G View FIGURE 6 ).
Lining epithelium of the ovovitelline ducts ciliated and cuboidal, wrapped by a thin circular muscle layer (5µm thick). Common glandular ovovitelline duct lined with cuboidal to columnar ciliated epithelium, followed by a muscle layer with circular fibres and some oblique and longitudinal fibres (10–15µm thick). Similarly to the ascending portions of the ovovitelline ducts, it receives erythrophil secretion from shell glands ( Figs. 5B–D View FIGURE 5 , 6C–E, H View FIGURE 6 ). Female canal lined with ciliated and columnar epithelium, pierced by fine erythrophil granules, followed by circular muscle fibres (5–10µm thick). Lining epithelium of female atrium with stratified appearance, except near the gonopore, where it is columnar ( Figs. 5B–D View FIGURE 5 , 6C–E, H View FIGURE 6 ). Abundant fine granular erythrophil secretion discharges into the atrial epithelium. Muscularis of female atrium composed of circular, oblique and longitudinal fibres (25–35µm thick).
Comparative discussion. The new species here described fits well with the diagnosis of Imbira Carbayo et al., 2013 . This genus, which includes only two species, both recorded in the Brazilian Atlantic Forest, characterizes geoplaninid flatworms with elongated (around 90 to 200mm in length, in maximum extension), slender and flattened body, and with parallel margins; eyes marginal along the body; and presence of longitudinal parenchymal muscle fibres. In addition, the reproductive system shows a tubular prostatic vesicle with paired proximal portions; male atrium folded with eversible penis; ovovitelline ducts ascending laterally to gonopore and joining each other dorsally to female atrium; short female canal dorso-anteriorly flexed; and lining epithelium of female atrium with multi-layered aspect ( Marcus 1951; Leal-Zanchet & Carbayo 2001; Carbayo et al. 2013).
Externally, Imbira negrita sp. nov., whose dorsal pigmentation is homogeneous black, is easily distinguished from the other species of the genus: in Imbira marcusi Carbayo et al., 2013 (see Geoplana goetschi after Marcus (1951)), the dorsal ground colour is dark green olive, sometimes brownish or black, with a sub-marginal yellowish stripe contouring the body ( Marcus 1951; Álvarez-Presas et al. 2011), whereas in Imbira guaiana ( Leal-Zanchet & Carbayo, 2001) , the dorsal surface is greyish-olive ( Leal-Zanchet & Carbayo 2001). Regarding the body shape, the new species resembles I. guaiana because both species look like a ribbon, whereas in I. marcusi , which is the widest species of the genus, the body is almost lanceolate ( Marcus 1951, photos in http://planarias.each.usp.br). The pharynx of I. negrita sp. nov. is similar to that of I. guaiana , which is collar-type ( Leal-Zanchet & Carbayo 2001). In the new species, it is between bell-shaped and collar-type, although in the largest sectioned specimen the pharynx is collar-type. Although Marcus (1951) stated that the pharynx in I. marcusi is cylindrical, with dorsal insertion at the level of the mouth ( Marcus 1951, p. 191), according to Álvarez-Presas et al. (2011), who reviewed Marcus material, the pharynx is bell-shaped. The copulatory apparatus of the three species is quite similar, differing in some details. The prostatic vesicle of I. negrita sp. nov. and I. guaiana exhibits two portions. Proximally, it consists of two long tubular extrabulbar branches which run backward parallel to each other, and distally they unite into an unpaired and tubular intrabulbar portion. This unpaired portion runs sinuously and opens into a short ejaculatory duct whose lumen is narrow. In I. marcusi , the proximal part is also bifurcated and extrabulbar, but it opens directly into the ejaculatory duct, which runs along the penis bulb and traverses the eversible penis ( Marcus 1951; Álvarez-Presas et al. 2011). It would be interesting to corroborate whether this long part identified by Marcus (1951) and Álvarez-Presas et al. (2011) as the ejaculatory duct is in fact the intrabulbar part of the prostatic vesicle. Similarly to that observed in I. negrita sp. nov., the eversible penis and the male atrium of I. marcusi and I. guaiana are lined by ciliated epithelium.
Regarding the female reproductive system, the new species can be distinguished from I. marcusi and I. guaiana because, in these species, the ovovitelline ducts emerge from the dorsal region of the ovaries ( Marcus 1951; Leal-Zanchet & Carbayo 2001), whereas in I. negrita , they emerge from the latero-ventral face of the ovaries. In the three species, the ovovitelline ducts join dorsally, at the level of the gonopore or slightly behind it, with some intraspecific variation, as observed both in I. negrita sp. nov. and the Brazilian species. The same applies to the point where the female genital canal (which is extremely short in all species) opens into the female atrium. In the largest specimen of I. negrita sp. nov., the female canal opens into the most distal part of the dorsal wall of the female atrium, whereas in the smaller specimens, the female canal opens into the distal third of the female atrium but not terminally. According to the schematic drawings of specimens studied by Marcus (1951), something similar appears to occur in I. marcusi . However, Marcus reconstructed the copulatory apparatus of an animal after copulation, therefore mature, with the female canal opening in the middle third of the female atrium.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |