Pahudioxylon bankurensis Chowdhury, Ghosh & Kazmi
|
publication ID |
https://doi.org/10.5252/geodiversitas2022v44a28 |
|
publication LSID |
urn:lsid:zoobank.org:pub:2611B0BC-F569-4135-A09C-6E527C2565A4 |
|
DOI |
https://doi.org/10.5281/zenodo.7157559 |
|
persistent identifier |
https://treatment.plazi.org/id/03B787F6-A211-FFA2-8953-FACEE34CEDAF |
|
treatment provided by |
Felipe |
|
scientific name |
Pahudioxylon bankurensis Chowdhury, Ghosh & Kazmi |
| status |
|
Pahudioxylon bankurensis Chowdhury, Ghosh & Kazmi
( Fig. 8 View FIG )
Pahudioxylon bankurensis Chowdhury, Ghosh & Kazmi, 1960: 22 , pl. 2, figs 1-6, ill. 1.
Albizzioxylon sahnii Ramanujam, 1960: 118 , pl. 21, ill. 25.
Pahudioxylon sahnii Ghosh & Kazmi, 1961: 96 , figs 1, 2.
Pahudioxylon deomaliense Prakash, 1965c: 433 , figs. 1, 2.
Ingoxylon sahnii – Müller-Stoll & Mädel 1967: 112.
Pahudioxylon welkitii Lemoigne & Beauchamp, 1972: 336 , pl. 12.
Pahudioxylon assamicum Prakash & Tripathi, 1975: 63 , pl. 2, figs 7, 9, 11, 12.
Pahudioxylon indicum Prakash, 1979: 66 , pl. 2, figs 1-5.
Pahudioxylon bengalensis Ghosh & Roy, 1982: 52 , figs 1, 2.
ORIGINAL HOLOTYPE. — Geological Survey of India ( G.S.I.) no. P2 /126.
MATERIAL. — MNHN.F.50183 (field number: 17FN03). Estimated minimal diameter: 14-34 cm.
LOCALITY. — Kalewa Township, Sagaing Region, Myanmar.
AGE. — Upper lower to lowermost middle Miocene.
DESCRIPTION
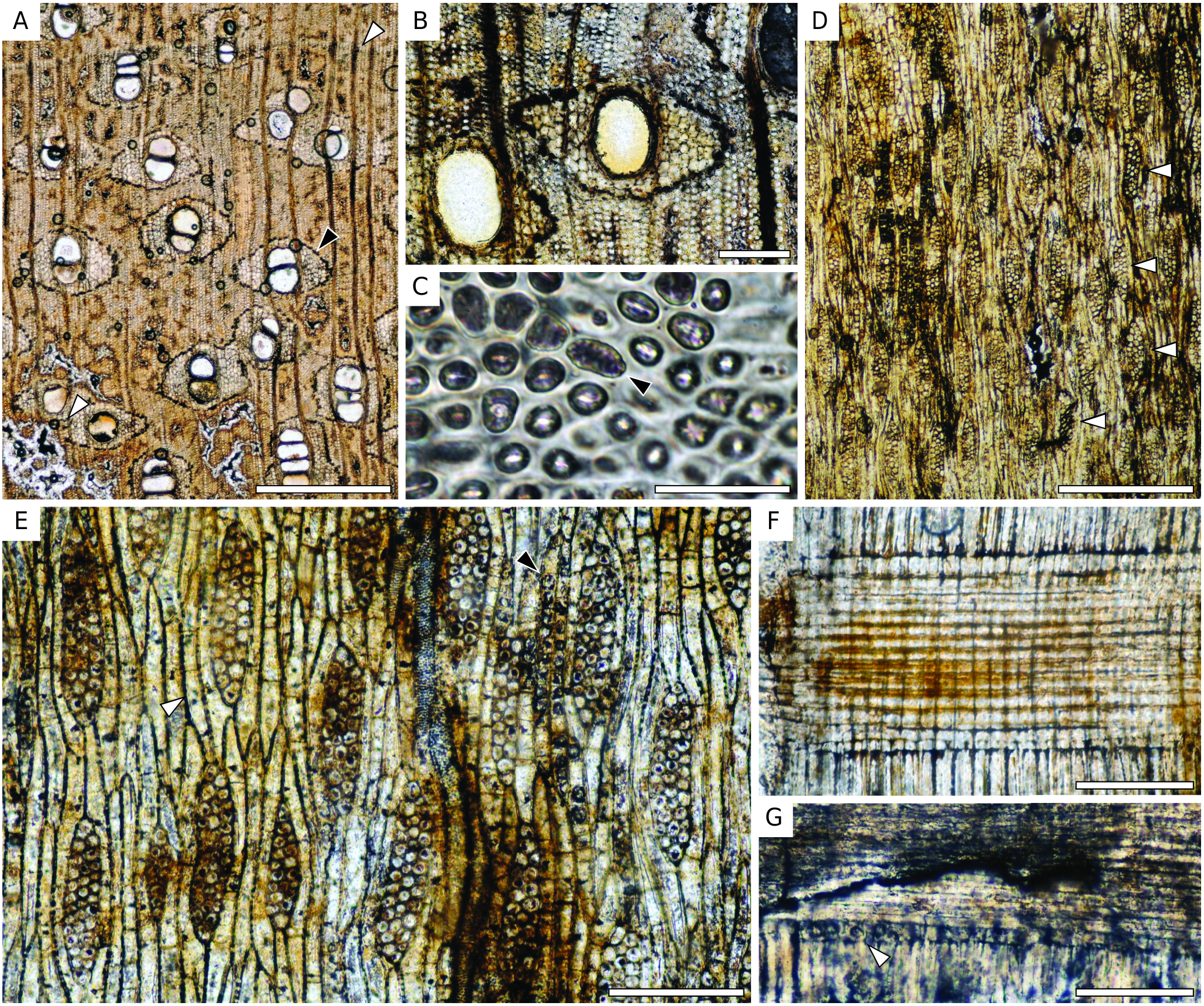
Wood diffuse-porous.Growth ring boundaries distinct, marked by marginal parenchyma bands ( Fig. 8 View FIG ). Vessels solitary (55%) as well as in radial groups of 2-4 (45%) ( Fig.8A View FIG ), round to oval, 1-5 per mm² (average: 3); tangential diameter 120-275 µm (average:185 µm).Tyloses absent.Vessel elements 120-370 µm (average: 225 µm) long. Perforation plates simple. Intervessel pits alternate, polygonal shaped, 4-7 µm (average: 6 µm) wide ( Fig. 8C View FIG ). Parenchyma paratracheal lozenge-aliform up to 13 cells wide for each wing and 5 cells around vessels, also confluent joining 2-4 vessels or groups of vessels ( Fig. 8A, B View FIG ), marginal bands 4-5 cells wide, sometimes surrounding narrower vessels; parenchyma cells 35-160 µm (average: 90 µm) long, 15-40 µm (average: 26 µm) wide; around 3-5 cells per parenchyma strand ( Fig. 8E View FIG ), crystals in chambered parenchyma cells at the margin of the aliform parenchyma ( Fig. 8E View FIG ), crystals are visible as black dots on the transversal section ( Fig. 8A, B View FIG ); possibly some rare diffuse parenchyma cells. Rays 1- to 3- seriate (mostly 3) ( Fig. 8E View FIG ), a very faint storied tendency in some parts of the section ( Fig. 8D View FIG ), 5-7 rays per mm (average: 6), 140-370 µm (average: 245 µm) or 8-21 cells high, mostly homocellular made of procumbent cells ( Fig. 8F View FIG ), rarely 1 row of marginal square (or larger procumbent) cells with sometimes crystals in them ( Fig. 8G View FIG ). Fibres non-septate, 7-30 µm (average: 19 µm) wide, thin-to-thick walled (lumina 1.6 times the double wall thickness in average).
DISCUSSION
This specimen is characterized by: 1) diffuse-porous wood, 2) exclusively simple perforation plates; 3) homocellular and 2- to 3-seriate rays; 4) aliform to aliform-confluent parenchyma; 5) marginal parenchyma; and 6) storied ray tendency in places. These combined features are shared with some Fabaceae (Baretta-Kuipers 1981; Gasson et al. 2003; Evans et al. 2006; InsideWood 2004 -onward), and in particular with many traditional Caesalpinioideae because most of the genera have heterocellular rays as well as rays more than 500 µm high and the number of cells per parenchyma strand often goes up to 4 and even more (compared to hardly exceeding 2 for the traditional Papilionoideae subfamily). Traditional Mimosoideae are ruled out as many genera have septate fibres (about 30 %), exclusively homocellular rays with small ray cells, rare storied structures and no crystals in ray cells. A comparison with Caesalpinioideae genera reveals that our specimen looks similar to the genera Afzelia and Intsia ( Gasson et al. 2003; InsideWood 2004 -onward), especially for their well-defined aliform parenchyma, sometimes confluent, their marginal parenchyma bands, the distinct crystals present in the margin of the aliform parenchyma (even in transverse section), and the strands commonly from 2 to 5 cells long. Both genera are hardly distinguishable based on xylological characters ( Prakash 1966; Müller-Stoll & Mädel 1967; Gasson et al. 2003; Ogata et al. 2008) although they have a different geographical distribution ( Léonard 1950). Afzelia is a genus of Africa and South-East Asia (mostly in Africa) ( Léonard 1950; POWO 2019). Intsia is a South and South-East Asian genus, with species also present in Australia, Madagascar or Tanzania ( POWO 2019). Based on a comparison with some species of Afzelia and Intsia from the InsideWood (2004 -onward) database and the Xylarium of the MNHN (specimen MNHN-P-P00396553), our specimen shows the closest affinity with Intsia bijuga (Colebr.) Kuntze because of the clear margin of crystalliferous aliform parenchyma cells, a tendency to irregular storied rays, clear marginal bands of parenchyma often in contact with small vessels and the fequency of vessels. Although, this species does not display any marginal square ray cells with crystals, this feature is only sporadically observed in our fossil and should not be used as a distinctive feature.
Based on their similar wood anatomy, fossil wood resembling both Afzelia and Intsia are described under the genus Pahudioxylon ( Chowdhury et al. 1960) . When compared with fossil species of this genus (a synthetic table is provided by Feng et al. 2015), all features of our fossil are compatible with the wide diagnosis of P. bankurensis . Our specimen is thus attributed to P. bankurensis .
Intsia trees are tropical and live in wet evergreen forests, with rainfall higher than 2000 mm per year, and is associated with Anisoptera Korth. and Hopea Roxb. ( Soerianegara & Lemmens 1993) . Intsia are mostly found in coastal habitat, near beaches, mangroves ( Loo & Tan 1997) and sometimes in peatlands ( Tanjung et al. 2020). Intsia bijuga is found along coasts, river edges, tidal or temporarily inundated places (sometimes salty water), in mangroves and back-mangroves, primary and secondary forests, up to 600 m altitude ( Soerianegara & Lemmens 1993; Hou et al. 1996; Orwa et al. 2009). Its seeds are sea-dispersed ( Lewis et al. 2005).
| G.S.I |
Conservatoire et Jardin botaniques de la Ville de Genève |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Pahudioxylon bankurensis Chowdhury, Ghosh & Kazmi
| Gentis, Nicolas, Licht, Alexis, Boura, Anaïs, Aung, Dario De Franceschi Zaw Win Day Wa & Dupont-Nivet, Guillaume 2022 |
Pahudioxylon bengalensis
| GHOSH P. K. & ROY S. K. 1982: 52 |
Pahudioxylon indicum
| PRAKASH U. 1979: 66 |
Pahudioxylon assamicum
| PRAKASH U. & TRIPATHI P. P. 1975: 63 |
Pahudioxylon welkitii
| LEMOIGNE Y. & BEAUCHAMP J. 1972: 336 |
Pahudioxylon deomaliense
| PRAKASH U. 1965: 433 |
Pahudioxylon sahnii
| GHOSH S. & KAZMI M. H. 1961: 96 |
Pahudioxylon bankurensis
| CHOWDHURY K. & GHOSH S. & KAZMI M. H. 1960: 22 |
Albizzioxylon sahnii
| RAMANUJAM C. G. K. 1960: 118 |