Cassida rubiginosa Müller, 1776
|
publication ID |
https://doi.org/10.5281/zenodo.182782 |
|
DOI |
https://doi.org/10.5281/zenodo.6228615 |
|
persistent identifier |
https://treatment.plazi.org/id/03BD1870-C013-9E50-20D7-2CF07255AE9A |
|
treatment provided by |
Plazi |
|
scientific name |
Cassida rubiginosa Müller, 1776 |
| status |
|
Cassida rubiginosa Müller, 1776 View in CoL
Identification. Live adults of C. rubiginosa are vivid green on dorsal surface, often with yellowish elytral margins, and are black underneath. This green pigment is ephemeral, and dried specimens preserved in collections quickly turn brownish. Its medium size ( 6-8 mm), confused elytral punctures, and the vivid light green color make adults recognizable immediately, even by non-experts ( Fig. 1).
The first instar larva was described and illustrated by Świçtojańska (2004). Egg bursters are absent ( Cox 1994a). The mature larva was described by Paterson (1931, sub Cassida viridis ) and illustrated by Peterson (1951). It can be identified using the key to species of the British cassidine larvae ( Van Emden 1962).
Descriptions and illustrations of the pupa of C. rubiginosa , are found in Paterson (1931) ( sub C. viridis ) and Palij & Klepikova (1957). A key, including C. rubiginosa , is provided by Palij & Klepikova (1957).
History and distribution. Cassida rubiginosa was first discovered in North America in 1902 in Lévis, near Québec City, where beetles were observed to be defoliating burdock ( Arctium minus (Hill.) Bernh. , Asteraceae ) ( Fyles 1902, 1903; Roy 1902). Schaeffer (1903, 1904a, 1904b) contributed to clarify its identity. It was subsequently reported by Brown (1940) on burdock, in Montréal, at Knowlton and Brome in the Eastern Townships of Québec, and in Shediac, New Brunswick. The species is now widely established both across Canada from Alberta east to New Brunswick, and in the United States from Maine south to Virginia and west through Ohio to Wisconsin and South Dakota ( Riley et al. 2003). In the Old World , it is found throughout continental Europe (not yet recorded in the Netherlands) from Fennoscandia south through the Baltic republics to Greece and Spain, in Great Britain, and across Siberia to the northern Far East of Russia ( Audisio 2005b).
Biology. Cassida rubiginosa has been well studied in Europe by Kleine (1917a) and Kosior (1975). In Eastern Canada, C. rubiginosa is found in habitats where Canada thistle and burdocks grow, such as agriculture lands, pastures, abandoned fields, sparsely wooded meadows, neglected orchards, clear cut areas, or similar open and/or disturbed habitats.
The species is univoltine in Ontario and Québec (LeSage, personal observations), in Virginia ( Ward & Pienkowski 1978a), and very likely so in the Maritime Provinces. Hibernation is in the adult stage. In Poland, Kosior (1975) observed a migration of adults from thistles to forest floor litter in the fall. In Virginia, Spring & Kok (1999) found that leaf litter was the preferred hibernaculum of field caged adults, and inability to access preferred hibernating locations combined with fluctuating winter temperatures may result in the survival of less that one in four beetles. Adults leave their winter shelters in early spring. The earliest specimens in the Canadian National Collection (CNC) were collected on April 2 in southern Ontario and on May 15 in the Ottawa area. The earliest adults collected in the Maritime Provinces are from May 31 (Kentville, Nova Scotia).
Females began to oviposit between mid-March and April in Virginia ( Ward & Pienkowski 1978a). In Europe, Kosior (1975) stated that oviposition started 3-7 days after mating according to temperature, day length, rain, and wind. Eggs are usually laid on the underside of the leaves, more rarely on the upper surface, and sporadically on the stem.
Ward & Pienkowski (1978a) referring to the midrib, observed that 74% of the oöthecae were deposited on the abaxial surface, and 18% on the adaxial surface. The number of eggs in oöthecae is variable but is usually three, although oöthecae with only one egg are not rare ( Kosior 1975). In Virginia, Ward & Pienkowski (1978a) found an average of 4.6 eggs per oötheca, and Spring & Kok (1997) found an average of 61.1 oöthecae laid per individual female over a 15-week period. In Maryland, Tipping (1993) observed that many oöthecae were laid on leaves closest to the soil. In laboratory rearing, the fecundity averaged 815 eggs per female; eggs hatched in two weeks at 18°C, in four days above 32°C ( Ward & Pienkowski 1978a). The egg-laying period lasted 12 weeks ( Kosior 1975).
The eggs are usually laid on the lower surface of the leaves. Each egg is enclosed in secretion which turns from whitish to brown in about ten minutes, and finally, the oötheca is closed with a layer of excreta ( Engel 1935; Bibolini 1973). Excremental coverings are thought to conceal eggs or act as physical or chemical barriers to repel natural enemies ( Muir & Sharp 1904; Damman & Cappucino 1991; Olmstead 1996). Egg deposition, coverings, and oviposition were reviewed by Hilker (1994), Olmstead (1994), and Selman (1994).
Damman & Cappucino (1991), who studied this form of double defense (egg clumping and excrement cover) in the hispine Microrhopala vittata (Fabricius) , found that the fecal covering significantly reduced egg mortality. Since the parasites generally attacked the bottom egg, and while the predators rarely penetrated the fecal covering, egg masses were virtually protected against all natural enemies likely to pose a threat. Such a protection can be expected in Cassida rubiginosa which also lays small oöthecae of a few eggs ( Kosior 1975; Ward & Pienkowski 1978a).
There are five larval instars in C. rubiginosa , each one easy to recognize by its exuviae attached to the caudal fork together with excrement. In addition to the shield and fork, the larvae possess lateral spines, or scoli, that are used as sensory organs, and when they are stimulated the shield is moved to cover the body ( Olmstead 1996). This distinctive structure, made of dried exuviae and accumulated fecal wastes, attracted the attention of early naturalists (ex. Réaumur 1737)."Stercoraceous parasol" ( Walsh & Riley 1869), "faeces pack" ( Rabaud 1915b), and "frass mask" ( Engel 1935), were used to described this structure but the term "fecal shield" popularized by Eisner et al. (1967) is now in use. The smallest exuviae (first instar) is attached at the extremity of the fork, the largest at base, each sliding on the branches of the new fork after each molt ( Rabaud 1915b). As the larvae feed again, the newly produced faeces accumulate behind the previous ones.
Consequently, the fecal shield grows and moves forward at each molt with the result that before pupation, this pack contains the whole series of exuviae and all the faeces produced by the larvae during their life ( Rabaud 1915b). The shield is also highly maneuverable and can be moved to cover areas of the body that are stimulated (Olmstead 1994). The anus is extrudable. Consequently, it can be projected and curved over the back and, through the aid of the fork and of some of the lateral spines, it forms the protective fecal shield ( Walsh & Riley 1869).
The role of the shield has been the subject of several hypotheses. According to Frisch (1720), cited by Engel (1935), the fecal shield protects the body from rain and pests, while for Huber (1846) and others, it is a protective shelter from insolation, but this assumption was recently rejected ( Bacher & Luder 2005). Larvae covered with their fecal shield are similar to droppings which may confuse large predators like birds, but not insect predators. For Eisner et al. (1967), ants are undoubtedly among its chief natural enemies. They were always seen foraging in large numbers in the dense herbage that included the beetle's host plants. Confronted with the inanimate shield rather than with the body of the larvae, they were quick to lose interest in such potential prey. Larvae deprived of their shield were bitten and killed, or carried away live into ant nests. On the other hand, these authors noted that the shield did not protect larvae from all predators. For instance, tests with a lycosid spider invariably resulted in the larvae being killed, an observation already made by Engel (1935) with the larvae of Cassida viridis Linnaeus. Engel (1935) also stated that the violent movements of the larvae when disturbed actually constitute a protection insofar as they chase away enemies. According to Franz (1941), the shield can protect against parasites although its effectiveness is not absolute.
Eisner et al. (1967) argued that the fecal shield of C. rubiginosa larvae was effective in deterring ants in laboratory tests. Vencl et al. (1999) found that shields were necessary for the survival of the larvae of Plagiometriona clavata (Fabricius) and that the chemicals derived from their nightshade host plant ( Solanum dulcamara L.) formed a deterrent barrier against the ant ( Formica subsericea Say ) attacks. They concluded that the incorporation of deterrent metabolites in shield defenses represented responses to selection from invertebrate predators. The role of these allelochemicals from host plants and other antipredator devices was reviewed by Blum (1994).
Olmstead & Denno (1992) estimated that the cost of bearing fecal shields was minimal. Being made of recycled waste products, fecal shields provide an inexpensive mode of protection from certain natural enemies. Tipping (1993) noticed that smaller larvae were very susceptible to generalist predators, especially coccinellids. However, Olmstead & Denno (1993) observed that predators with short mandibles, such as coccinellids, were effectively deterred by the larval shields of cassidines whereas nabids and pentatomids with long piercing mouthparts easily circumvented the shield defense. Consequently, a particular kind of defense does not provide complete protection against generalist predators in natural habitats.
Müller (2002) found that feeding by the lacewing larvae Chrysoperla carnea (Stephens, 1836) was not influenced by fecal shields, and concluded that the effectiveness of shields of tansy-feeding cassidine larvae ( C. denticollis and C. stigmatica ) varied with predators and might be based more on mechanical than on chemical modes of action. Shields also proved to be useless against the paper wasp Polistes domidulus Christ and ultraviolet radiation ( Bacher & Luder 2005).
Larvae of C. rubiginosa are not very mobile and tend to remain on the same ramet throughout their development. The main larval feeding activity is postponed to a period when the nitrogen content of the leaves had dropped to 50% of its initial value, but when the host plant biomass had increased by 30% ( Obermaier & Zwölfer 1999). Early-season larval feeding had no measurable effect on Canada thistle growth while late-season feeding significantly reduced shoot growth ( Bacher & Schwab 2000).
When full grown, the larvae discard their load of excrement and exuviae, attach their last 2-3 ventral abdominal segments to the plant by means of a sticky secretion, and pupate there. The preferred site for pupation is along the midrib of leaves or on the main plant stalk ( Ward & Pienkowski 1978a). According to these authors, the pupation period averaged 9.6 days at 17.8°C and 3.5 days at 32.5°C. In Germany, adults of the new generation appear in mid-July onwards with a peak in late August ( Obermaier & Zwölfer 1999), an emergence period that roughly corresponds to what we have observed in Eastern Canada.
Larvae and adults do not eat leaves from the edges but attack them from a surface, usually the upper face. Rabaud (1915a) stated that the under face remained untouched but this is true only for younger larvae. Adults and larger larvae can skeletonize and severely damage the leaves ( Batra et al. 1981).
According to Koji & Nakamura (2006) in Japan, the population of C. rubiginosa was characterized by a high density of over-wintered adults and a low variability in annual population density for both over-wintered and new adults. This exceptional stability for a herbivorous insect population was attributed to the longevity of the adults, some being still alive after three years.
Parasitism. Parasitism of Chrysomelidae by Hymenoptera and Diptera was extensively reviewed by Cox (1994b), and this topic is treated in detail for several European Cassida species, including C. rubiginosa , by Kosior (1975). The following paragraphs primarily concern the North American populations.
According to Tschanz et al. (2005), the degree of exposure of C. rubiginosa larvae on plants had a significant effect on predation but the rate of parasitism was equal on exposed or hidden plants. Ten years after the release of this cassidine in southwestern Virginia, Ang & Kok (1995) estimated that parasitism and incomplete development were two major mortality factors.
In North America, six species of parasitoids have already been identified from larvae and pupae of C. rubiginosa , but none from eggs and adults ( Ward & Pienkowski 1978b; Tipping 1993; Ang & Kok 1995; Olmstead 1996). In Europe, Girault (1914) reported the hymenopteran eulophid Oomyzus gallerucae Fonscolombe ( sub Tetracampe galerucae Jonsc.) as an egg parasite but questioned its identity. Delucchi (1960) listed the mymarid Anaphes pannonica Soyka as an egg parasite of C. rubiginosa . According to Muir & Sharp (1904), the fecal covering of the oöthecae do not protect against parasites.
Tetrastichus rhosaces (Walker) View in CoL is a gregarious obligate European eulophid endoparasitoid of the larvae and pupae ( Kaufmann 1933; Graham 1991). According to Ward & Pienkowski (1975, 1978b), it is the most important parasitoid of C. rubiginosa View in CoL in North America, accounting for 14.2-23.8% of the total parasitism. The numbers of parasites reached an average of 9.9 per host, most of them being commonly located in the anal or cephalic regions. In larvae, signs of parasitism were not evident until the last larval instar when parasitized larvae showed a noticeable discoloration. Affected pupae became slightly darker. Tipping (1993) did not find it in Maryland in his field study of C. rubiginosa View in CoL on Canada thistle. Ang & Kok (1995) assumed that the Aprostocetus View in CoL sp. collected by them in southwestern Virginia was T. rhosaces View in CoL which parasitized up to 9% of the larvae and as much as 96% of the pupae.
The endoparasitoid solitary tachinid fly, Eucelatoria dimmocki Aldrich View in CoL , accounted for 2.7-7.5% of the total parasitism in a study of the parasites in northern Virginia ( Ward & Pienkowski 1978b), 0-2% in southwestern Virginia (Ang & Kok 1995), and 19.5% in Maryland ( Tipping 1993). It is a native polyphagous parasitoid of larvae and pupae which has been previously recorded from several other cassidine hosts including Chelymorpha cassidea (Fabricius) View in CoL , Plagiometriona clavata View in CoL (Fabricius, sub Deloyala clavata ), Charidotella sexpunctata bicolor View in CoL (Fabricius, sub Metriona bicolor ), and Agroiconota bivittata View in CoL (Say, sub Metriona bivittata ) ( Aldrich 1932; Stearns 1933).
Ward & Pienkowski (1978b) measured only 0.7-2.1% of parasitism by the hymenopteran chalcidid Conura albifrons (Walsh, sub Spilochlacis albifrons ). It is a polyphagous, solitary endoparasitoid. In North America, Musesebeck et al. (1951) and Peck (1963) listed 38 other hosts in 14 families in 4 orders. This chalcid is also a hyperparasite of Tachinidae and Braconidae ( Ward & Pienkowski 1978b).
Tipping (1993) reported that Conura side (Walker, sub Spilochalcis side ) emerged from 1.4% of the pupae. It is another polyphagous, solitary endoparasitoid with at least 45 known hosts, mostly Lepidoptera ( Peck 1963). In western Virginia, this parasitoid ( sub Cornura torvina (Walsh)) appeared late in the season and accounted for the second highest rate of parasitism with 0-8% of the pupae parasitized (Ang & Kok 1995).
The hymenopteran eupelmid Macroneura vesicularis (Retzius, sub Eupelmella vesicularis ) is another polyphagous, solitary endoparasitoid, but of negligible importance in C. rubiginosa . Its level of parasitism in this species did not exceed 0.1% ( Ward & Pienkowski 1978b). At least 70 additional hosts, in various orders and families, are known for this parasitoid ( Muesebeck et al. 1951; Krombein 1958; Peck 1963).
In their two-year study of the parasites of C. rubiginosa, Ward & Pienkowski (1978b) recovered only one individual of the solitary ichneumonid, Itoplectis conquisitor (Say) . In Maryland, it accounted for 1.4% of the total parasitism ( Tipping 1993). In southern Virginia, it was found at all sites but without exceeding 2% of the hosts affected (Ang & Kok 1995). This solitary parasite is polyphagous with over 100 hosts recorded in North America ( Muesebeck et al. 1951; Krombein 1958; Peck 1963; Krombein & Burks 1967).
Parasitic nematodes are known but not yet reported in North America. In Europe, Hexamermis sp. (96%) is the most important parasite of larvae and pupae, whereas Mermis sp. (2%) and Agamermis sp. (2%) ( Mermithidae ) are occasionally found; adults were primarily parasitized by Mermis sp. (84%) and Hexamermis (15%) ( Kosior 1975). Loktin & Ivanova (1970), using suspensions of Neoaplectana sp. ( Steinernematidae ) larvae, killed 33-60% of the larvae of C. rubiginosa in the field.
Predation. Very little is known about the predators of C. rubiginosa in North America, although the subject has been well treated in Europe ( Kosior 1975; Olmstead 1996). In Virginia, Ward & Pienkowski (1978b) reported that the phalangiid Leiobunum sp., the reduviids Arilus sp. and Sinea diadema (Fabricius) , the pentatomid Podisus maculiventris (Say) , and the neuropteran chrysopid Chrysoperla ( sub Chrysopa) carnea (Stephens) fed on C. rubiginosa in the field but did not specify the life stage preyed upon. In Maryland, Olmstead & Denno (1993) studied the predation of some mandibulate, piercing/sucking, and chelicerate predators of Charidotella sexpunctata bicolor (Fabricius) and of Deloyala guttata (Olivier) . The selected mandibulate predators were the coccinellid beetles Coleomegilla maculata (DeGeer) , Coccinella septempunctata Linnaeus , and the carabid Lebia fuscata Dejean. Geocoris punctipes (Say) was a short rostrum species whereas the heteropteran nabid Nabis americoferus Carayon , and P. m a c u l i v e n t r i s had long beak. For the chelicerate predators, two spiders were selected: Oxyopes salticus Hentz (Oxyopidae) and a Phidippus sp. ( Salticidae ). The larvae were protected against the short mandibulate coccinellids but not from haustellate bugs which could pierce the shield or insert their long rostrum under the shield and feed. All these predators are generalists and common in the Northeast. Consequently, they are very likely to prey on C. rubiginosa larvae as well.
Research on insect predators of C. rubiginosa is currently ongoing in Europe. In Switzerland Schenk & Bacher (2002) estimated that the paper wasp Polistes dominulus Christ (Vespidae) was responsible for 99.4% of the predation on C. rubiginosa larvae, and thus may cancel the effect of mass releases of the beetle as biocontrol agent. Tschanz et al. (2005) in Switzerland, found that exposure significantly affected predation by this wasp, and consequently larvae on hidden plants were less likely to be killed than larvae on exposed leaves. Schenk & Bacher (2004), also in Switzerland, demonstrated that predation by Nabis mirmicoides Costa (Heteroptera: Nabidae ), and Chrysoperla carnea can be proved using the monoclonal antibody (Mab) CRL5-1.
Host plants. From the extensive literature review of Clark et al. (2004) it is clear that C. rubiginosa is polyphagous, but upon closer examination, it is apparent that it exhibits preferences for the Cardueae in the Asteraceae ( Zwölfer & Eichhorn 1966; Batra et al. 1981; Obermaier & Zwölfer 1999; Bacher & Schwab 2000).
In North America, it was first reported on burdock ( Arctium minus (Hill.) Bernh. ) from Lévis, near Québec City ( Roy 1902; Fyles 1902; Brown 1940). Peterson (1951) mentioned this plant species in his description of the larva from Ohio, whereas Wilcox (1954) in his treatment of leaf beetle fauna of this state, mentioned that it had been reported from Canada on thistle but did not give the sources of his information. Later, he added burdock to the known list of host plants ( Wilcox 1979).
Chagnon (1917, 1939, 1940), and Chagnon & Robert (1962) mentioned both thistle ( Cirsium sp.) and burdock ( Arctium sp.) as hosts in Québec but did specify which species was involved. From our own collecting, and on the basis of the specimens studied here, only A. minus and Cirsium arvense (L.) Scop. are true hosts of C. rubiginosa in eastern Canada.
In Saskatchewan, Maw (1976) reared C. rubiginosa from Cirsium arvense , Centaurea jacea , Arctium sp. and Carduus sp. In West Virginia, Hacker (1977a) first reported that larvae fed heavily on leaves of bull thistle ( Cirsium vulgare (Savi) Tenore ) at Mineral Wells, and later collected adults and larvae in various counties on Cirsium pumilum (Nutt.) Spreng. , Carduus crispus L., and Carduus nutans L. ( Hacker 1977b). In Maryland and Pennsylvania, Batra (1978) observed that C. rubiginosa was most abundant on Carduus nutans and C. acanthoides , but later reported its preference for Cirsium , Carduus , and Silybum , also mentioning some feeding on Cynara , Carthamus and other plants ( Batra et al. 1981). In Virginia, Ward & Pienkowski (1978a) listed five host thistles: bull thistle ( C. vulgare ), Canada thistle ( C. arvense ), field thistle ( C. discolor (Mühl.) Spreng. ), musk thistle ( Carduus nutans ), and plumeless thistle ( C. acanthoides L.). Peschken (1984a) stated that C. rubiginosa overwhelmingly fed on introduced weeds, with only one report on the native Cirsium muticum Michx. and C. discolor (Muhl. ex Willd.) Spreng.
Of the host plant genera in the Asteraceae reported by Clark et al. (2004), all except Pulicaria and Saussurea are found in the Maritime Provinces, and many species are widely distributed in the region ( Erskine 1960; Hinds 1986; Roland 1998).
Biocontrol potential. Canada thistle, known in Europe as the creeping thistle, is one of the world's worst weeds ( Holm et al. 1977; Hays 1991; White 1996). It is a very aggressive colonizer that quickly produces stands on lands disturbed by agriculture or other human activities. The weed is difficult to control because the root system can extend several feet down and spread extensively horizontally, and the roots can persist even when broken by plowing. Their prolific seed production, seed longevity, competitive ability, and the lack of natural enemies are additional reasons of the success of thistles ( Kok 1998).
The world catalogue of agents and their target weeds of Julien & Griffiths (1998) gives the origins of the weeds, the dates of release of control agents, their status, and the degree of control of the weeds. Closer to this region, the reviews of Kok & Gassmann (2002) on bull thistle, McClay (2002) on Canada thistle, and Gassmann & Kok (2002) on musk thistle in eastern United States, also includes information on the situation in Canada. In this country, the biological control attempts of weeds were reviewed four times. Cassida rubiginosa View in CoL was not mentioned in the first review ( McLeod 1962) . In the second review , for the years 1959-1968, Harris & Zwölfer (1971) stated that C. rubiginosa View in CoL larvae and adults skeletonized leaves and were capable of damaging the plumeless ( C. acanthoides View in CoL ) and nodding thistle ( C. nutans View in CoL ), but high parasitism seemed to prevent the build up of populations sufficient to control thistles. In the same review, Peschken (1971) mentioned that C. rubiginosa View in CoL was one of the most conspicuous enemies of Cirsium arvense View in CoL , but could not control the weed below the economic level. In a 1969-1980 review , the authors focused on the impact of the weevils Hadroplontus litura (Fabricius) View in CoL , Rhinocyllus conicus (Froelich) View in CoL , and Trichosirocalus horridus (Panzer) View in CoL , the flea beetle Altica carduorum (Guérin-Méneville) , the tephritid flies Urophora cardui (Linnaeus) View in CoL and U. stylata (Fabricius) View in CoL (Harris 1984; Harris & Wilkinson 1984; Peschken 1984a). Peschken (1984b) indicated that C. rubiginosa View in CoL was also present in the release pastures of H. litura View in CoL , together with the rust Puccinia punctiformis (Str.) Rohl. and the weevil Cleonus piger (Scopoli) , and consequently assumed that the decline of some thistle patches was caused by one or a combination of these agents. In the last review available, from 1981-2000, McClay et al. (2002) cited the previous works of Zwölfer (1969) in Europe and Ward & Pienkowski (1978a) in Virginia, but without new information on C. rubiginosa View in CoL in Canada.
The first concerted attempt at biological control of thistles was initiated in Virginia in 1969 against musk thistle. This program started with the introduction of a head-feeding weevil, Rhinocyllus conicus View in CoL , subsequently followed by the introduction of two rosette feeding weevils, Trichosirocalus horridus View in CoL and Hadroplontus trimaculatus (Fabricius) View in CoL ( Kok 1978, 1979). Cassida rubiginosa View in CoL was also considered as a potential biological agent.
In Saskatchewan, Maw (1976) noted that C. rubiginosa could be sufficiently numerous at times to defoliate thistles in Saskatchewan. Batra (1978) in Maryland, however, pointed out that the beetle did not significantly reduce the vigor of thistles although leaves could be extensively damaged in some areas. In their study of the stress caused by five organisms on Canada thistle on the island of Montréal (Québec), Forsyth & Watson (1985) estimated that defoliation over 50% was required to reduce weed vigor, but that C. rubiginosa rarely achieved this. Cartwright (1984) estimated that an infestation by C. rubiginosa was equivalent to a 50- 75% mechanical defoliation.
In Maryland, Tipping (1992) reported 72% seed reduction of musk thistle but these exceptional results were obtained with field-caged individuals protected from parasites and predators. In Virginia, Cartwright & Kok (1990) found that the beetle could significantly affect the growth of musk thistle when more than half of the foliage was eaten. Cartwright & Kok (1990) also studied the effects of defoliation by C. rubiginosa on the growth of musk thistles in Virginia. They found no reduction in seed yield despite an average 23.6% defoliation, but observed that growth was more adversely affected by multiple than single defoliations, particularly when 50% or more of the foliage was removed. Thus, although it is unlikely that C. rubiginosa can control Carduus thistles by itself, it can add substantially to the total stress on the weeds if used in conjunction with other biological control agents. The recent attempts to establish the beetle in South Dakota did not succeed ( Liu et al. 2000; Reed et al. 2006).
Ang et al. (1994, 1995) looked at the competitive growth of Canada thistle, tall fescue ( Festuca arundinacea Schreb. ), and crown vetch ( Coronilla varia L.) in the presence of C. rubiginosa . Their results indicated that C. rubiginosa enhanced the impact of the plant competitors by inhibiting the ability of thistle to compete against other plants.
In another approach, Tipping (1993) in Maryland studied the interaction between Canada thistle, C. rubiginosa , and the rust Puccinia punctiformis (Strauss.) Röhl. (Uredinales: Pucciniaceae ). He found that infected plants suffered or did not bloom but were not more attractive than healthy ones to the beetle. The following year, Kok & Abad (1994) demonstrated that C. rubiginosa adults could disperse spores of the rust Puccinia carduorum Jacky , an autoecius rust fungus imported from Turkey for the control of musk thistle ( Baudoin et al. 1993). The spores were attached to the tarsal setae of the adults and consequently the beetles increased the effective control of this weed. According to Kok et al. (1996) and Kok (1998, 2001), the longevity, egg production, and larval development of C. rubiginosa were not adversely affected by the rust fungus. However, Kluth et al. (2001, 2002) in Germany demonstrated that this interaction between C. rubiginosa and the rust fungus P. punctiformis was antagonistic. Although C. rubiginosa transferred spores, the biomass of the adult beetles was significantly reduced, the larval and pupal development tended to be prolonged, and the mortality increased when feeding on infected plants. These results were called into question by the laboratory investigations of Kok et al. (1996) who found that adults and larvae of C. rubiginosa preferred healthy leaves or pustule-free areas of infected leaves, and consequently the rust did not significantly interfere with the development and reproduction of the musk thistle herbivores.
In Germany, Kruess (2002) examined the indirect interactions between the necrotrophic fungus Phoma destructiva (Plowr.) ( Pleosporales : Leptosphaeriaceae ), Canada thistle, and C. rubiginosa . Adults avoided infected thistles for egg deposition and feeding, the larval development was negatively affected, and larval and pupal mortality was higher.
In Switzerland, Bacher & Schwab (2000) concluded that high levels of plant competition combined with herbivory of C. rubiginosa larvae led to 50% mortality of Canada thistle during the growing season. Consequently, they believed that an increase of herbivores in combination with breaking the root system by tillage and the establishment of competitive plants may be a feasible way to control the weed.
Locality records. A total of 57 specimens were examined:
NEW BRUNSWICK: Albert Co.: 7.VII.2004, R.P. Webster, Cirsium arvense , (1, RWC); Westmoreland Co.: 2.VII.1939 and 4.VII.1939, Shediac, W.J. Brown, on Arctium minus , (9, CNC). NOVA SCOTIA: Annapolis Co.: Bridgetown, 7.IX.1958, C.R. MacLellan, (1, ACNS); Granville Ferry, 30.VI.2002, C.G. Majka, coast field on thistle, (1, CGMC); Hampton, 7.VIII.2005, Cirsium arvense , (2, CGMC); Cape Breton Co.: Glace Bay, 3.VIII.1994, D.B. McCorquodale, (1, CBU); Schooner Pond, 6.IX.2003 and 11.IX.2003, C.W. D'Orsay, (2, CBU); Sydney Tar Ponds, 7.VI.1996, L.A. Hudson, (1, CBU); Colchester Co.: Debert, 12.IX.1999, J. Ogden, (1, NSNR); Shubenacadie, 2.VI.2003, J. Ogden, (1, NSNR); Truro, 6.VII.1982, M.A. Bulger and L.H. Lutz, (2, NSAC); Cumberland Co.: Amherst, no date indicated, J. Ogden, (1, JOC); Halifax Co.: Halifax, 3.VII.1967, K.A. Neil, (1, NSMC); Point Pleasant Park, 30.VI.2001 and 15.VIII.2001, C.G. Majka, Cirsium arvense , (2, CGMC); south-end Halifax, 19.VI.2001 and 26.VI.2001, Cirsium arvense , (2, CGMC); Hants Co.: Enfield, 22.VIII.2005, C.G. Majka, Cirsium arvense , (1, CGMC); Maitland, 25.VI.2002, A.J. Hebda, dikeland, (1, NSMC); Noel Shore, 2.VII.2002 and 9.VII.2002, A.J. Hebda, garden, (3, NSMC); Inverness Co.: Scotsville, 10.VII.1975, no collector indicated, (1, NSMC); Kings Co.: no location indicated, 27.VI.1956, H.B. Specht, (11, NSMC); Canard Creek, 12.VI.1987 and 4.VII.1988, J.A. Adams, (2, NSAC); Kentville, 16.VII.1950 and 6.VIII.1952, V.R. Vickery, (3, NSAC); Kentville, 31.V.2005, C. Sheffield and S. Westby, (1, ACNS); Porter Pt., 15.VII.1948, K.D. Archibald, (1, NSMC); Lunenburg Co.: Bridgewater, 5.IX.2002, G.D. Selig, (1, GSC). PRINCE EDWARD ISLAND: no locality indicated, 1974-83, UPEI; Prince Co.: Freetown, 31.VII.1991, M.E.M. Smith, potato field, (1, ACPE); Queens Co.: Cavendish, 14.VII.2002, C.G. Majka, coastal lagoon, (1, CGMC); Charlottetown, 12.VI.1957, F.M. Cannon, (1, ACPE); Charlottetown, 18.VI.1982, L.S. Thompson, (1, ACPE).
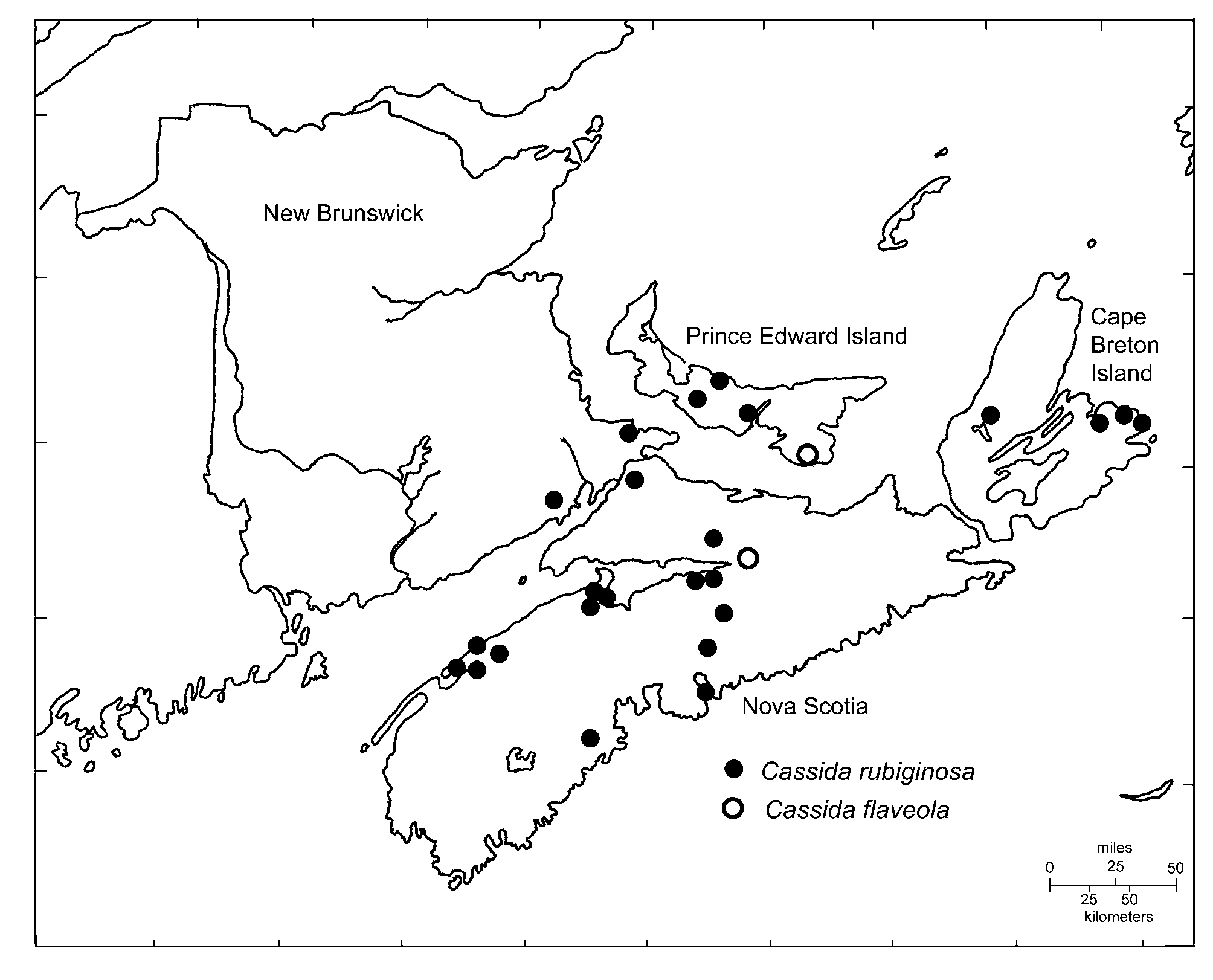
Distribution in the Maritime Provinces. The distribution of C. rubiginosa in the Maritime Provinces is indicated in Fig. 3 View FIGURE 3 . It is newly recorded in both Nova Scotia (commencing in 1948) and Prince Edward Island (commencing in 1957). Although the distribution of the species within Nova Scotia and Prince Edward Island is somewhat uneven, it appears broadly distributed in appropriate open habitats, given the uneven past collecting effort in the region. Collecting of Coleoptera in New Brunswick has been particularly spare and uneven, so further fieldwork there is required to determine if it occurs beyond the southeastern portion of the province where it has been recorded. In some portions of Nova Scotia, C. rubiginosa co-occurs on thistles with the adventive leaf beetle, Sphaeroderma testaceum (F.) ( Majka & LeSage 2006).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Cassida rubiginosa Müller, 1776
| Majka, Christopher G. & Lesage, Laurent 2008 |
review
| Peschken 1971 |
review (
| McLeod 1962 |