Tropocyclops matanoensis, Defaye, Danielle, 2007
|
publication ID |
https://doi.org/10.5281/zenodo.177864 |
|
DOI |
https://doi.org/10.5281/zenodo.5684854 |
|
persistent identifier |
https://treatment.plazi.org/id/039C87D0-FFB7-8C26-FF42-447EFDC0FA31 |
|
treatment provided by |
Plazi |
|
scientific name |
Tropocyclops matanoensis |
| status |
sp. nov. |
Tropocyclops matanoensis n. sp.
Material examined —Females and males collected in Lake Matano, Indonesia, by Doug Haffner and Lis Sabo, 05.II.2002, 90 m depth.
Type material: All type material deposited in the Muséum national d’Histoire Naturelle, Paris.
Holotype female, dissected on 2 slides, registration number MNHN-Cp2398. Allotype male, dissected on 2 slides, reg. number MNHN-Cp2399. Paratypes on slides: dissected females, MNHN-Cp2400 to MNHN- Cp2405; dissected males, MNHN-Cp2406 to MNHN-Cp2408. Ethanol-preserved paratypes: 30 females and 5 males, MNHN-Cp2409. Four paratypes mounted on SEM stubs: 2 males and 2 females, MNHN-Cp2410.
Type locality: Lake Matano, Sulawesi, Indonesia.
Etymology. The species name refers to the type-locality.
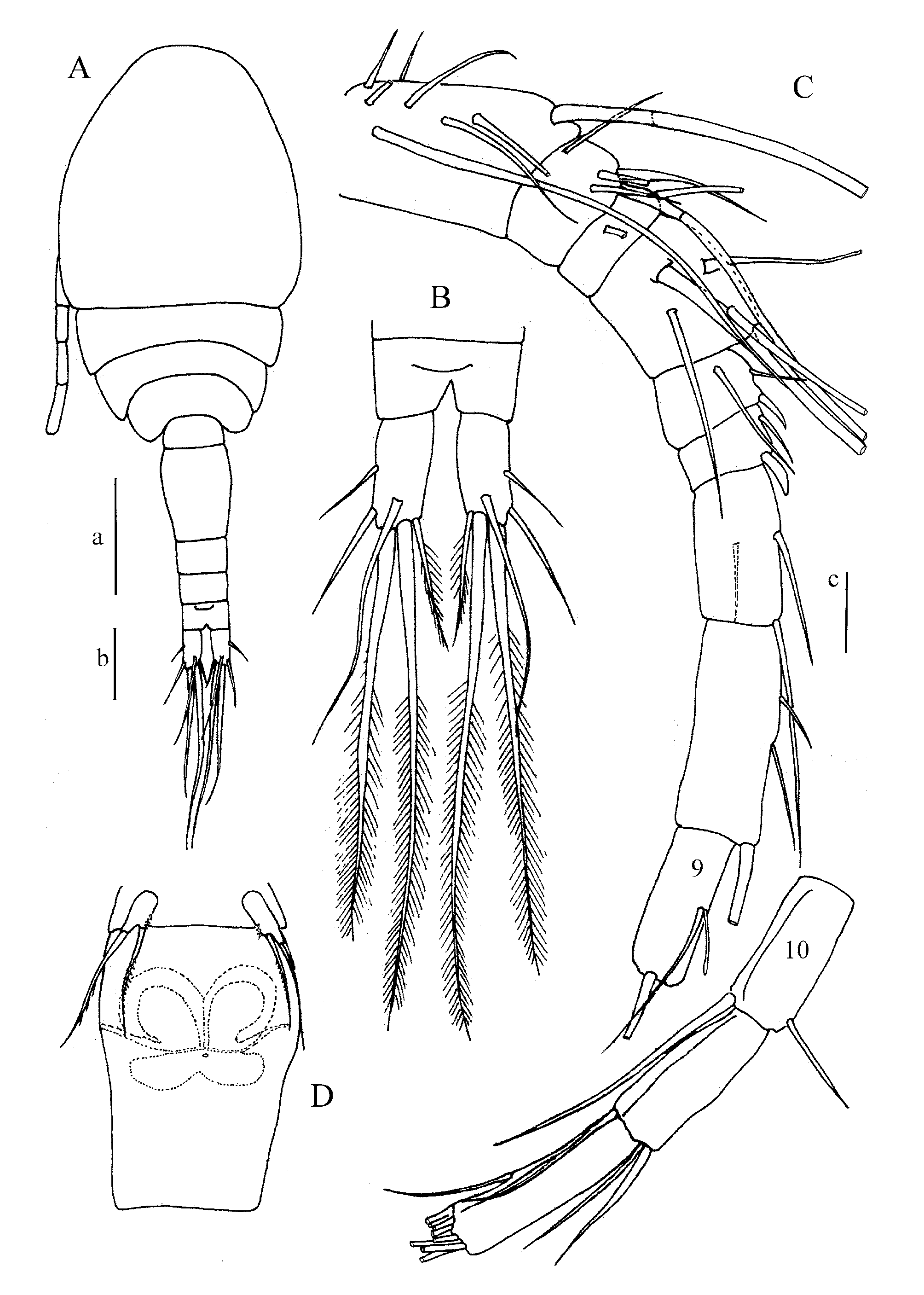
Description of holotype (female)—( Figs 1–4 View FIGURE 1 View FIGURE 2 View FIGURE 3 View FIGURE 4 , 5 View FIGURE 5 A, B, D)
Length excluding furcal setae, 650 µm (other females: 650–700 µm, mean length: 664 µm, n=6).
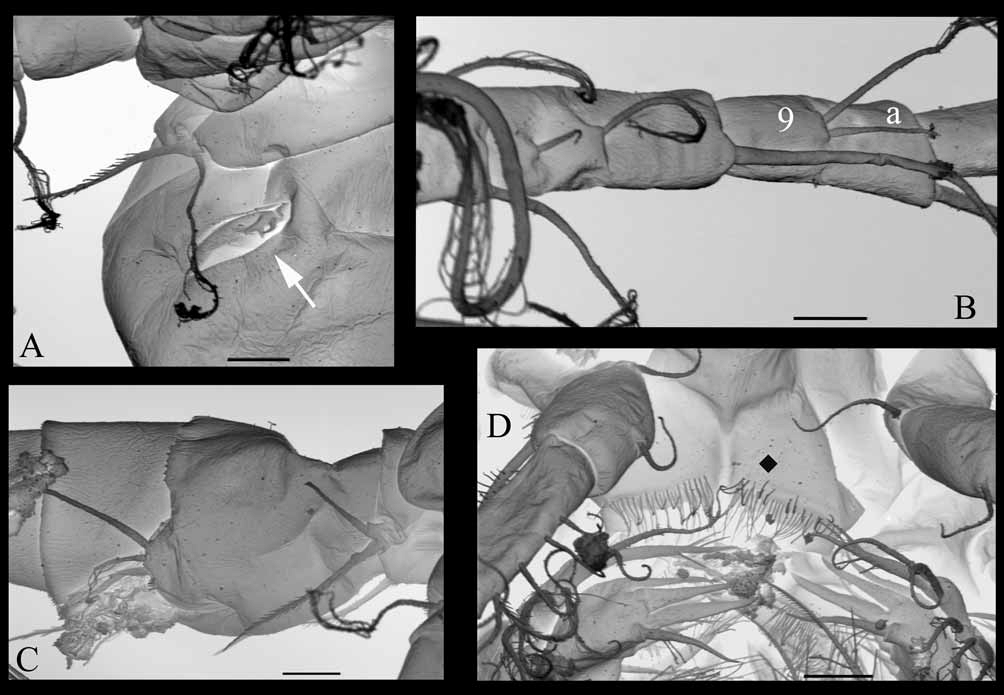
Cyclopid of medium size, slender, widest at the posterior part of cephalothorax ( Fig. 1 View FIGURE 1 A). Cephalothorax almost twice as long as the following pedigerous somites. Urosome narrow. Somite bearing leg 5 slightly narrower than genital double-somite and devoid of lateral hairs. Genital double-somite, 1.5 longer than wide, hardly broader at anterior section than at posterior part. Seminal receptacle butterfly-like shaped, situated in the proximal half of the genital double-somite ( Fig. 1 View FIGURE 1 D). Copulatory pore located proximal to centre of somite. The 2 following urosomites of same width and length, 0.44 times length of genital double-somite. Anal somite ( Fig. 1 View FIGURE 1 B), shorter than each of these 2 urosomites, bearing the anal operculum, with margin almost straight and located in anterior half of somite. No ornamentation at the distal margin of urosomites.
Furcal rami ( Fig. 1 View FIGURE 1 B) rather short, 2.1 times longer than broad, with the usual 6 setae. Furcal setae: median outer seta inserted slightly dorsal, midway (55%) along base of the furcal ramus; outermost seta and innermost seta of similar length, longer than a ramus; dorsal seta, long, more than twice length of furcal rami and outermost seta; median terminal inner seta twice length of dorsal seta and 1.14 length of outermedian terminal seta. All furcal seta plumose (not figured in illustrations). No spine or denticles at the insertion of furcal setae.
Antennule 12-segmented ( Figs 1 View FIGURE 1 C, 5B), characterized by long setae, especially on segments 1, 2, 4, 7, 8, 9; setation from proximalmost to distalmost segments (number of setae into brackets, a: aestetasc, s: spine): 1–(8), 2–(4), 3–(2), 4–(6), 5–(3), 6–(1+s), 7–(3), 8–(3), 9–(2+a), 10–(2), 1–(2) and 12–(7+1a). Segmental fusion patterns of the antennule as I–V, VI–VII, VIII, IX–XI, XII–XIII, XIV, XV–XVI, XVII–XX, XXI– XXIII, XXIV, XXV, XXVI–XXVIII. A very thin hyaline lamella, hardly visible, on last 3 segments.
Antenna ( Fig. 2 View FIGURE 2 E) 4-segmented, composed of the basipodite and the 3-segmented endopodite. Basipodite with 1 inner seta, inserted antero-distally ( Fig. 5 View FIGURE 5 D), 1 long, outer exopodal seta inserted distally. Spinule ornamentation visible on the frontal side of basipodite, as a short row of long hairs inserted medially on the outer half of the segment, 1 oblique small row of long spinules at the base of the inner margin on caudal side, located more basally ( Fig. 2 View FIGURE 2 E). Endopodite segments 1, 2, 3 with 1, 8 and 7 setae respectively; all 3 segments ornamented with row of setules or spines on the outer margin, very long hairs inserted at the median part of the outer margin of the last segment.
Labrum composed of 12 teeth and covered externally of long, distal hairs ( Fig. 5 View FIGURE 5 D).
Mandible ( Fig. 2 View FIGURE 2 A) with gnathobase ending by long, slender teeth and the usual barbed spinule, palp consisting of 2 long, plumose setae and 1 thin, small seta, without vestigial segment.
Maxillule ( Fig. 2 View FIGURE 2 B) with precoxopodite ending in the 3 usual strong terminal spines, 5 spines laterally and 1 strong, hairy spine. Maxillulary palp bearing 3 short spines distally, and distal part, representing endopodite, with 3 long setae and 1 outer spiniform seta.
Maxilla ( Fig. 2 View FIGURE 2 C) 5-segmented (as usual in Tropocyclops ) composed of precoxopodite, coxopodite, basipodite and 2-segmented endopodite. Precoxopodite with lobate proximal endite with 2 spinules, distal endite with 2 long, thin spines. Basipodite with endite armed with 3 elements, 1 forming a strong claw. Endopodite with 2 setae on first segment and 3 slender setae, distal one short and thin.
Maxilliped ( Fig. 2 View FIGURE 2 D) 4-segmented with 3 barbed setae on syncoxopodite, 2 on basipodite; endopodite 2- segmented, proximal segment with 1 long, barbed seta, distal segment with 3 setae. Syncoxopodite and basipodite bearing long setae on the external margin (not all figured), and additional group of setae on the basipodite near the insertion of barbed setae.
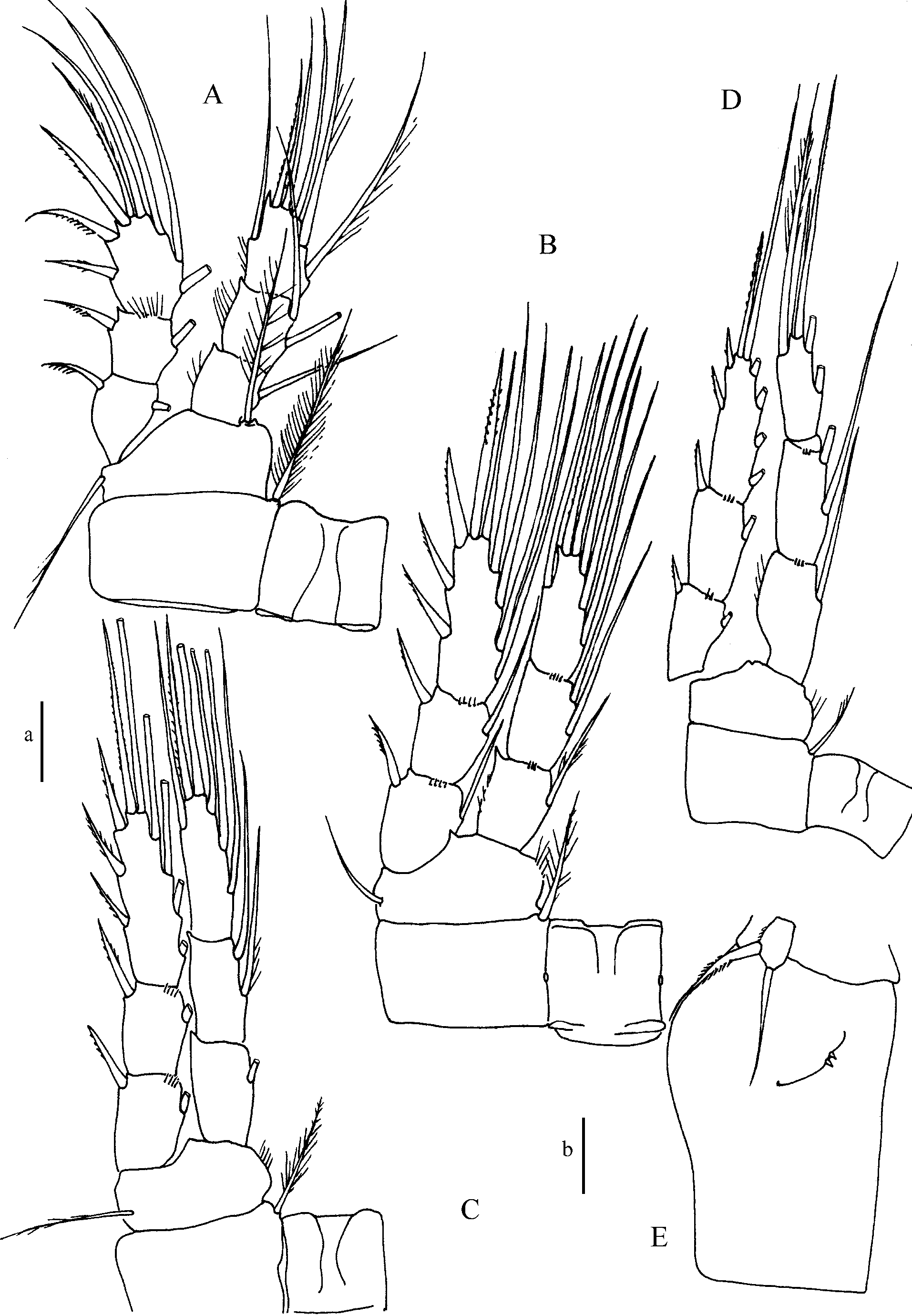
P1–P4 ( Fig. 3 View FIGURE 3 A–D) with 3-segmented rami. Spines and setae formula of distal segments of exopodites 3.4.4.3 and 5.5.5.5 respectively. Segments and spines slender. Intercoxal plates of P1–P4 with distal margin straight, and without ornamentation. P1 basipodite with 1 very long spine on the inner margin, reaching the third segment of endopodite of P4. Armature formula as follows (spines in roman numerals, setae in arabic numerals; *setiform spine; 1)not figured).
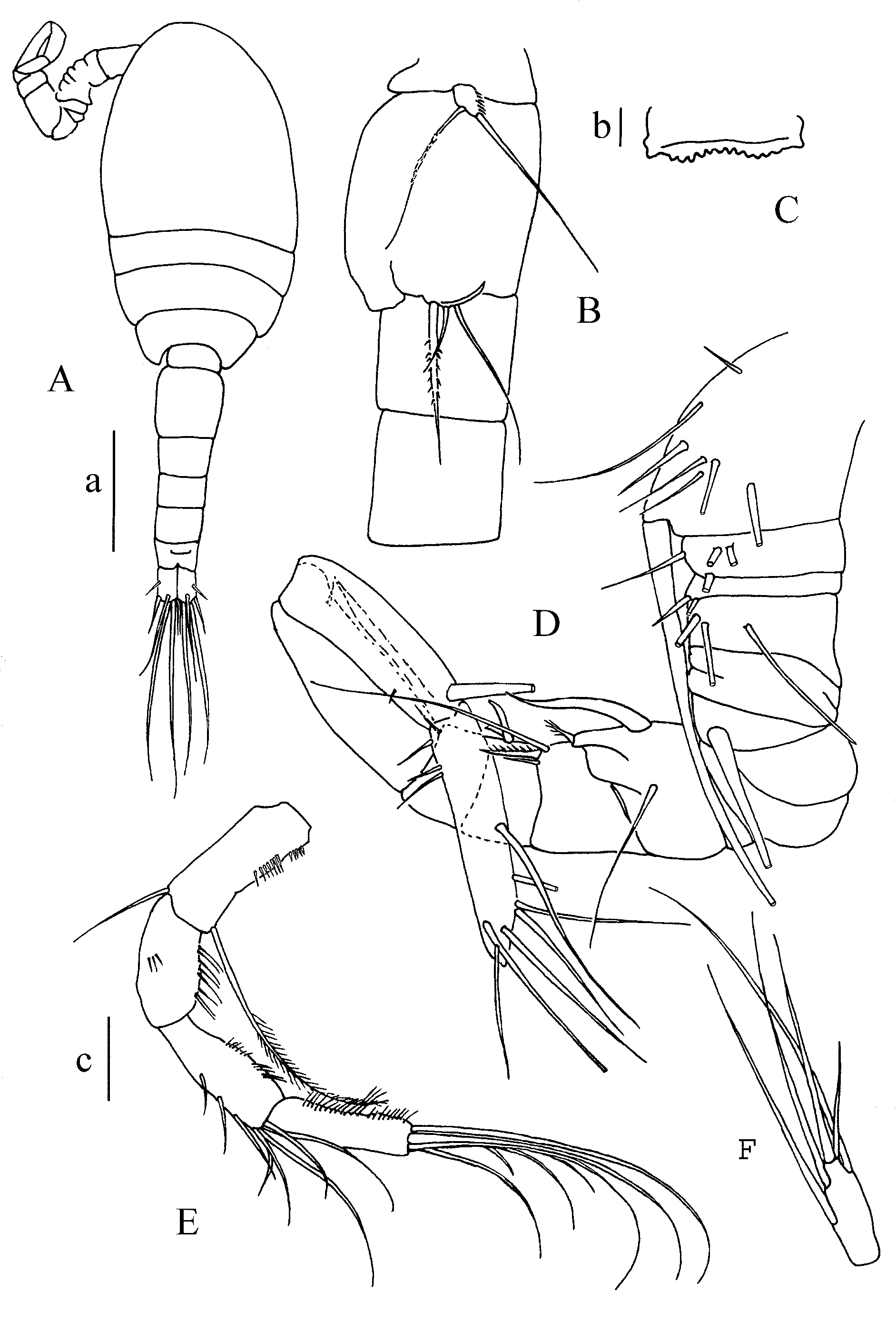
P5 ( Figs 1 View FIGURE 1 D, 3E, 5A): 1-segmented, more or less oval in lateral view, ornamented laterally with a small, distal row of short spinules, bearing 2 unequal elements: 1 long spine inserted sub-laterally on the distal half of the inner margin and 1 distal seta inserted apically, slightly longer than the spine.
P6 ( Figs 3 View FIGURE 3 E, 5A) barely visible, located dorsolaterally, only 2 short outer spines visible.
No colour observed.
No egg–sac observed.
Description of male —( Figs 4 View FIGURE 4 , 5 View FIGURE 5 C). Length: 510–585 µm (mean length: 553 µm). Anterior part of body same shape as female. Lateral corners of fourth pedigerous somite enveloping first urosomite. Urosome with 5 somites, the somite 1 devoid of setules laterally; genital somite largest, as long as wide ( Fig. 4 View FIGURE 4 B); 3 following urosomites slightly narrower than genital somite. Anal somite slightly shorter than these urosomites. Furcal rami length 2 times width. Furcal setae same as for female; terminal furcal setae plumose (not figured). No spines or denticles at insertion of furcal setae. No ornamentation at the posterior margin of urosomites.
Antennule ( Fig. 4 View FIGURE 4 D), geniculate, 16-segmented; segments 4 and 5 difficult to distinguish; very long setae on segments 1, 4, 6 and 15. Aesthetascs not distinguishable from the other small setae, probably present on segments 1, 2, 3, 11(?), 13(13). Spines of different lengths on segments 3, 10, 12, spiniform strong seta on segments 10, 11. Antenna ( Fig. 4 View FIGURE 4 E) and buccal appendages not significantly different from female. Segmentation and armature of P1–P4 (last segment of endopodite of P4: fig.4F) identical to female. P5 ( Figs 4 View FIGURE 4 B, 5C), similar to that of female, with apical external seta as long as genital somite. P6 ( Figs 4 View FIGURE 4 B, 5C), composed of 3 elements inserted close to each other on a small cuticular plate: 1 inner strong spine, the extremity of which reaches half way along the fourth urosomite, 1 small, median, thin seta and 1 outer seta a little longer than the inner spine.
Discussion. The new species is very similar to many other species of Tropocyclops by way of the shape and size of the body and the segmentation of the swimming legs. However, it differs from other Tropocyclops species in the following ways: 1) somite bearing P5 without setules on the lateral sides; 2) antenna with only one seta at postero-distal angle of the basipodite; 3) P5 bearing only two elements: one apical seta and an inner long spine inserted on the middle of the internal margin of the segment; 4) shape of the seminal receptacle. The peculiar structure of the P5 has been reported only in Tropocyclops jamaicensis Reid & Janetzky, 1996 , which was found in phytotelmata in leaf axils of a bromeliad from Jamaica.
The genus Tropocyclops currently comprises 20 species: 19 reported by Dussart and Defaye (2006) and T. bopingi , recently described by Dumont (2006) from southern China. The diagnosis of the cyclopid genera has, since Kiefer (1929), been based upon the morphology of the P5. It is defined in Tropocyclops as being composed of a “broad free segment with an anterior margin more or less triangular, bearing an external and a terminal seta and an internal spiniform seta” ( Dussart & Defaye 2001). This diagnosis has been modified by Reid and Janetzky (1996) by the addition of “except T. jamaicensis n. sp. lacking lateral seta”. The type-species, Tropocyclops prasinus ( Fischer, 1851) has long been considered a cosmopolitan species. This species is highly variable, and 12 subspecies have been described to date, differing mainly in the shape and relative lengths of the furca, the relative lengths of the furcal setae, the morphology of endopodite 3 of P4 and the relative lengths of its two terminal spines. The variability observed in the relative lengths of the furca and of the P4 Enp3 and their setae, which might be linked to ecological factors, needs to be studied in more details. However, it is possible that Tropocyclops prasinus represents in fact a complex of species. The genus Tropocyclops , which is composed of species of small size (generally around 500 µm; the largest, Tropocyclops prasinus aztequei Lindberg, 1955 , reaches 900 µm) has not been revised since Kiefer (1931a) and the systematic relationships between species of this eucyclopine genus have been little investigated until now.
Tropocyclops matanoensis n. sp. shares with T. jamaicensis the absence of lateral setules on the P5 bearing somite, but differs from the latter species in the following ways: 1) the antenna bears only one seta on the antero-distal corner; 2) the number of setae on the second segment of the antenna ( 8 in the new species, 9 in T. jamaicensis ); 3) the spine formula of the swimming legs (3.4.4.3. in T. matanoensis , 3.4.3.3. in T. jamaicensis ); and 4) the shape of the seminal receptacle.
The antennules of all other Tropocyclops species are 12-segmented. The characters of the antenna have not been considered systematically in the descriptions of the Tropocyclops species and this lack of information prevents any discussion. Re-examination of most Tropocyclops species will be necessary in order to evaluate the taxonomic importance of the antennal characters.
As the first elements of a discussion related to the antennae, it should be noted that T. prasinus and its subspecies have an external exopodal seta, and two setae on the inner distal corner of the basipodite, which is the basic pattern in Cyclopidae . Exceptions are found in two taxa: Reid (1991b) reported only one distal inner seta on basipodite, the exopodal seta being present in a population of T. prasinus from Balbina (Amazonas); the same ornamentation was figured by Gaviria for the subspecies Tropocyclops prasinus altoandinus Gaviria, 1994 from Colombia. Different patterns have been observed for the ornamentation of the basipodite of the antenna and these can be used to help in the identification of subspecies and possibly of species. Thus, in T. nananae Reid, 1991 b, the basipodite of the antenna possesses two distal internal setae and no exopodal seta; T. federensis Reid, 1991 b, has a distal internal seta and an exopodal seta. The loss of an internal seta on the basipodite of the antenna has not only been observed in species of the genus Tropocyclops , but also in different Cyclopinae genera, such as Diacyclops , Metacyclops , Thermocyclops ( Reid, 1991a) . The other important systematic character of the antenna is the number of setae on the second segment of the endopodite, which has maximum of nine in Cyclopidae . There are nine setae in T. prasinus and T. jamaicensis , but only eight in T. matanoensis ; however, information is lacking for most of the other species. The pattern of setation of the antenna, as well as the microcharacters of the basipodite, are beginning to be analyzed in more detail now that their systematic interest has been demonstrated (Fiers & Van de Velde 1984), particularly in recent studies of genera such as Mesocyclops ( Holyńska 2006) , Paracyclops ( Karaytug & Boxshall 1998) , Acanthocyclops ( Mirabdullayev & Defaye 2002, 2004).
Kiefer (1931a) defined two groups in Tropocyclops , based on the spine formula of the last segment of the swimming legs exopodites: the “quaterni”-group with the formula 3.4.4.3 and the “terni”–group with 3.4.3.3. The “quaterni”-group is composed of T. extensus ( Kiefer, 1931b) , T. multicolor ( Lindberg, 1935) , T. schubarti (Kiefer, 1935) , T. breviramus Hsiao, 1950 , T. onabamiroi Lindberg, 1950 , T. levequei Dumont, 1981 and T. piscinalis Dussart, 1984 . Two other spine formulae have been observed: 2.3. 3.3 in T. pseudoparvus Dussart & Fernando, 1986 and 3.3. 3.3 in T. federensis Reid, 1991 . All Tropocyclops species have 3-segmented legs and the chaetotaxy is well conserved, the only variation consisting of the presence or absence of the seta (or spine) inserted at the internal margin of the basipodite of P1. It is conserved in T. prasinus , T. extensus , T. schubarti , T. breviramus , T. rarus Dussart, 1983 , T. piscinalis , T. pseudoparvus , T. federensis , T. nananae and T. matanoensis n. sp., but absent in T. tenellus ( G. O. Sars, 1909) , T. confinis ( Kiefer, 1930) , T. onabamiroi , T. polkianus Einsle, 1971 , T. levequei and T. jamaicensis . However, the validity of this distinction between the “terni” and “quaterni” groups will have to be re-examined as Dumont (2006), in his redescription of T. parvus Kiefer, 1931 a from Lake Petén Itzá ( Guatemala), mentioned the presence of both spine formula in a single population. Other variations concern the intercoxal plates, which may be ornamented with one or two rows or groups of setules on the anterior part or along the margin. No ornamentation has been observed in the new species.
The P5 of Tropocyclops has been considered, until recently, to be a highly conserved, more or less triangular plate, bearing an external and a terminal seta and an inner spiniform seta. The finding, after T. jamaicensis , of a second species with a P5 bearing two elements raises questions about the value of P 5 in defining the genera in the Cyclopidae , particularly the Eucyclopinae. There are no examples of Cyclopidae in which the P5 has lost a setal element and this represents an important event in the evolution of Eucyclopinae. In T. jamaicensis, Reid and Janetzky (1996) suggested this loss might be related to life in a restricted habitat. In T. matanoensis n. sp., which is a planktonic species living in deep waters, no explanation is available as to why this seta has been lost.
A typical character of the genus Tropocyclops is the presence of setules on the lateral margins of the P5 bearing somite. These setules are absent in T. matanoensis n. sp. and T. jamaicensis , as well as in two other species: T. tenellus and T. pseudoparvus .
Another characteristic of the genus Tropocyclops is the shape of the female seminal receptacle, which is composed of two lobes: an anterior lobe with two strongly S-curved arms and a posterior bi-lobed part, typical of the T. prasinus -group. Other forms are only seen in a few species: H-shaped in T. multicolor and T. breviramus , rounded lobes in T. federensis . The butterfly-shape observed in T. matanoensis is unique, although the anterior part can be considered similar to that of T. jamaicensis .
The other characteristics of the new species fit the general diagnosis of the genus. Considering all the species of Tropocyclops , the main differences between species are the presence of one or two setae on the anterodistal corner of the antenna, the chaetotaxy of the swimming legs (quaterni and terni groups) and the P5 having two or three armature elements (excluding setules). Other differences include the number of the setae on the second endopodal segment of antenna and the presence or absence of a spine on the inner corner of the basipodite of P1. The antennule of the male in all species has setae without true sensory clubs, as observed in Eucyclops or Austriocyclops , for instance. Thus, in spite of an apparent variability, the genus is well characterized. Tropocyclops jamaicensis and T. matanoensis , the two species with bisetal P5 differ from other species in the genus in the following characters: 1) the chaetotaxy of the antenna, 2) the presence ( T. matanoensis only) of a basipodal spine on P1, and 3) the spine formula.
A detailed revision of the genus Tropocyclops will be required to determine whether T. jamaicensis and T. matanoensis should be given a new taxonomic status, either as a subgenus of Tropocyclops or a new Eucyclopinae genus. Such a revision will have to include careful observation of female and male morphological characters, including microcharacters of the buccal appendages.
Ecology. From an ecological point of view, the fact that T. matanoensis n. sp. has been encountered only at a depth of 90–100 m is remarkable. Sabo (2006) showed that Lake Matano has fully mixed waters to approximatively 80–90 m, after which an oxycline begins; the water temperature decreases slightly and chemical changes also occur, indicating that the fully mixed water column does not extend beyond a depth of 100 m. It appears that the Tropocyclops population lives in the water layer representing the lower limit of favourable conditions. The significance of this behaviour is not clear, but it is possible that it is in response to predation pressure by zooplanktivorous fish (14 fish species are present in Lake Matano ( Wirjoatmodjo et al. 2004)) and perhaps in relation to a possible vertical migration of the cyclopids during the night.
Another characteristic of the population of T. matanoensis is the near absence of egg sacs in the females, already noted as a general feature in Lake Matano ( Haffner et al. 2001); according to these authors, this would explain the low production and recruitment rates. However, the Tropocyclops population is always present, even if the abundance of ovigerous females is less than two individuals/m3 ( Sabo 2006). This suggests the nauplii hatch rapidly, and that the egg-sacs are rapidly lost by the females, perhaps as a means to avoid predation. An alternative explanation for the lack of egg-sacs may be the method of sampling, which might have caused the loss of egg-sacs.
The only other copepod species known from Lake Matano is the diaptomid Eodiaptomus wolterecki , reported by Haffner et al. (2001) and Sabo (2006). In the sample examined for the present study, there were some diaptomids co-occurring with T. matanoensis n. sp.: a few female copepodites and a male. The male has been identified as Eodiaptomus wolterecki . The male characters fit the description by Brehm (1933b), particularly the details of the antennule and the morphology of P5. As the females were not adults, it was not possible to establish whether they belong to the “var.” matanensis mentioned by Brehm (1933b) as this form differs from the typical wolterecki in the spinule ornamentation on the dorsal integument of Th2, Th3, Th4 (in the female), as well as in the small number of eggs (only two or three).
The copepod fauna of Sulawesi is little known, but it is remarkable for the rarity of the species recorded until now. For cyclopids, only Mesocyclops friendorum Hołyńska, 2000 (female only) was known from South Sulawesi. Diaptomids are represented by a few species. Eodiaptomus wolterecki wolterecki (Lakes Towuti and Mahalone) and two forms of this species: Eodiaptomus wolterecki var. matanensis (Lake Matano) and Eodiaptomus wolterecki var. tominangae (Lake Tominanga) were described by Brehm (1933b, 1942) from specimens collected during the Woltereck expedition in 1932; the taxonomic status of these varieties is uncertain and will need to be clarified. Eodiaptomus wolterecki celebensis (Lake Wawontoa) , was first described by Brehm (1933c) as Eodiaptomus celebensis . Neodiaptomus lymphatus , first described as Diaptomus lymphatus by Brehm (1933a) has been also only reported from Sulawesi. Tropodiaptomus (Tropodiaptomus) doriai (Richard, 1894) is known also from Java and Sumatra and Tropodiaptomus (Tropodiaptomus) malaicus (Grochmalicki, 1915) also from Java, and Bali ( Dussart & Defaye 2002).
The presence of a new species of Tropocyclops in Lake Matano constitutes a new addition to the fauna of this ancient lake. In the case of T. matanoensis , we can hypothesize that the long isolation of the lake fauna has resulted in this particular form, which is the second of the genus possessing only one seta and one spine on P5. The copepod fauna of the lake is probably far from completely known and further sampling in this lake and other ancient lakes in Indonesia, particularly in the deep biotopes, which are rarely studied in fresh waters, will probably reveal new taxa. Further studies will allow a better evaluation of the biodiversity of the copepod fauna, notably concerning their possible endemicity. Such studies are of importance because the biogeographical area of Wallacea, which represents one of the 20 global hotspots of biodiversity faces threats to many of its habitats ( Myers et al. 2000).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |