Sinocallipus simplipodicus Zhang, 1993
|
publication ID |
https://doi.org/ 10.5281/zenodo.294081 |
|
publication LSID |
lsid:zoobank.org:pub:4E13D217-051D-4B09-8C57-B141A3E37722 |
|
DOI |
https://doi.org/10.5281/zenodo.6276562 |
|
persistent identifier |
https://treatment.plazi.org/id/03AF9150-9D5B-2C26-FE89-FD48FE080106 |
|
treatment provided by |
Admin |
|
scientific name |
Sinocallipus simplipodicus Zhang, 1993 |
| status |
|
Sinocallipus simplipodicus Zhang, 1993 View in CoL
Figs. 3–14 View FIGURES 3 – 5 View FIGURES 6 – 10. S View FIGURES 11 – 14. S
Sinocallipus simplipodicus Zhang, 1993:129 , figs. 1–16.
Type specimens. Male holotype, female allotype, and 15 male (one juvenile) and 5 female (one juvenile) paratypes collected by F. Zhang, D. Wang, X. Chen, and H. Lu, 8 and 13 April 1992, in Xiao Cave, Hekou Yaozu Autonomous County, Yunnan Province, China (not examined). The repository is not stated specifically in the original description (Zhang, 1993) but presumably is the Institute of Zoology, Chinese Academy of Sciences, Beijing.
Diagnosis. With the characters of the genus.
Description of Laotian specimens. Color (after two years in preservative, but presumably close to this in life): Ocellar patches black; interantennal and epicranial regions of head brownish. Collum, segment 2, prozonum and anterior half of metazonum of 3rd pleurotergite, and epiproct white, completely unpigmented; remaining pleurotergites mottled brownishgray with a light, narrow middorsal stripe.
Segment counts as follows (because of the difficulty in distinguishing segments at the caudal end, where they are smaller and closely packed together, an error of + 2 segments is assumed): adult males (with gonopods) with 54–68 segments, females with 61–69. Measurements (on two relatively straight individuals of both sexes): males 44.0 & 46.0 mm long, 2.3 & 2.4 mm wide; females 48.0 & 51.7 mm long, 2.8 & 3.0 mm wide.
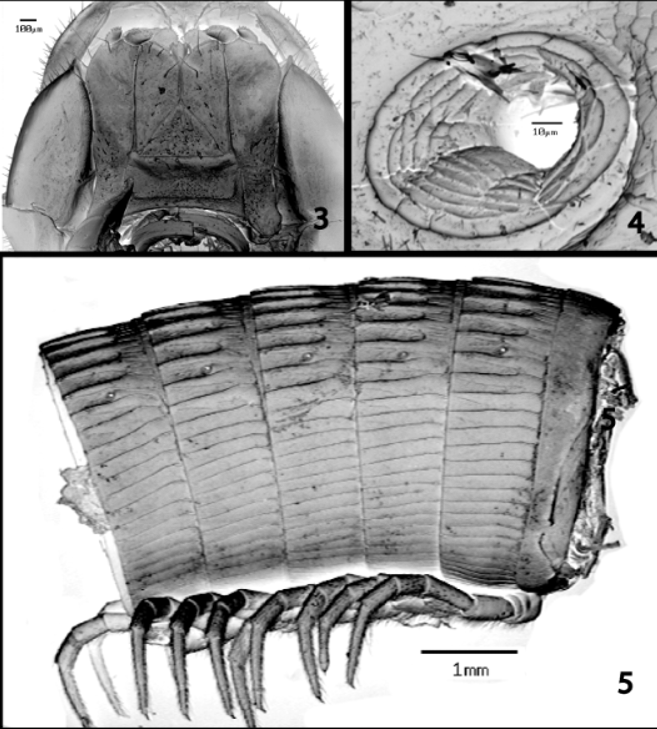
Head densely setose, especially frontal and subantennal regions, setae diminishing in number and becoming longer on epicranium. 41 ocelli in right ocellarium in one adult male. Gnathochilarium ( Fig. 3 View FIGURES 3 – 5 ): laminae linguales broad, touching distal to midlength in midline, with distal setae but without palps, narrowing proximad and separated by broad, triangular mentum; stipes long and broad, extending for entire length of gnathochilarium along lateral margins of lamina linguales, separated basally by broad, subrectangular prebasilare, with two apical palps apiece, each with a row of apical papillae.
Collum and second segment smooth; crests arising on 3rd pleurotergite, low, broad, and poorly defined, only slightly more distinct on 4th pleurotergite; carinae on remaining pleurotergites low and narrow, not prominent, becoming progressively lower laterad and grading into striae ventrolaterad ( Fig. 4 View FIGURES 3 – 5 ), first crest on each side of midline inconspicuous; only primary crests present, secondary and tertiary ones absent, without crest transition. Ozopores ( Fig. 5 View FIGURES 3 – 5 ) relatively broad, arising on segment 6, located between 3rd and 4th crests on each side of midline. Segments with strong, distinct strictures between pro and metazona ( Fig. 4 View FIGURES 3 – 5 ). Dorsal setae sparse and scattered, short and inconspicuous, seemingly broken and apparently obliterated by abrasion with substrate, present only on anteriormost segments, extending to segments 4–5 in most individuals but to segments 6 and 9 in two females; all setae on anterior positions on carinae, no evidence of setal migration. Epiproct short and apically broad, margin nearly linear, with two long, prominent spinnerets projecting caudad from ventral surface.
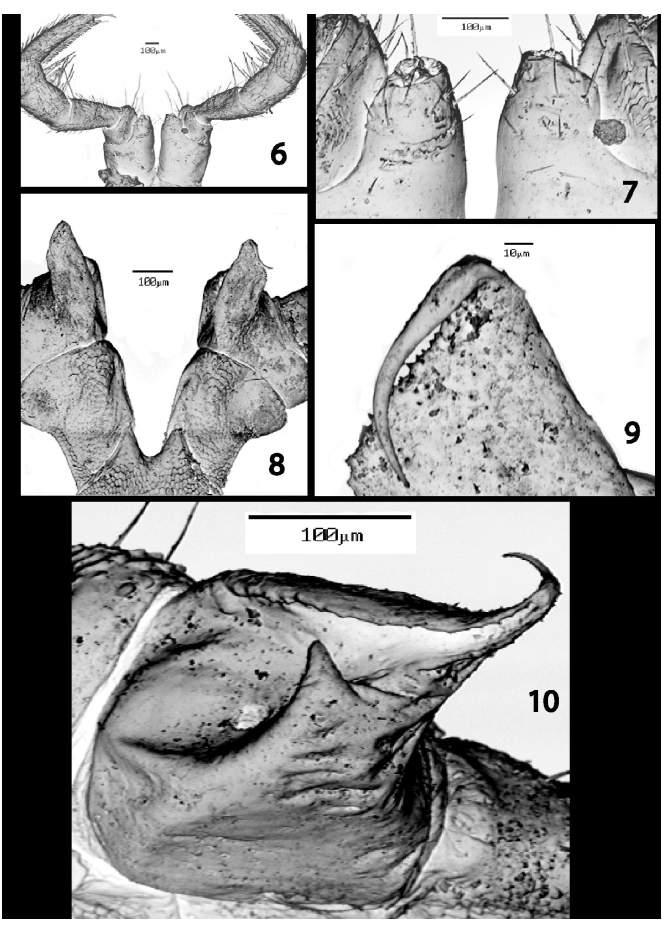
Males with first two legs slightly incrassate, first comprising only six articles, second with seven podomeres and gonapophyses arising mediad from coxae ( Figs. 6–7 View FIGURES 6 – 10. S ). Remaining legs composed of 7 articles; trochanters of 9th legs with short, broad, deeply excavated, caudomedial lobes ( Figs. 8–10 View FIGURES 6 – 10. S ), possessing central pore, a short spur on anteriomedial corner, and curved, spiniform projection on caudomedial corner, lying across distal margin of lobe on some legs to form a “cheliceralike” appearance ( Figs. 10 View FIGURES 6 – 10. S ). Prefemora on legs in anterior 2/3 of males with ventral pads; all tarsi with ventral comb rows ( Fig. 6 View FIGURES 6 – 10. S ).
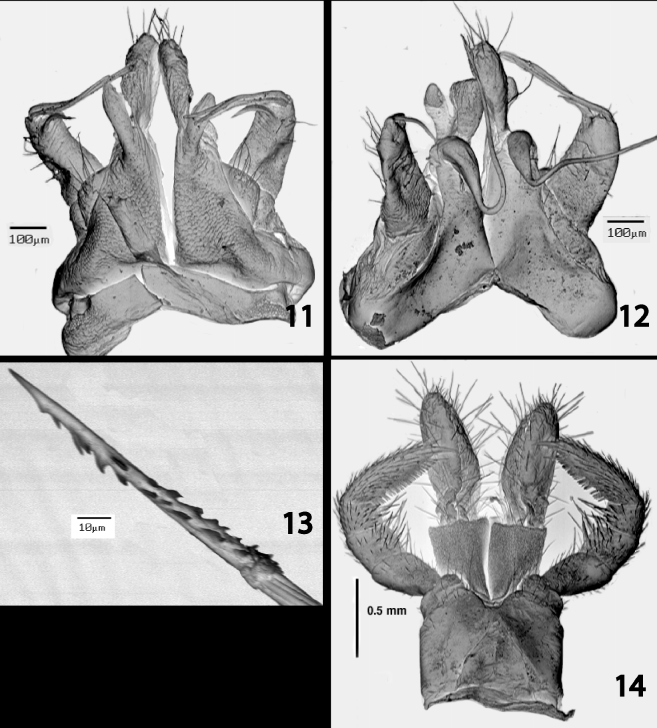
Gonopods in situ with telopodal projections directed anteromediad, constituting anteriormost part of structure; apices of cannulae directed anteriolaterad, overhanging midlength of telopodal projections. Gonopod structure as follows ( Figs. 11–12 View FIGURES 11 – 14. S ): Coxae connected basally by broad sternum extending for entire breadth of gonopods, each with two clavate processes extending directly ventrad on anterior side, a short, glabrous, anterior process arising basally from anterior side of longer, caudal process, latter apically setose, with several long setae on lateral margin at articulation with telopodite. Telopodite short and broad, positioned lateral to coxa and extending directly ventrad, without prostatic groove, with three slender, acicular, narrowly separated terminal projections directed strongly mediad; caudalmost projection longest, in some males divided near midlength into two subequal, closely appressed subbranches, curvilinear, overlapping longer coxal process and in one male (not photographed) extending slightly beyond medial margin of latter and nearly meeting corresponding projection from opposite gonopod; medial projection slightly shorter, also curvilinear, overhanging longer or shorter coxal processes; anteriormost projection shortest, broad and subfalcate, extending slightly beyond telopodal margin and terminating well short of coxal processes. Cannula long and slender, not coiled, arising near midlength of caudal side of coxa at level of origin of shorter anterior process, angling dorsomediad basally and converging with opposite member, then curling and diverging, angling anteriolaterad (anteriad in dissected specimens), apices either overhanging coxal processes or midlength of telopodal projections, or dislodged (as in right cannula in Fig. 12 View FIGURES 11 – 14. S ) and remaining on caudal side of telopodite, terminating freely, not inserting into groove or pit in telopodite, with minute barbs distally and grooves curving around structure for an unknown distance proximad ( Fig. 13 View FIGURES 11 – 14. S ).
Female ovipositor as in fig. 14. Second legs with tarsal combs.
Var ia t io n. The Laotian specimens show a few differences from Zhang’s characterization (1993) of the Chinese specimens beyond the obvious one of pigmentation. The spinnerets arise from beneath the epiproct, instead of on its caudal margin, and extend well beyond the latter. Zhang characterized the family Sinocallipodidae as one in which the diplosegments lack interzonal strictures, but as shown in fig. 4, they are distinct in the Laotian material. The gonopods agree closely with Zhang’s illustrations of Chinese males (the anterior view is of a paratype, the caudal view, the holotype [Zhang, 1993, figs. 12 & 13, respectively]), the main difference being the length and configuration of the cannula. In the Yunnan specimens, the structure is short and coiled, and lies wholly on the caudal side of the coxal processes, perhaps to some degree an artifact of the illustrations reflecting foreshortening; however, the cannula in Laotian males curves but does not coil and is much longer, extending in situ ventrolaterad and over (ventral to) the telopodal projections. The number of the latter can also vary; it usually is three but occasionally there are four caused by division, near midlength, of the caudalmost branch into two equivalent subbranches that are closely appressed to each other. In one Laotian male, the left telopodite has four branches and the right, three.
In the few individuals that could be measured and whose segments could be counted, females are longer and broader than males. Segment counts are comparable; the available males have lower counts, but females show less variation.
The pigmentation and number of ocelli (41) in the Laotian specimens, compared to the lack of pigmentation and reduced number of ocelli (11–16) in those from Yunnan, imply a degree of cave adaptation in the latter.
Natural history observations. The Laotian specimens were encountered at around 984 ft. (300 m) elevation, 4–5 cm deep in leaf litter and humus in a wet evergreen forest. No information is available on the microhabitat of the Yunnan specimens other than the fact that they were taken in a cave.
Distribution. Same as that of the genus. Xiao Cave is in the southern fringe of Yunnan Province, near the town of Hekou on the Hong River near Lao Cai, Vietnam. This part of Yunnan projects southward into Laos, and Xiao Cave is at the tip of this point, just on the Chinese side of the border. The Laotian site (details below) is some 600 mi (960 km) to the southsoutheast in the southernmost provinces of Laos, ca. 50 mi (80 km) from both the Thailand and Cambodian borders at their closest points.
LAOS: Champasak/Attapu Provs., Dong Hua Sao National Biodiversity Conservation Area, along the Houry Phak River near the SW edge of Bolavens Plateau (lat.15 o0 4' 37"N, long. 106o10'45"E), 5M, 9F, September 1999, H. Heatwole (North Carolina State Museum of Natural Sciences and Virginia Museum of Natural History collections).
Remarks. Definitive studies on the roles of the parts of the gonopod in reproduction have not been conducted, but even though the telopodal branches are, in situ, the anteriormost part of the gonopod of S. simplipodicus , we believe the cannula may be the “functional” element that directly inseminates females particularly because it terminates freely and the telopodite lacks a prostatic groove. The telopodite is also firmly attached and stationary, whereas the cannula is movable at the articulation with the coxa as evidenced by how easily it is dislodged during dissection. In the introduction to his work, Zhang (1993) stated “seminal furrow (= prostatic groove) obscurely visible,” but it is not shown on his figures nor mentioned in the gonopodal characterization in the species’ description. Careful examination of the Laotian gonopods reveals no evidence of a prostatic groove, and we believe it is indeed absent. Without the groove and a connection between the cannula and telopodite, the most plausible method of spermatophore/ seminal fluid transfer seems to be by the cannula directly, which in the Laotian males is the longest gonopodal structure, could most easily contact the openings of the vasa deferentia to be “charged,” and subsequently penetrate the female cyphopods during copulation. Observing the gonopods in situ, it is evident that the body could be flexed and the cannulae “pushed” forward to contact the gonapophyses and cyphopods of females. If the cannulae are the “functional” element instead of the telopodite as in other callipodidans, this could be interpreted as a plesiomorphy, an early stage in gonopod development in which a “functional” telopodite is obviated by a more direct method of spermatophore transmittal that requires a less intricate structure. The presence on the 9th legs of the trochanteral lobe with short projections and an internal duct of some sort as evidenced by the central pore ( Figs. 8–10 View FIGURES 6 – 10. S ) is an unknown here. This is another unique feature in Sinocallipus , as it is unknown from any other callipodidan.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |