Trichomycterus pascuali, Datovo, Alessio, 2017
|
publication ID |
https://doi.org/10.11646/zootaxa.4319.3.7 |
|
publication LSID |
lsid:zoobank.org:pub:5Ed288D1-Bb13-4Deb-9B97-9E5C4902D5B8 |
|
DOI |
https://doi.org/10.5281/zenodo.5683961 |
|
persistent identifier |
https://treatment.plazi.org/id/03A6B93D-F406-FFCD-D38C-FB1605E7FD30 |
|
treatment provided by |
Plazi |
|
scientific name |
Trichomycterus pascuali |
| status |
sp. nov. |
Trichomycterus pascuali , new species
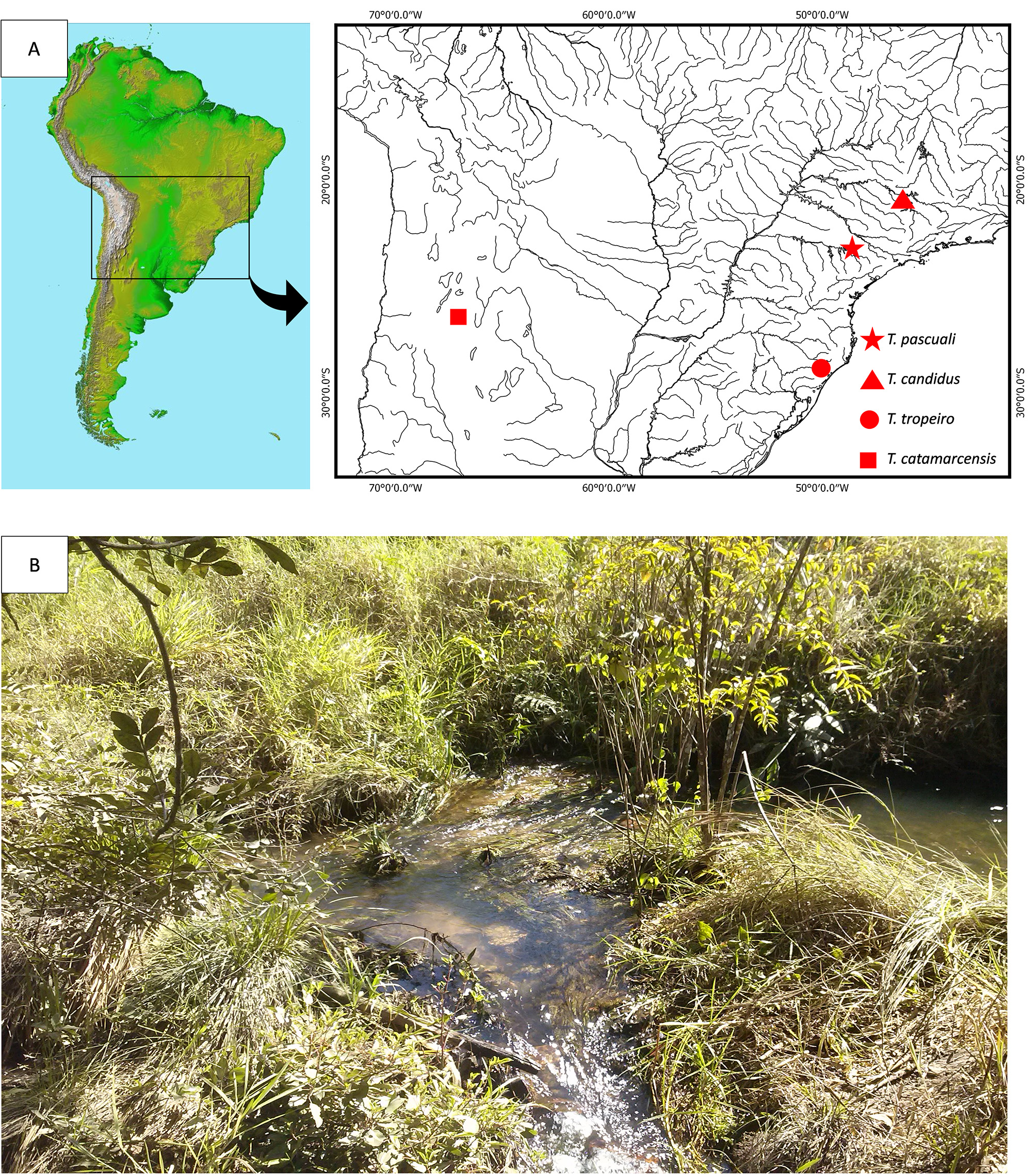
( Figs. 1–3 View FIGURE 1 View FIGURE 2 View FIGURE 3 , Table 1)
Holotype. MZUSP 121681 View Materials , 48.8 View Materials mm SL; Brazil: São Paulo State, Municipality of Itatinga; unnamed tributary of Tamanduá river, Paranapanema Basin , 23°13’27.06”S 48°31’45.34”W, G.S.C. Silva, G.J. Costa e Silva, L.E. Ochoa, 14 Feb 2017 GoogleMaps .
Paratypes. LBP 23323, 8 ( 20.76-46.53 mm SL), MZUSP 121682 View Materials (2 ex): 2 CS (20.76, 22.30 mm SL), 1 MS (46.53), 1 genetype ( 20.76 mm, Genbank accession number: MF034463 View Materials - COI, MF034462 View Materials -16 About COI S); same data as holotype GoogleMaps .
Diagnosis. Trichomycterus pascuali is readily distinguished from its congeners by two remarkable features: possession of five pectoral-fin rays ( vs. six or more) and absence of pelvic fin, girdle, and muscles ( vs. presence). Within Trichomycterus , only T. candidus (Rio Grande basin, Brazil), T. catamarcensis ( Laguna Blanca basin, Argentina), and T. tropeiro (laguna dos Patos basin, Brazil) also lack pelvic structures. Trichomycterus pascuali differs from these three species in having the aforementioned five pectoral-fin rays ( v s. six, eight, and seven, respectively) and seven branchiostegal rays ( vs. eight, five or six, and nine, respectively). It addition, it differs from T. candidus by the possession of anterior and posterior cranial fontanels completely separated from each other by a broad epiphyseal bar ( vs. fontanels partially or completely fused, with epiphyseal bar absent or minute and incomplete) and 37 post-Weberian vertebrae ( vs. 38–39 in T. candidus ); from T. catamarcensis by the first pectoral fin prolonged as a small filament ( vs. pectoral filament absent in most specimens), 14 ribs ( vs. 18–20 in T. catamarcensis ), 17–18 dorsal procurrent caudal-fin rays ( vs. 12–13), and 15–16 ventral procurrent caudal-fin rays ( vs. 11–12); and from T. tropeiro by the presence of pectoral filament ( vs. absence), 17–18 dorsal procurrent caudal-fin rays ( vs. 13–14), 15–16 ventral procurrent caudal fin rays ( vs. 9–10), and absence of pores i1 and i3 ( vs. presence).
Members of the so-called “ Trichomycterus ” hasemani group, a clade of highly derived Amazonian miniature trichomycterids, also exhibit a low count of pectoral-fin rays and one of its species, “T.” anhanga , further lacks pelvic fins. However, the “T.” hasemani group is demonstrably a new subfamily that is more closely related to the Vandelliinae-group than to trichomycterines (de Pinna 1989; Dutra et al. 2012; Datovo 2014, Wosiacki 2002). The nominal assignment of these species to the genus Trichomycterus is merely due to taxonomic inertia, as the formal description of the new subfamily is still underway (by W. Wosiacki and M.C.C. de Pinna, pers. comm.). In any event, T. pascuali differs from all species of the T. hasemani group by the relatively large body size, with up to 49.4 mm SL ( vs. miniature body, with less than 18.6 mm SL); presence of an ossified neurocranial roof with two small fontanels ( vs. roof of neurocranium mostly unossified and forming a single wide fontanel); and 14 ribs ( vs. 1–3).
Description. Morphometric data are given in Table 1. Body elongate, maximum total length 52.2 mm SL, cross section of trunk roughly cylindrical at pectoral girdle and progressively more compressed towards caudal-fin. In lateral view, dorsal profile of body straight from snout tip to interorbital region, slightly convex to dorsal-fin origin, and slightly convex to end of caudal peduncle. Ventral surface of body convex from tip of snout to anal-fin insertion. Caudal peduncle smoothly continues profile of trunk region, with dorsal and ventral profiles slightly convex. Greatest body depth at dorsal-fin origin. Head wide, longer than wide, nearly trapezoidal in dorsal view. Eyes elliptical, covered by thin and translucent skin separate from surface of eyeball. Anterior nostril larger than posterior one, surrounded by fleshy flap of integument continuous with base of nasal barbel. Posterior nostril partially surrounded anteriorly by thin flap of skin. Seven branchiostegal rays visible in c&s individuals. Mouth subterminal, its corners laterally or slightly posteriorly oriented. Lower lip with conspicuous fleshy lobes along lateral limits, lobes situated internal to base of rictal barbels. Lower lip with anterior and, to lesser degree, anteroventral surfaces covered by small papillae. Anterior margin of upper lip rounded with numerous papillae. Barbels relatively short and with larger bases. Nasal barbel thick always surpassing posterior margin of eye but not reaching opercular patch of odontodes. Maxillary barbel reaching posterior tip of interopercular patch of odontodes. Rictal barbel reaching interopercular patch of odontodes. Interopercular patch of odontodes posteriorly elongate with 11 or 12 conic odontodes imbedded in flesh. Opercular patch of odontodes very small, with 10–12 thin odontodes with thick tips.
Cephalic laterosensory system including incomplete supraorbital and infraorbital canals. Infraorbital canal restricted to its posterior fragment corresponding to pores i10 and i11. Supraorbital pores s1, s3 and s6. Two bilaterally paired pores s6. Lateral line of trunk reduced, comprising two pores dorsal to middle of pectoral fin. Vertebra 37, ribs 14.
Pectoral fin narrow, with rounded margin. Pectoral-fin rays i,4, first one longest, unbranched, prolonged as small filament. Dorsal fin with distal margin rounded, ii,5-7 rays (modally ii,6,i). Anal fin slightly smaller than dorsal fin, with distal margin rounded, ii,4-5,i rays, origin at vertical through posterior portion of dorsal fin. Pelvic fin, girdle and muscles absent. Caudal fin with distal margin truncate with rounded corners and dorsal and ventral edges slightly convex, i,5+6,i principal rays, branched rays splitting tree times. Procurrent caudal-fin rays 17 or 18 dorsally and 15 or 16 ventrally.
Color in alcohol. Two main body-coloration patterns. First pattern ( Fig. 2A View FIGURE 2 ) observed in specimens 20.8–48.8 mm SL. Ground color of body yellowish. Head covered by irregular brown spots and two regular longitudinal stripes along lateral region of body, one dorsolateral and another midlateral. Ventrolateral row of spots observed at abdominal region and caudal peduncle. Dorsal, anal and caudal fins slightly pigmented.
Second color pattern ( Fig. 2B View FIGURE 2 ) found in specimens 49.4–52.2 mm SL. Head with some regular brown spots in the dorsal region, gradually lighter laterally; barbels slightly pigmented with the same coloration as head spots. Trunk yellowish with dorsal and abdominal lateral portion of the body covered by irregular spots from the lateral head region to caudal peduncle without defined pattern. Pectoral fin unpigmented, dorsal, anal, and caudal fins rays pigmented with the same coloration as the body spots.
Etymology. The name “ pascuali ” was given in honor to José Pascual Ochoa, L. Ochoa’s father.
Distribution. Known only from the type locality, a small stream tributary of Tamanduá river, Paranapanema river, Upper Paraná, close to the town of Itatinga, São Paulo state, southeastern Brazil ( Fig. 4 View FIGURE 4 ).
Habitat, ecological notes, and conservation status. The new species was discovered in a small stream with clear water and moderate water flow at the altitude of approximately 600 meters above sea level. The specimens were collected under rocks and aquatic vegetation in a muddy bottom area bordered by riparian vegetation. The region is highly impacted by agricultural activity, more specifically eucalyptus monoculture. Rivers in the region are poor in both alpha diversity and specimen abundance. Trichomycterus pascuali was the only species of fish found in the type locality and the number of individuals sampled was low. This species has a restricted area of occupancy and its vulnerability to the threats from anthropic impacts could drive the taxon to critically endangered in a short time. According to the IUCN (2001) criteria, T. pascuali should be classified in the category vulnerable VU D2.
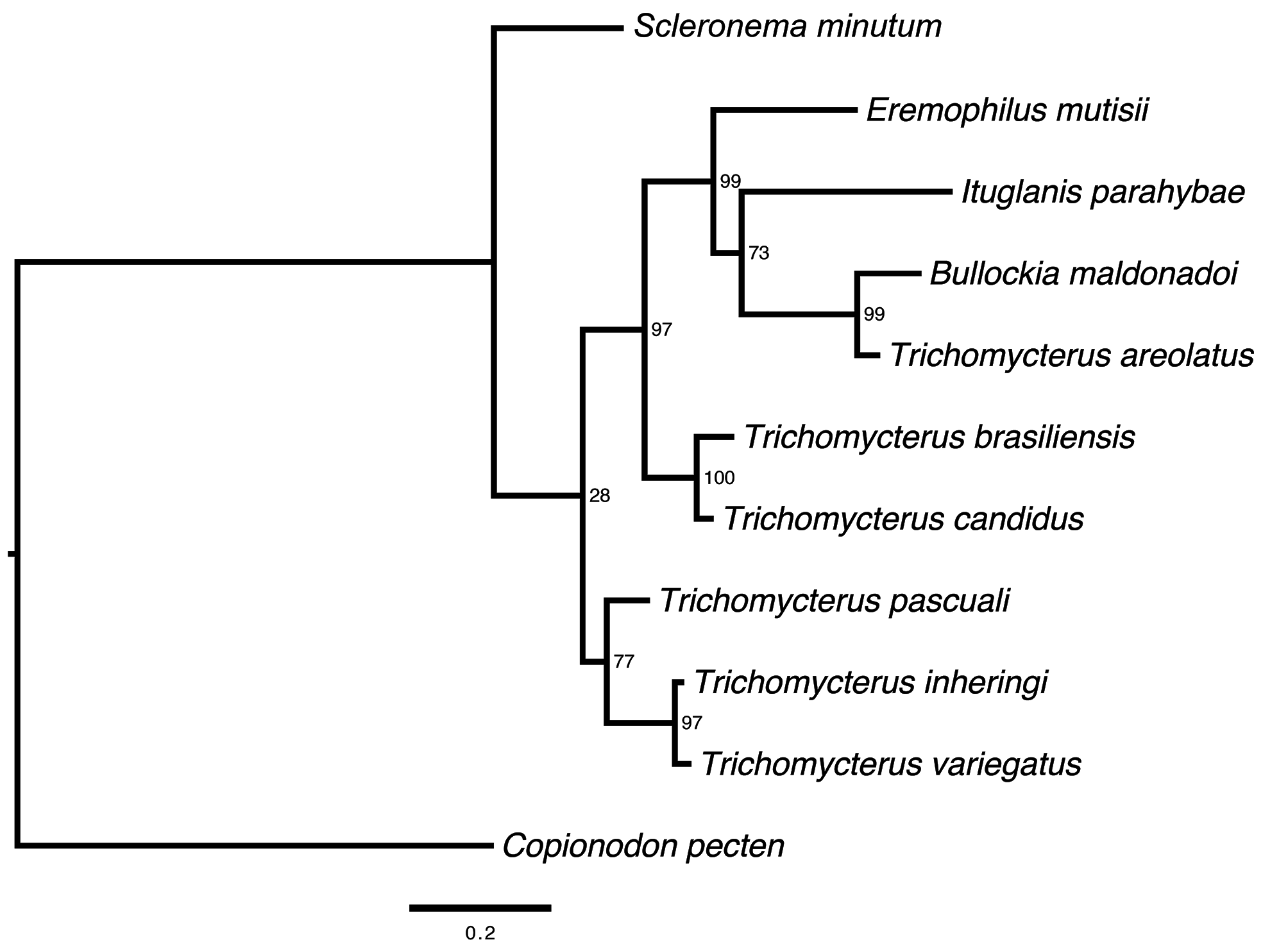
Genetic identification. Aligned sequences were obtained for 573 bp of COI from the reference set of 11 tissue samples of trichomycterines, the translation of sequences did not result in stop codons, indicating the amplification of functional domains. The nucleotide composition of the COI matrix was 29.8% (T), 26.8% (C), 25.1% (A), 18.3% (G), with a proportion of 400 invariables sites, 173 variables sites and 113 parsimony informative sites. The overall mean genetic distance based in the COI gene was 0.138±0.011. The genetic divergence between species was superior to 3% and the results show concordance with the morphological identification. The pairwise species comparison ranged from 0.5 to 24.1% ( Table 2 View TABLE 2 ) and the divergence genetic within the Trichomycterus species was 0.097±0.009. Trichomycterus pascuali presented a genetic divergence with its congeneric species from 9.3 to 11.7%, with T. candidus and T. brasiliensis respectively. the genetic divergence between E. mutisii and T. pascuali was (10.8%) similar with other species of the Trichomycterus , the maximum likelihood analysis using a concatenated matrix with COI and 16S genes (1122 pb, 30.2%(T), 25.0 (C), 23.7%(A), 21.1%(G), with 112 conserved sites, 254 variables sites and 145 parsimony informative sites) shows that T. pascuali is more related with T. inhering and T. variegatus than other genera included in this analysis with a bootstrap support of 77% ( Fig. 5 View FIGURE 5 ).
| COI |
University of Coimbra Botany Department |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |