Pseudopoda
|
publication ID |
https://doi.org/ 10.11646/zootaxa.3999.3.3 |
|
publication LSID |
lsid:zoobank.org:pub:A97BB1A1-0536-465B-9222-2209A7B07027 |
|
DOI |
https://doi.org/10.5281/zenodo.5680191 |
|
persistent identifier |
https://treatment.plazi.org/id/03B7B251-FF92-FF90-FF0F-FADF0362FE6A |
|
treatment provided by |
Plazi |
|
scientific name |
Pseudopoda |
| status |
|
Pseudopoda View in CoL View at ENA wu spec. nov.
Figs 115–130 View FIGURES 115 – 123 View FIGURES 124 – 129 View FIGURE 130
Type material. CHINA: Sichuan: Holotype: male (PJ 3489) Lugu Lake, 27°46’10.04’’ N, 100°45’52.47’’E, 3100 m, extremely dry (secondary) Pinus forest, J. Martens leg., by hand, 27 May 2011 ( SMF).
Paratypes: 1 female (PJ 3490, SD 1134), with same data as for holotype ( SMF). Yunnan: 1 male (PJ 3491, SD 1135), with same data as for holotype, but: 27°37’55.90’’ N, 100°49’04.06’’E, 3300 m, 28 May 2011 ( IZCAS).
Etymology. The species name is derived from the Chinese word “ ⊥Γ ⅛], wú” meaning “without” and referring to the fact that this new species is the first one of the genus Pseudopoda with an almost entirely reduced conductor; term in apposition.
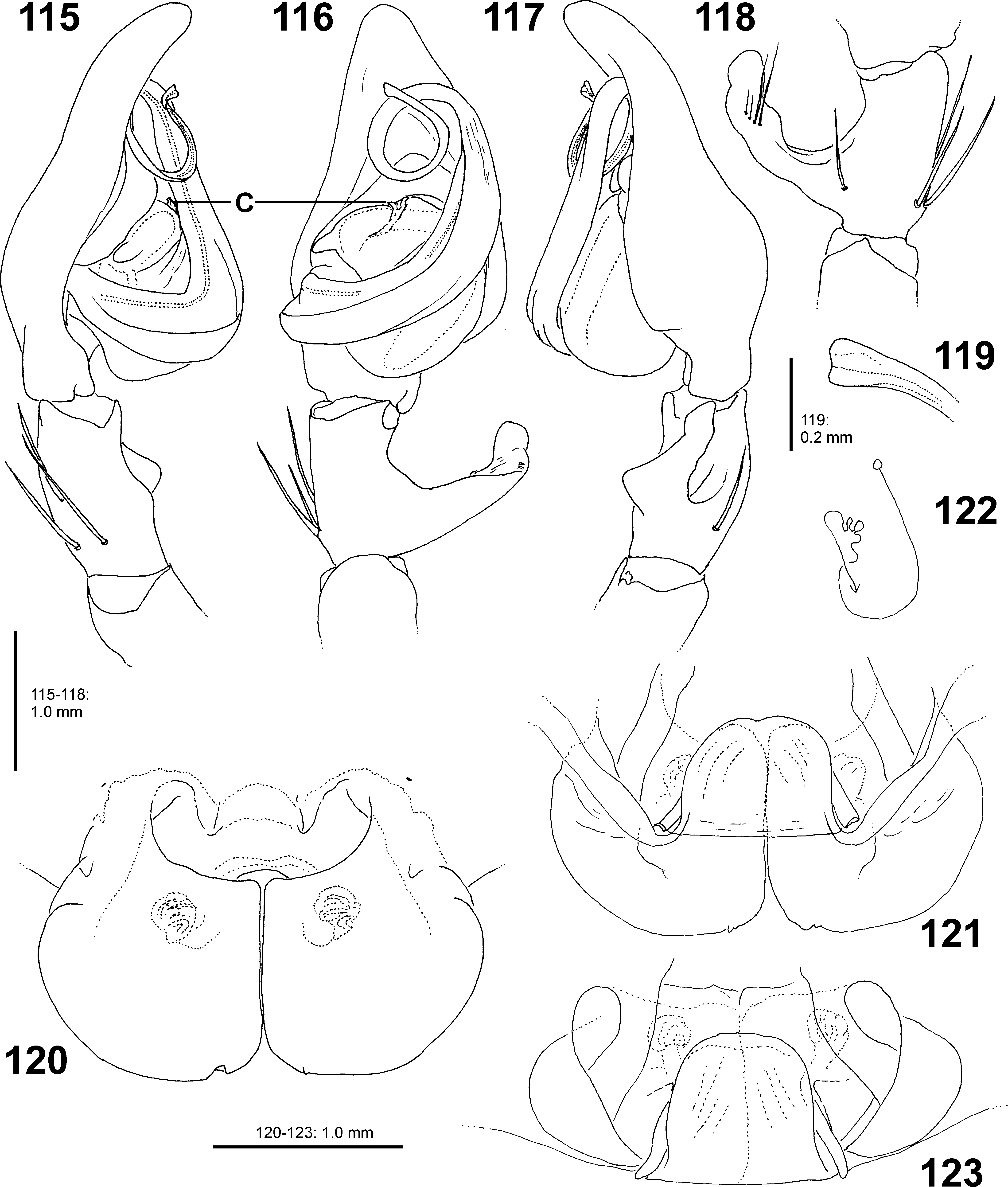
Diagnosis. Medium sized representatives of Pseudopoda , body length of males 11.0–11.6, of females 12.6. Male palp of the new species differs from all other species of the genus by its C reduced to a small inconspicuous structure ( Figs 115–116 View FIGURES 115 – 123 : C). Proximal part of E similar to that of P. signata or related species, but distinguished by 1) Distal end of E with one coil, 2) RTA with simple tip, i.e. not divided in two apices ( Figs 115–119 View FIGURES 115 – 123 ). Females resemble those of P. signata in anterior margins of LL broadly U-shaped and ventral windings situated close to anterior margin of LL, but are distinguished by 1) Ventral windings narrower and accumulated in a small spherical structure, 2) First windings diverging posteriorly more strongly, 3) LL more rounded posteriorly ( Figs 120–122 View FIGURES 115 – 123 ).
Description. Male (holotype): DS length 5.5, width 5.1, AW 3.0, OS length 6.1, width 3.6. Eyes: AME 0.28, ALE 0.39, PME 0.30, PLE 0.40, AME–AME 0.19, AME–ALE 0.11, PME–PME 0.36, PME–PLE 0.42, AME– PME 0.40 ALE–PLE 0.42, clypeus height at AME 0.38, at ALE 0.33. Spination: palp: 131, 001(small), 2101; legs: femur I 313(324), II 323, III 3 (2)33, IV 33 (2)1; patella I–III 0 0 1, IV 001(0); tibia I–IV 2026; metatarsus I 2014, II 2024, III 3025, IV 3037(6). Metatarsus IV ventrally with several bristles along entire length, metatarsus III with few bristles in proximal half. Leg formula: 2143. Measurements of palp and legs: palp 8.1 (2.6, 1.2, 1.3, -, 3.0), leg I 24.3 (6.5, 2.8, 6.6, 6.2, 2.2), leg II 26.5 (7.2, 2.9, 7.3, 6.9, 2.2), leg III 22.1 (6.2, 2.5, 5.8, 5.6, 2.0), leg IV 23.2 (6.5, 2.2, 6.0, 6.3, 2.2). Cheliceral furrow with ca. 25 denticles in restricted patch at largest anterior tooth. Promargin of chelicerae with 3 teeth, retromargin with 4 teeth.
Palp as in diagnosis ( Figs 115–119 View FIGURES 115 – 123 ). RTA arising proximally from tibia, with distinct ventral hump in retrolateral view. Cymbium more than twice as long as tibia, with prolateral and retrolateral bulge. Tegulum with proximal bulge, prolatero-distal elongate nipple. Spermophor wide in tegulum, very narrow within E. E arising in 9-o’clock-position from tegulum, strong, in distal coil narrower, E tip widened, disto-prolaterad. C arising in 12- o’clock-position from tegulum, small, not reaching embolus tip as in other Pseudopoda species.
Colouration ( Figs 124–126 View FIGURES 124 – 129 ): Yellowish to reddish-brown with dark brown markings. DS with fovea and striae marked, the latter with rows of dots, margins slightly darker than rest. Sternum with 6 dots at coxae I–III. Coxae and femora ventrally indistinctly dotted, labium and gnathocoxae distally lighter. Chelicerae reddish-brown, without pattern. Legs with coxae to tibiae yellowish, metatarsi and tarsi reddish-brown, femora with spine patches, tibiae proximally with few irregular patches. OS dorsally with heart patch and lateral markings in anterior half; in posterior half with dark chevrons and write transversal line. OS ventrally with several spots.
Female (paratype): DS length 5.4, width 4.8, AW 3.0, OS length 7.2, width 4.7. Eyes: AME 0.24, ALE 0.35, PME 0.29, PLE 0.35, AME–AME 0.23, AME–ALE 0.09, PME–PME 0.38, PME–PLE 0.45, AME–PME 0.36, ALE–PLE 0.37, clypeus height at AME 0.38, at ALE 0.30. Spination: palp: 131, 101, 2121, 1014; legs: femur I–II 323, III 323(2), IV 331; patella I–III 0 0 1, IV 000; tibia I–IV 2026; metatarsus I–II 2024, III 3025, IV 3037. Metatarsus IV ventrally with many bristles along entire length, metatarsus III with few bristles in proximal half. Leg formula: 2143. Measurements of palp and legs: palp 6.9 (2.0, 1.1, 1.5, -, 2.3), leg I 18.7 (4.9, 2.4, 4.8, 4.1, 1.5), leg II 19.1 (5.5, 2.6, 5.1, 4.4, 1.5), leg III 16.1 (4.8, 2.2, 4.0, 3.7, 1.4), leg IV 17.5 (5.3, 1.8, 4.3, 4.4, 1.7). Cheliceral furrow with ca. 45 denticles in restricted patch close to median anterior tooth. Promargin of chelicerae with 3 teeth, retromargin with 4 teeth. Palpal claw with 8 teeth.
Copulatory organ as in diagnosis ( Figs 120–122 View FIGURES 115 – 123 ). Epigynal field wider than long, with trilobate anterior margin and anterior bands inconspicuous and included in epigynal field. Part of LL extending posteriorly beyond epigastric furrow longer than part anterior of epigastric furrow. Dorsal part of LL developed as prominent anterior hump with rills. Fertilisation ducts latero-posteriad.
Colouration ( Figs 127–129 View FIGURES 124 – 129 ): As in male but generally markings more distinct. Sternum and coxae ventrally with few indistinct dots. OS dorsally as in male paratype (see variation), transversal line with anterior margin distinct, posterior margin frayed; OS ventrally with narrow V-shaped marking and few additional dots.
Variation. Males (n=1): DS length 4.9, OS length 6.1. Spination: femur I 322(3), II 323, III 322, IV 331; patella I–II 101, III 0 0 1, IV 001(0); tibia 2026; metatarsus I–II 2024, III 3025, IV 3037. Colouration: Markings more distinct ( Figs 124–126 View FIGURES 124 – 129 ) than in holotype. DS with dots and markings forming 2 indistinct longitudinal bands. Sternum with several dots also centrally. OS dorsally with darker heart region accompanied by light areas and light patches, transversal line in posterior half very distinct. OS ventrally with dots fused to V- to U-shaped marking, in front of epigastric furrow with 2 parallel markings.
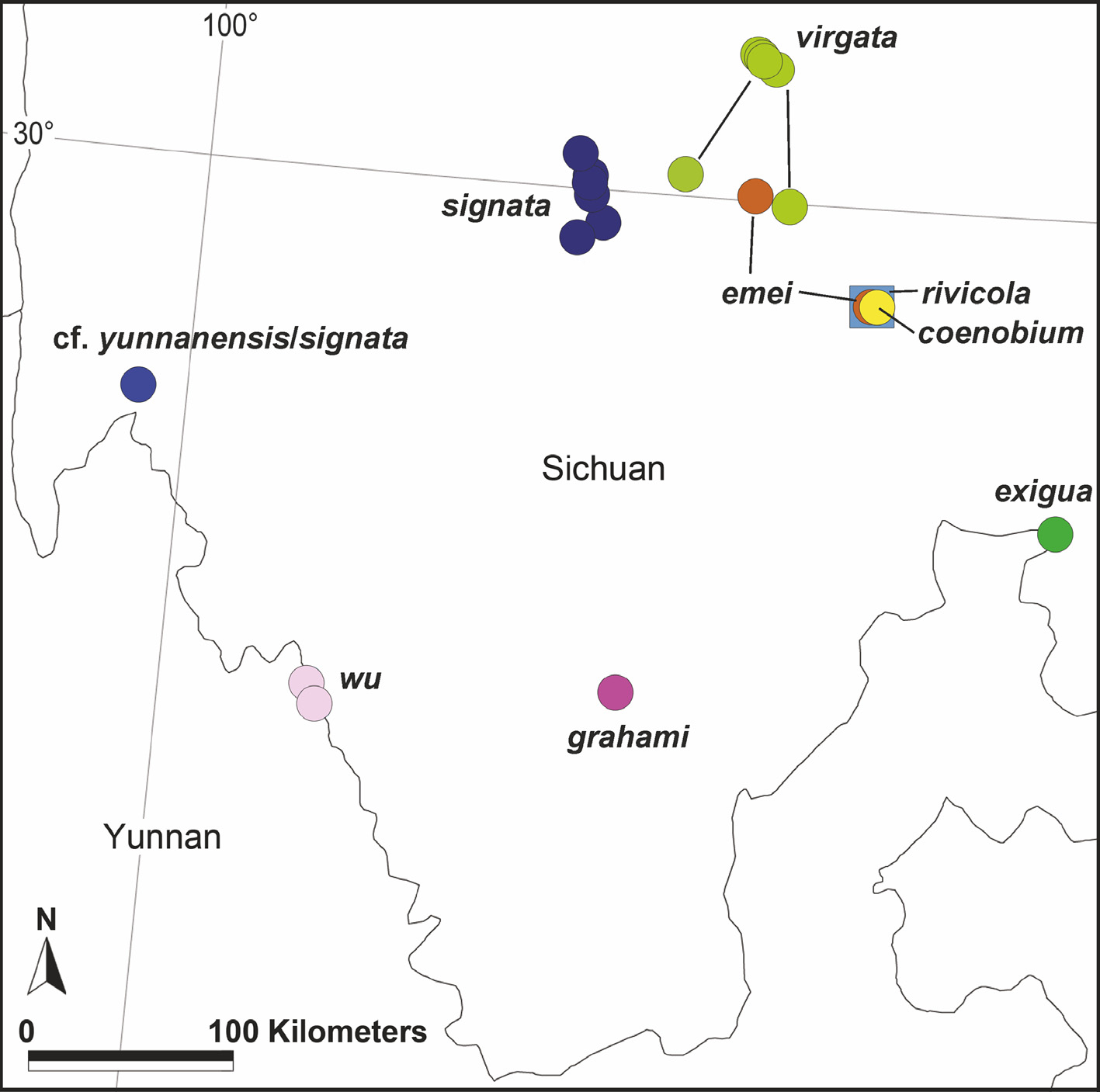
Distribution. China: Yunnan, Sichuan. Known from the type locality in South Sichuan and a nearby locality in North Yunnan ( Fig. 130 View FIGURE 130 ).
Notes. A reduction and arrangement of a conductor as exhibited in P. w u spec. nov. is an extraordinary character only known from few species within the Sparassidae (presently with 1148 species known; World Spider Catalog 2015). In several Sparassidae the conductor is developed as sheath-like structure, most likely functioning as guiding support during the copulation process. This type is known for instance from the genera Heteropoda Latreille, 1804 , Yiinthi Davies, 1994 , Gnathopalystes Rainbow, 1899 and Damastes Simon, 1880 . In many species the conductor is membranous and more or less spoon-shaped with the embolus tip close to the conductor’s distal part, e.g. in Pseudopoda , Sinopoda Jäger, 1999 , Spariolenus Simon, 1880 , Leucorchestris Lawrence, 1962 or Palystella Lawrence, 1928 . In the Deleninae, the conductor is spiral shaped or screw-like and supports the wound embolus at least in its resting position (e.g., in Neosparassus Hogg, 1903 ). In other species it is reduced and sits attached with a broad base directly at the tegulum, but still with the embolus tip close to it (many species of Olios Walckenaer, 1837 ). In species of, for instance, the genera Bhutaniella Jäger, 2000 , Cebrennus Simon, 1880 , Rhitymna Simon, 1897 , Vindullus Simon, 1880 , and in Origes pollens Simon, 1897 , the conductor is almost entirely or entirely reduced. Pseudopoda wu spec. nov. represents a morphological and most likely functional novelty within the genus. It might be explained by the rigid embolus that does not need any supportive structure. Behavioural observations and analyses are necessary to support this hypothesis.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.