Erythromelana, , Townsend, 1919
|
publication ID |
https://doi.org/10.11646/zootaxa.3621.1.1 |
|
publication LSID |
lsid:zoobank.org:pub:99CC048E-C555-4178-8932-70E93A3B255C |
|
persistent identifier |
https://treatment.plazi.org/id/03BF7F25-FFAD-AB57-FF03-FDB03D2AFA17 |
|
treatment provided by |
Felipe |
|
scientific name |
Erythromelana |
| status |
|
ERYTHROMELANA View in CoL View at ENA HOST ASSOCIATIONS
After seven years of inventorying caterpillars, the Caterpillars and Parasitoids of the Eastern Andes in Ecuador project has reared over 1000 tachinids from 16 families of Lepidoptera , and preliminary identifications of these specimens suggest the existence of over 230 tachinid species ( Miller & Dyer 2009, Stireman et al. 2009, Stireman unpub. data). Of these records, 28 Erythromelana specimens were reared from Eois spp. ( Lepidoptera : Geometridae ), and one specimen from an unknown pyralid larva ( Lepidoptera : Pyralidae ). Given the nearly perfect association of Erythromelana with Eois hosts and that most of the caterpillars sampled at the site and day of collection of the pyralid-reared specimen were geometrids, this latter record is suspected to be erroneous. The 28 Erythromelana specimens were reared from 24 caterpillars collected from host plants in the genus Piper , two from Peperomia , one on Sarcorachis ( Piperaceae ), and a single specimen from the genus Siparuna (Monimiaceae) ( Table 3).
One problem in understanding insect diversity and host relationships of herbivores and parasitoids is the presence of large numbers of rare species ( Novotny & Basset 2000). In the Ecuadorian lepidopteran and parasitoid inventory project mentioned above, with a total parasitism by tachinids of about 9%, more than 50% of 150 morphospecies were represented by a single individual ( Stireman et al. 2009). In particular, Erythromelana appear to be extremely rare. From 5810 successful Eois spp. rearing events with a total percent of parasitism of 8.2% (including Braconidae and Ichneumonidae ), the percent of tachinid parasitism (e.g., by Eribella Mesnil , Calolydella Townsend , Siphona Meigen , Eucelatoria Townsend , and Phytomyptera Rondani ) is less than 2%, and the percent parasitism by Erythromelana is less than 0.5%. Additionally, two years of monthly samples from Malaise and pan traps from the area where these caterpillars were collected recovered only a single Erythromelana specimen from a collection of over 2000 individual tachinids. Although the rarity of these species makes it difficult to analyze host associations, a brief examination is provided below.
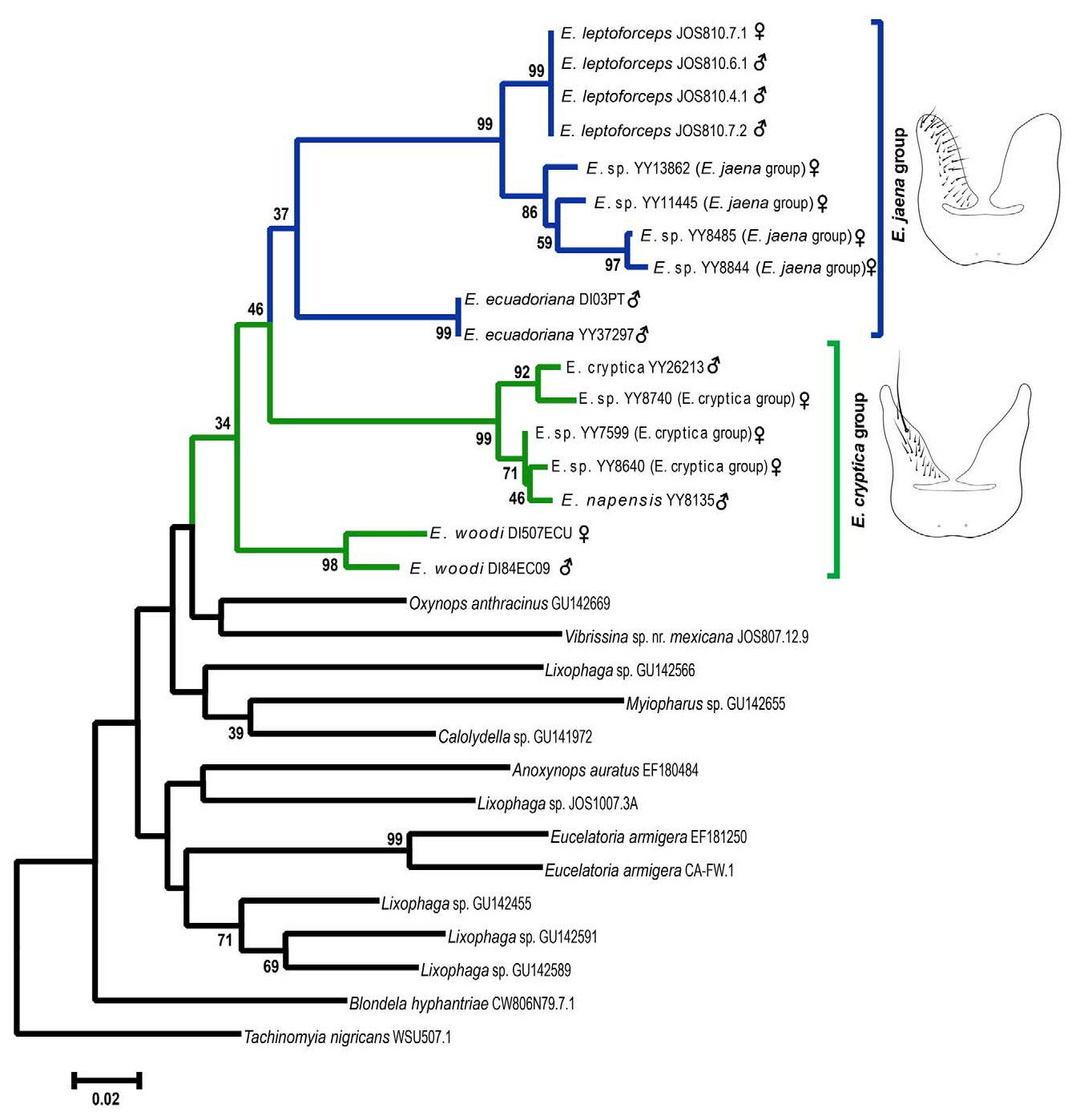
Host specialization appears to be a key factor in species diversification ( Schluter 2000). Specifically, specialization can increase genetic differentiation of populations by linking resource use and mate choice (e.g., Bush 1975, Feder et al. 1988, Hawthorne & Via 2001). At least five Erythromelana species appear to be specialized on the geometrid genus Eois ( Table 3). Of these records, E. curvifrons is the only species where the host genus is unknown, although it is likely Eois as it was identified as a geometrid and was collected on a host plant on which Eois feeds. The specificity of Erythromelana species on particular Eois species is difficult to evaluate because most of the caterpillar hosts were identified only to genus. Still, several Erythromelana species appear to parasitize the same host species and it is likely that some parasitize several Eois species. For example, E. napensis and the two morphospecies, E. sp. ( E. cryptica group) and E. jaena -group specimens with small palpus, all appear to parasitize the same host in the Eois encina group; and E. sp. ( E. cryptica group) parasitizes Eois on at least five host-plants, which likely represents multiple Eois species ( Table 3). This suggests that Erythromelana are largely specialized on the genus Eois , but individual species may use more than one Eois host. From these records it appears unlikely that host-associated ecological speciation by itself can explain the diversification of Erythromelana . However, a recent analysis of Eois diversification has revealed widespread cryptic speciation in this group, as well as the use of multiple hosts by some lineages ( Wilson et al. 2012), suggesting that host-related diversification of Erythromelana remains a possibility.
GEOGRAPHIC PATTERNS IN ERYTHROMELANA RICHNESS
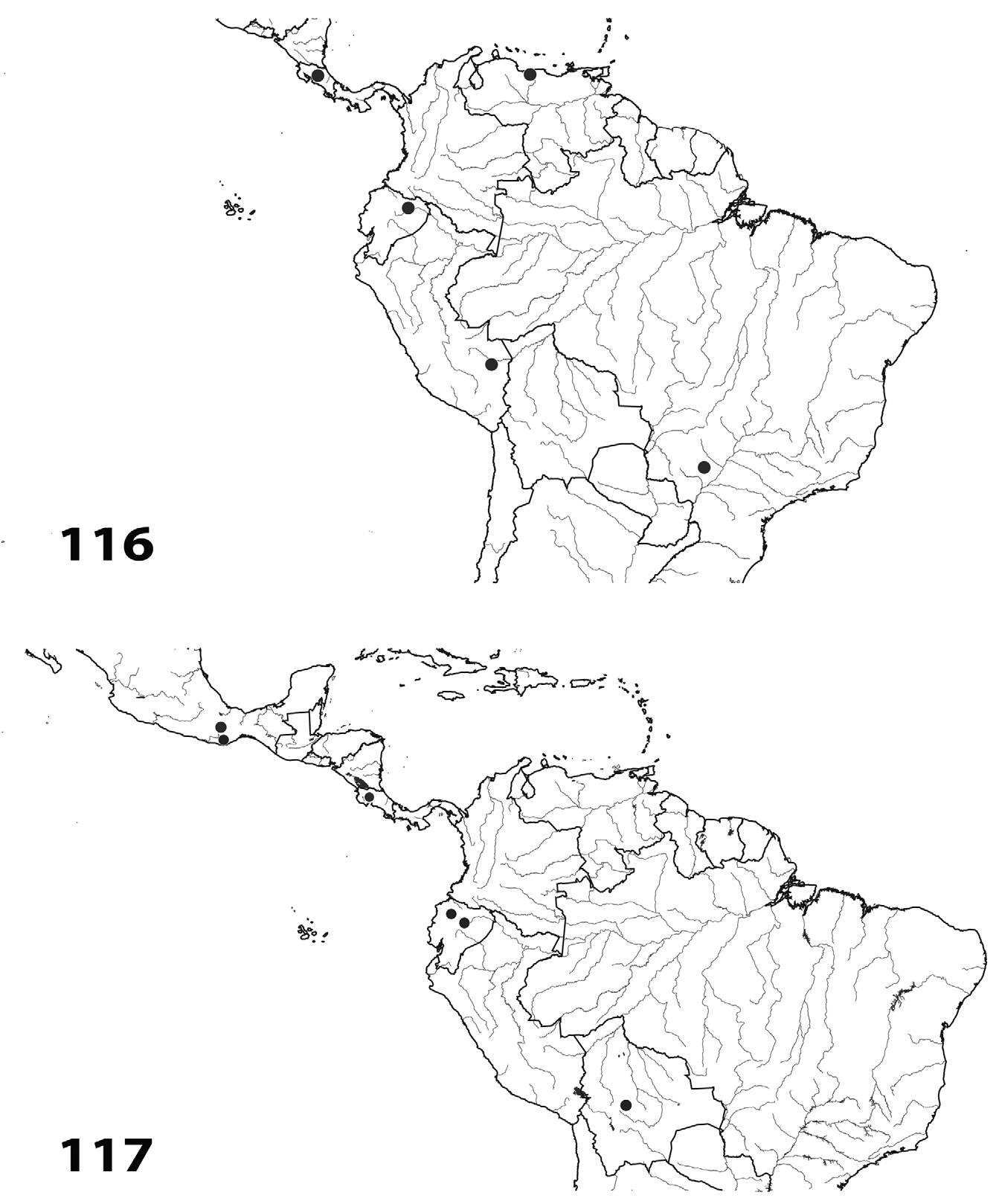
Species in the genus Erythromelana are broadly distributed across the Neotropical Region ( Figs. 112–117 View FIGURES 112–113 View FIGURES 114–115 View FIGURES 116–117 ). In general, the genus is poorly sampled, as the fauna between Mexico and Costa Rica and between southern Brazil and the Andes Mountains remains unknown. Erythromelana species occur in a variety of habitats ranging from lowland to montane tropical forest across a range of elevations (e.g., Santa Catarina, Brazil, 300–500 m to Napo, Ecuador 2000–2600 m).
Given the parasitoid lifestyle of Erythromelana species , their distribution must correspond to the distribution and diversity of their lepidopteran hosts and their host plants. The primary caterpillar host of Erythromelana , Eois , is widely distributed in the Neotropics, from Mexico to Argentina, where it comprises an important part of the geometrid fauna ( Strutzenberger et al. 2009). Eois includes 239 Neotropical species, but estimates of the diversity of this genus suggest the existence of perhaps 2000 species in this region ( Strutzenberger et al. 2009, Rodriguez- Castañeda et al. 2010, Brehm et al. 2011). The bulk of Eois species diversity appears to occur in Neotropical montane regions ( 1600–1800 m), where more than 500 species are estimated to occur ( Rodriguez-Castañeda et al. 2010, Brehm et al. 2011). Plants in the genus Piper , the predominant hosts of Eois caterpillars, exhibit similar distributions and diversity patterns. Piper is widely distributed in the Neotropics, including about 700 described species ( Jaramillo & Manos 2001). Similar to Eois , pipers are diverse at mid elevations ( 1600–1800 m), particularly in the Andes where there are at least 300 described species ( Jaramillo & Manos 2001, Rodriguez- Castañeda et al. 2010). In summary, Erythromelana , Eois , and Piper are similarly distributed across the Neotropical Region, and the Andes Mountains appear to be a hotspot of diversity for each of these genera.
Altitude appears to be a major factor in determining the distribution and diversification of Erythromelana species. Erythromelana species can be divided into species occurring only at low elevations (< 1000 m), mid elevations ( 1000–1800 m), and high elevations (> 1800 m). For example, E. catarina appears to be restricted to low elevations, whereas E. convexiforceps , E. napensis , E. jaena , E. curvifrons , E. ecuadoriana , and E. eois appear to be restricted to high elevations. The species E. arciforceps , E. distincta , E. leptoforceps , and E. nigrithorax occur at both low and mid elevations, and E. abdominalis and E. woodi occur at mid and high elevations. Erythromelana cryptica is the only species that has been collected across a wide range of elevations, from relatively low elevations in Costa Rica ( 700 m) to high elevations in Ecuador ( 2000–2600 m). Interestingly, it is this species, along with E. woodi , that is most likely to contain additional cryptic species. Overall, Erythromelana exhibits a lower species diversity in low elevations and higher diversity in mid and high elevations. This distribution of diversity across different altitudes correlates well with the distribution of their caterpillar hosts. The number of low elevation Eois species is estimated to be 260 species, whereas 570 species are estimated to inhabit higher elevations (Rodriguez- Castañeda et al. 2010, also see Brehm et al. 2011).
Most Erythromelana species are found in South America with a few species restricted to Central America. For example, E. convexiforceps View in CoL and E. nigrithorax have been collected only from southern Mexico and El Salvador. There are only two species, E. woodi View in CoL and E. cryptica View in CoL , that are widely distributed from Mexico to the Andes in South America (but see the note about these two taxa above). All the other species occur in South America, with E. leptoforceps View in CoL , E. napensis View in CoL , E. arciforceps View in CoL , and E. distincta View in CoL also occurring in Costa Rica. From a phylogenetic perspective, this distribution suggests that Erythromelana View in CoL may have originated in the Andes Mountains and expanded to Central America and the Amazon lowlands. The distribution and phylogenetic position of E. woodi View in CoL (see Figs. 110 View FIGURE 110 , 111 View FIGURE 111 and 117 View FIGURES 116–117 ) suggests that it may represent the most basal lineage, perhaps dispersing from the Andes and radiating into the species found in Central America and Mexico. The radiation of Erythromelana View in CoL could be related to the rapid uplift of the Andes Mountains in South America and the Talamanca highlands in Central America. The current elevation of the Andes is the result of a rapid uplift that occurred during the last ten million years, whereas the Talamanca highlands rose within the last five million years ( Hooghiemstra & Van der Hammen 1998, Gregory-Wodzicki 2000, Grafe et al. 2002). The Andes Mountains, with their great topographic heterogeneity, represent one of the most biologically diverse regions on Earth ( Lomolino 2001, Molau 2004, Beck et al. 2008). Of the 14 Erythromelana species described in this revision, five ( E. jaena View in CoL , E. abdominalis View in CoL , E. curvifrons , E. ecuadoriana View in CoL , and E. eois View in CoL ) appear to be restricted to the Andes. This Andean fauna includes all species with yellow abdomens. Erythromelana woodi View in CoL is the only species that contains individuals with abdomens ranging from mostly yellow to mostly black, but specimens with mostly yellow abdomens are from the Andes and specimens with black abdomens occur in Central America. The varied topography and habitat heterogeneity of the Andes could promote isolation of populations and increase the likelihood of differentiation among them. For example, single female and male E. woodi View in CoL specimens were collected at approximately the same latitude in Ecuador, but the female is from Mindo on the western slope and the male from is from YBS on the eastern slope of the Andes. These specimens appear morphologically to be a single species, but the molecular analysis suggests that they may represent distinct species (see phylogeny section and Fig. 111 View FIGURE 111 ). The lack of morphological differences between these specimens may reflect recent geography-associated divergence of this species, in which eastern and western populations are isolated by the high Andean paramo.
Further systematic, biogeographic and ecological studies on this group will undoubtedly allow more rigorous evaluation of the evolutionary origins, patterns of diversification, and host-relationships of this fascinating genus.
ACKNOWLEDGMENTS
We thank all those who, in various ways, made this work possible. Jim O’Hara, Monty Wood (Invertebrate Biodiversity, Agriculture and Agri-Food Canada, CNC), Norm Woodley (Systematic Entomology Laboratory, NMNH), Nigel Wyatt (Natural History Museum, Department of Entomology, BMNH), Manuel Zumbado (National Biodiversity Institute of Costa Rica, INBio) and Pierfilippo Cerretti (U. of Rome La Sapienza) provided specimen loans and expertise and training in tachinid identification. We also thank Tom Rooney (Wright State U.) and Greg Dahlem (Northern Kentucky U.) for their guidance, advice, and training while serving on D.J.I.’s graduate committee. Special thanks to Jeremy Heath and Hilary Devlin for their support and advice provided to D.J.I. Thanks to Jim O’Hara and two anonymous reviewers for carefully reviewing and editing this manuscript. This study was made possible through support by National Science Foundation grant DEB 0717092 (to J.O.S.), a Biology Award for Research Excellence from the Department of Biological Sciences at Wright State University, and a Dipterology Fund award from the North American Dipterists Society (to D.J.I.).
REFERENCES
Amorim, D. de S., Silva, V.C. & Balbi, M.I.P.A. ( 2002) Estado do conhecimento dos Diptera Neotropicais. In: Costa, C., Vanin, S.A., Lobo, J.M. & Melic, A. (eds.), PrIBES 2002 [Proyecto de Red Iberoamericana de Biogeografía y Entomología Sistemática]. Sociedad Entomológica Aragonesa, Zaragoza. Vol. 2, pp. 29 – 36.
Beck, E., Makeschin, F., Haubrich, F., Richter, M., Bendix, J. & Valarezo, C. ( 2008) The ecosystem (Reserva Biológica San Francisco). In: Beck, E., Bendix, J., Kottke, I., Makeschin, F. & Mosandl, R. (eds.), Gradients in a tropical mountain ecosystem of Ecuador. Ecological Studies, no.198. Springer, Berlin Heidelberg New York, pp. 1 – 14.
http://dx.doi.org/10.1007/978-3-540-73526-7_1
Benson, D.A., Karsch-Mizrachi, I., Lipman, D.J., Ostell, J., Rapp, B.A. & Wheeler, D.L. ( 2000) GenBank. Nucleic Acids Research, 28, 15 – 18.
http://dx.doi.org/10.1093/nar/28.1.15
Belshaw, R. ( 1993) Tachinid flies. Diptera: Tachinidae. Handbooks for the identification of British Insects, 10, Part 4a(i). Royal Entomological Society of London, London. 169 pp.
Brehm, G., Bodner, F., Strutzenberger, P., Hünefled, F. & Fiedler, K. ( 2011) Neotropical Eois (Lepidoptera: Geometridae): checklist, biogeography, diversity, and description patterns. Annals of the Entomological Society of America, 104, 1091 – 1107.
http://dx.doi.org/10.1603/AN10050
Brown, B.V. ( 2001) Flies, gnats, and mosquitoes. In: Levin S.A. (ed.), Encyclopedia of Biodiversity, Vol. 2. Academic Press, London, pp. 815 – 826.
http://dx.doi.org/10.1016/B0-12-226865-2/00123-1
Brown, B.V. ( 2005) Malaise trap catches and the crisis in Neotropical dipterology. American Entomologist, 51, 180 – 183.
Burns, J.M., Janzen, D.H., Hajibabaei, M., Hallwachs, W. & Hebert, P.D. ( 2008) DNA barcodes and cryptic species of skipper butterflies in the genus Perichares in Area de Conservacion Guanacaste, Costa Rica. Proceedings of the National Academy of Sciences, 105, 6350 – 6355.
http://dx.doi.org/10.1073/pnas.0712181105
Bush, G.L. ( 1975) Sympatric speciation in phytophagous parasitic insects. In: Price, P.W. (ed.), Evolutionary strategies of parasitic insects and mites. Plenum Press, New York, pp. 187 – 206.
http://dx.doi.org/10.1007/978-1-4615-8732-3_9
Connahs, H., Rodriguez-Castañeda, G., Walters, T., Walla, T. & Dyer, L.A. ( 2009) Geographical variation in host specificity and parasitism rates of an herbivore (Geometridae) associated with the tropical genus Piper. Journal of Insect Science, 9 (Article 28), 11 pp.
http://dx.doi.org/10.1673/031.009.2801
Cumming, J.M. & Wood, D.M. ( 2009) Adult morphology and terminology. In: Brown, B.V., Borkent, A., Cumming, J.M., Wood, D.M., Woodley, N.E. & Zumbado, M. (eds.), Manual of Central American Diptera, Vol. 1. NRC Research Press, Ottawa, pp. 9 – 50.
Dyer, L.A., Miller, J.S., Rab Green, S.B., Gentry, G.L., Greeney, H.F. & Walla, T.W. ( 2012). Caterpillars and parasitoids of the Eastern Andes in Ecuador. Available from: http://caterpillars.unr.edu/lsacat/ecuador/index.htm (accessed 3 June 2012).
Feder, J.L., Chilcote, C.A. & Bush, G.L. ( 1988) Genetic differentiation between sympatric host races of the apple maggot fly Rhagoletis pomonella. Nature, 336, 61 – 64.
http://dx.doi.org/10.1038/336061a0
Felsenstein, J. ( 2005) PHYLIP [ Phylogeny Inference Package] version 3.69. Distributed by the author. Department of Genetics, University of Washington, Seattle.
Grafe, K., Frisch, W., Villa, I.M. & Meschede, M. ( 2002) Geo-dynamic evolution of southern Costa Rica related to low-angle subduction of the Cocos Ridge: constraints from thermochronology. Tectonophysics, 348, 187 – 204.
http://dx.doi.org/10.1016/S0040-1951(02)00113-0
Gregory-Wodzicki, K.M. ( 2000) Uplift history of the Central and Northern Andes: a review. Geological Society of America Bulletin, 112, 1091 – 1105.
http://dx.doi.org/10.1130/0016-7606(2000)112%3C1091:UHOTCA%3 E2.0.CO;2
Guimarães, J.H. ( 1971) Family Tachinidae (Larvaevoridae). A catalogue of the Diptera of the Americas south of the United States, Vol. 104. São Paulo, 333 pp.
Hawthorne, D.J. & Via, S. ( 2001) Genetic linkage of ecological specialization and reproductive isolation in pea aphids. Nature, 412, 904 – 907. http://dx.doi.org/10.1038/35091062
Hebert, P.D., Cywinska, A., Ball, S.L. & deWaard, J.R. ( 2003) Biological identifications through DNA barcodes. Proceedings of the Royal Society of London, 270, 313 – 321.
http://dx.doi.org/10.1098/rspb.2002.2218
Hooghiemstra, H. & Van der Hammen, T. ( 1998) Neogene and Quaternary development of the Neotropical rain forest: the forest refugia hypothesis, and a literature overview. Earth -Science Reviews, 44, 147–183.
http://dx.doi.org/10.1016/S0012-8252(98)00027-0
Irwin, M.E., Schlinger, E.I. & Thompson, F.C. ( 2003) Diptera, true flies. In: Goodman, S.M. & Benstead, J.P. (eds.), The natural history of Madagascar. University of Chicago Press, Chicago, pp. 692 – 702.
Jaramillo, M.A. & Manos, P.S. ( 2001) Phylogeny and the patterns of floral diversity in the genus Piper (Piperaceae). American Journal of Botany, 88, 706 – 716.
http://dx.doi.org/10.2307/2657072
Kjaerandsen, J. ( 2009) The genus Pseudexechia Tuomikoski re-characterized, with a review of European species (Diptera: Mycetophilidae). Zootaxa, 2056, 1 – 45.
Lomolino, M.V. ( 2001) Elevation gradients of species-density: historical and prospective views. Global Ecology and Biogeography, 10, 3 – 13.
http://dx.doi.org/10.1046/j.1466-822x.2001.00229.x
Meier, R., Shiyang, K., Vaidya, G. & Ng, P.K.L. ( 2006) DNA barcoding and taxonomy in Diptera: A tale of high intraspecific variability and low identification success. Systematic Biology, 55, 715 – 728.
http://dx.doi.org/10.1080/10635150600969864
Miller, J.S. & Dyer, L.A. ( 2009) Special feature: diversity of insect-plant interactions in the eastern Andes of Ecuador. Journal of Insect Science, 9 (Article 26), 3 pp.
http://dx.doi.org/10.1673/031.009.2601
Molau, U. ( 2004) Mountain biodiversity patterns at low and high latitudes. Ambio Special Report, 13, 24 – 28.
Molbo, D., Machado, C.A., Sevenster, J.G., Keller, L. & Herre, E.A. ( 2003) Cryptic species of fig-pollinating wasps: implications for the evolution of the fig-wasp mutualism, sex allocation, and precision of adaptation. Proceedings of the National Academy of Sciences, 100, 5867 – 5872.
http://dx.doi.org/10.1073/pnas.0930903100
Monaghan, M.T., Balke, M., Gregory, T.R. & Vogler, A.P. ( 2005) DNA-based species delineation in tropical beetles using mitochondrial and nuclear markers. Philosophical Transactions of the Royal Society, 360, 1925 – 1933.
http://dx.doi.org/10.1098/rstb.2005.1724
Novotny, V. & Basset, Y. ( 2000) Rare species in communities of tropical insect herbivores: pondering the mystery of singletons. Oikos, 89, 564 – 572.
http://dx.doi.org/10.1034/j.1600-0706.2000.890316.x
O’Hara, J.E. ( 1989) Systematics of the genus group taxa of the Siphonini (Diptera: Tachinidae). Quaestiones Entomologicae, 25, 1 – 229.
O’Hara, J.E. ( 2002) Revision of the Polideini (Tachinidae) of America north of Mexico. Studia dipterologica, 10, 1 – 170.
O’Hara, J.E. ( 2011) World genera of the Tachinidae (Diptera) and their regional occurrence. Version 6.0. PDF document, 75 pp. Available from: http://www.nadsdiptera.org/Tach/Genera/Gentach_ver6.pdf (accessed 3 June 2012).
O’Hara, J.E. & Wood, D.M. ( 2004) Catalogue of the Tachinidae (Diptera) of America north of Mexico. Memoirs on Entomology, International, 18, 410 pp.
Oksanen, J., Kindt, R., Legendre, P., O’Hara, B. & Stevens M. ( 2007) vegan: Community Ecology Package. R package version 1.15 – 4. Available from: http://r – forge.r – project.org/projects/vegan (accessed 3 June 2010).
R Development Core Team ( 2010) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. Available from: http://www.R–project.org (accessed 18 May 2010).
Rodriguez-Castañeda, G., Dyer, L.A., Brehm, G., Connahs, H., Forkner, R.E. & Walla, T.R. ( 2010) Tropical forests are not flat: how mountains affect herbivore diversity. Ecology Letters, 13, 1348 – 1357.
http://dx.doi.org/10.1111/j.1461-0248.2010.01525.x
Schluter, D. ( 2000) The Ecology of Adaptive Radiation. Oxford University Press, Oxford, 288 pp.
Shorthouse, D.P. ( 2010) SimpleMappr, an online tool to produce publication-quality point maps. Available from: http:// www.simplemappr.net (accessed 9 May 2010).
Smith, M.A., Woodley, N.E., Janzen, D.H., Hallwachs, W. & Hebert, P.D. ( 2006) DNA barcodes reveal cryptic host-specificity within the presumed polyphagous members of a genus of parasitoid flies (Diptera: Tachinidae). Proceedings of the National Academy of Sciences, 103, 3657 – 3662.
http://dx.doi.org/10.1073/pnas.0511318103
Smith, M. A, Wood, D.M., Janzen, D.H., Hallwachs, W. & Hebert, P.D. ( 2007) DNA barcodes affirm that 16 species of apparently generalist tropical parasitoid flies (Diptera, Tachinidae) are not all generalists. Proceedings of the National Academy of Sciences, 104, 4967 – 4972.
http://dx.doi.org/10.1073/pnas.0700050104
Stireman, J.O., III ( 2002) Phylogenetic relationships of tachinid flies in subfamily Exoristinae (Tachinidae: Diptera) based on a 28S rDNA and elongation factor-1-alpha. Systematic Entomology, 27, 409 – 435.
http://dx.doi.org/10.1046/j.1365-3113.2002.00187.x
Stireman, J.O., III, O’Hara, J.E. & Wood, D.M. ( 2006) Tachinidae: evolution, behavior, and ecology. Annual Review of Entomology, 51, 525 – 555.
http://dx.doi.org/10.1146/annurev.ento.51.110104.151133
Stireman, J.O., III, Greeney, H.F., & Dyer, L.A. ( 2009) Species richness and host associations of Lepidoptera-attacking Tachinidae in the northeast Ecuadorian Andes. Journal of Insect Science, 9 (Article 39), 19 pp.
http://dx.doi.org/10.1673/031.009.3901
Strutzenberger, P., Brehm, G., Bodner, F. & Fiedler, K. ( 2009) Molecular phylogeny of Eois (Lepidoptera, Geometridae): evolution of wing patterns and host plant use in a species-rich group of Neotropical moths. Zoologica Scripta, 39, 603 – 620.
http://dx.doi.org/10.1111/j.1463-6409.2010.00440.x
Tachi, T. & Shima, H. ( 2010) Molecular phylogeny of the subfamily Exoristinae (Diptera, Tachinidae), with discussions on the evolutionary history of female oviposition strategy. Systematic Entomology, 35, 148 – 163.
http://dx.doi.org/10.1111/j.1365-3113.2009.00497.x
Tamura, K., Peterson, D., Peterson, N., Stecher, G., Nei, M. & Kumar, S. ( 2011) MEGA5.05: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Molecular Biology and Evolution, 28, 2731 – 2739.
http://dx.doi.org/10.1093/molbev/msr121
Townsend, C.H.T. ( 1919 a) New muscoid genera, species and synonymy (Diptera). Insecutor Inscitiae Menstruus, 6, 157 – 182.
Townsend, C.H.T. ( 1919 b) New genera and species of muscoid flies. The Proceedings of the United States National Museum, 56, 541 – 592.
http://dx.doi.org/10.5479/si.00963801.2301.541
Townsend, C. H. T. 1934 –1942. Manual of myiology in twelve parts. Charles Townsend & Filhos, Itaquaquecetuba, São Paulo, Brazil.
Whinnett, A., Zimmermann, M., Willmott, K.R., Herrera, N., Mallarino, R., Simpson, F., Joron, M., Lamas, G. & Mallet, J. ( 2005) Strikingly variable divergence times inferred across an Amazonian butterfly “suture zone ”. Proceedings of the Royal Society of London, 272, 2525 – 2533.
http://dx.doi.org/10.1098/rspb.2005.3247
Wilson, J.S., Forister, M.L., Dyer, L.A., O’Connor, J.M., Burks, K., Feldman, C.R., Jaramillo, M.A., Miller, J.S., Rodríguez- Castañeda, G., Tepe, E.J., Whitfield, J.B., & Young, B. ( 2012) Host conservatism, host shifts and diversification across three trophic levels in two Neotropical forests. Journal of Evolutionary Biology, 25, 532 – 546.
http://dx.doi.org/10.1111/j.1420-9101.2011.02446.x
Winterton, S.L., Wiegmann, B.M., & Schlinger, E.I. ( 2007) Phylogeny and Bayesian divergence time estimations of smallheaded flies (Diptera: Acroceridae) using multiple molecular markers. Molecular Phylogenetics and Evolution, 43, 808 – 832.
http://dx.doi.org/10.1016/j.ympev.2006.08.015
Wood, D.M. ( 1985) A taxonomic conspectus of the Blondeliini of North and Central America and the West Indies (Diptera: Tachinidae). Memoirs of the Entomological Society of Canada, 132, 1 – 130.
http://dx.doi.org/10.4039/entm117132fv
Wood, D.M. ( 1987) Chapter 110. Tachinidae. In: McAlpine, J.F., Peterson, B.V., Shewell, G.E., Teskey, H.J., Vockeroth, J.R. & Wood, D.M. (eds.), Manual of Nearctic Diptera, Vol. 2. Agriculture Canada Monograph, 28, 1193 – 1269.
Wood, D.M. & Zumbado, M. ( 2011) Tachinidae (tachinid flies, parasitic flies). In: Brown, B.V., Borkent, A., Cumming, J.M., Wood, D.M., Woodley, N.E. & Zumbado, M. (eds.), Manual of Central American Diptera, Vol. 2. NRC Research Press, Ottawa, pp. 1343 – 1417.
Wulp, F.M. van der ( 1890) Family Muscidae. In: Godman, E.D. & Salvin, O. (eds.), Biologia Centrali -Americana, Insecta, Diptera, Vol. 2. Taylor & Francis, London, pp. 41–208.
APPENDIX 1
Variables used in morphological ordinations and species descriptions. The numbers in brackets “[]” and parentheses “()” represent the sequential number of the variable and numbers assigned to a particular state of the character, respectively.
Continuous variables:
[1] Total body length – measured in profile from the pedicel to tip of the abdomen excluding abdominal setae. Head
[2] Ommatrichia length; measured from the back of the head using a white background to improve contrast of the setae. For each individual, the measurement was the average of two to three setae taken from the center region of the right eye.
[3] Head height, [4] eye height; [5] pedicel length, and [6] flagellum length were measured in profile as shown in Fig. 5 View FIGURES 5–9 .
[7] Maximum head length: measured in profile from the point of antennal insertion to the back of the head as shown in Fig. 6 View FIGURES 5–9 .
[8] Flagellum width: measured in profile at their maximum width, typically near the end as shown in Fig. 6 View FIGURES 5–9 .
[9] Parafacial width: measured from a viewpoint perpendicular to it at the minimum width of the parafacial.
[10] Facial ridge setae height: measured in profile from the vibrissal seta until the uppermost seta on the facial ridge as shown in Fig. 5 View FIGURES 5–9 .
[11] Palpus length: measured in profile as shown in Fig 5 View FIGURES 5–9 . For specimens in which measurement was not possible because the palpus was not visible, NA was reported.
[12] Head width: measured in frontal view as shown in Fig. 3 View FIGURES 3–4. 3 .
[13] Frontal vitta (FV) and [14] vertex width were measured in frontal view at their narrowest point as shown in Fig 3 View FIGURES 3–4. 3 .
Thorax
[15] Total thorax length: measured in dorsal view, at the center of the thorax from the anterior edge of the prescutum to the posterior edge of the scutum.
[16] Ultimate fore-tarsomere length: measured in profile at the center of the right fore-tarsomere.
[17] Fore-claw length: measured in profile from the right fore-claw.
[18] Wing length: measured in profile at the right wing from the end of the basicosta to the wing apex.
[19] Percentage of setae on wing vein R 4+5: calculated as the setose portion of R 4+5 as a proportion of the distance between base of R 4+5 and cross vein r-m on the right wing.
Male terminalia:
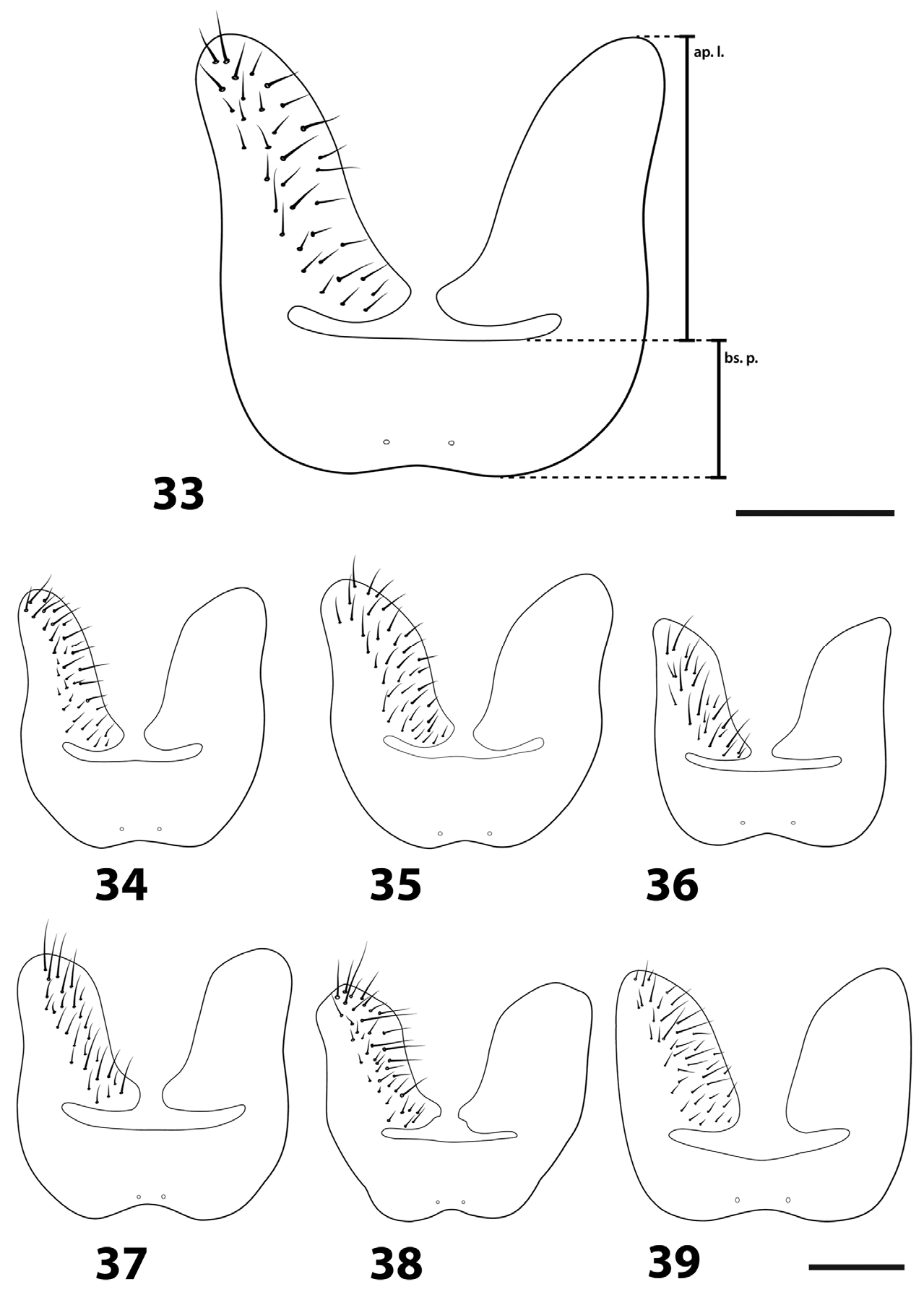
[20] St5 basal plate length, [21] apical lobe length, and [22] st5 total length were measured as shown in Fig. 35 View FIGURES 33–39 . [23] Cercus and [24] surstylus length were measured in profile.
The cerci were divided in three sections: [25] upper lobe length, [26] medial section length, and [27] apical cleft length, which were measured in posterior view as shown in Fig. 63 View FIGURES 61–67 .
Female terminalia:
[28] St5 width and [29] length: measured at the center of each sternite.
[30] Sternite 6 and [31] sternite 7 length: measured at the center of each sternite.
[32] Tergite 10 and [33] cercus length.
Discrete variables:
The following discrete characters were recorded:
Head:
[34] Fronto-orbital plate (FOP) color: (1) dull silver, (2) dull silver and black, and (3) black.
[35] FOP pruinescence: (0) absent and (1) golden pruinosity present.
[36] Vertex color: (1) dull silver, (2) dull silver and black, and (3) black.
[37] Vertex pruinescence: (0) absent and (1) golden pruinosity present.
[38] Palpus color: (1) brown, (2) yellow, (3) black, (4) brown with black at bases, and (5) brown-yellowish.
[39] Ommatrichia: (0) almost bare, (1) well developed, and (2) poorly developed.
[40] Ommatrichia density: (0) sparse, (1) dense.
[41] Fronto-orbital seta: Number of setae on right and left sides
[42] Inner-orbital seta: Number of setae on right and left sides
[43] Outer-orbital seta: Number of setae on right and left sides
[44] Subvibrissal seta: Number of setae
[45] Ocellar seta: (0) absent, (1) medioclinate, (2) lateroclinate, (3) proclinate, and (4) reclinate.
[46] Arista microtrichia: (1) pubescent, microtrichia length no more than the widest point of the arista, (2) plumose, microtrichia longer than width of arista.
Thorax:
[47] Postpronotum setae: Number of setae
[48] Postpronotum setae alignment: (1) forming a triangle, (2) in a line.
Setae on presutural scutum: [49] number of acrostichal, [50] dorsocentral, [51] intra-alar, and [52] supra-alar setae.
Setae on postsutural scutum: [53] number of acrostichal, [54] dorsocentral, [55] intra-alar, and [56] supra-alar setae.
[57] 1 st supra-alar seta on postsutural scutum: (0) absent, (1) smaller than 2 nd supra-alar setae.
[58] Katepisternum: Number of setae
Scutellar setae: (0) absent, (1) present for [59] basal, [60] discal, [61] lateral, [62] subapical, and [63] apical setae. [64] Tibia color: (1) yellow, (2) yellowish with black, (3) black.
Setae on mid tibia: Number of well-developed setae on [65] anterodorsal, [66] dorsal, [67] posterodorsal, [68] posterior, [69] posteroventral, and [70] ventral sections.
Setae on hind tibia: Number of well-developed setae on [71] posterodorsal and [72] anteroventral sections.
Wing cell color: (1) hyaline, (2) light fumose, and (3) dark fumose for: [73] c, [74] sc, [75] r 1, [76] r 2+3, [77] r 4+5, and [78] dm cells.
[79] R 4+5 dorsally with setae on (1) only at the base, (2) more that half way between the base and the cross with the vein r-m.
[80] Number of setae at the base of R 4+5 the right wing
[81] M1 vein ending at wing apex: (1) separately or (2) with R 4+5.
Abdomen:
[82] Abdomen color in dorsal view: (1) entirely yellow, (2) mostly yellow, (3) mostly black, (4) fully black, and (5) equally yellow and black.
[83] If abdomen mostly yellow, dorsally with black on: (1) tg1+2; (2) tg3; (3) tg4; (4) tg5; (5) tg1+2, tg3, and tg4; (6) tg3 and tg4; (7) tg1+2 to tg5; and (8) tg1+2, tg4, and tg5.
[84] If abdomen mostly black; dorsally with yellow on: (1) tg1+2; (2) tg3; (3) tg4; (4) tg5; (5) tg1+2, tg3, and tg4; (6) tg3 and tg4; (7) tg1+2 to tg5; and (8) tg1+2, tg4, and tg5.
[85] White pruinescence forming apical bands on: (0) absent, (1) 1/3 tg3, tg4, & ½ tg5; (2) 1/3 tg3, tg4 & 2/3 tg5; (3) 1/4 tg3, tg4 & 1/3 tg5; (4) 1/2 tg3, tg4, & 4/5 tg5; and (5) 1/4 tg3 & tg5.
[86] Discal setae on: (0) absent, (1) tg3 to tg5, (2) tg5, (3) tg3 & tg4, and (4) tg3.
[87] Median marginal setae on: (1) tg1+2 to tg5, and (2) tg3 to tg5.
[88] Sex patches: (0) absent, (1) on ventral tg4 and tg5.
Male terminalia:
[89] Setae on apical lobes of st5: (1) one long bristle on each lobe, (0) absent.
[90] Shape of apical lobes of st5: (1) pointed lobes, (2) rounded lobes.
[91] Surstyli internally with setae: (0) absent, (1) small, (2) medium, (3) large.
[92] Surstyli with a few small setae -like spines at anterior side of the tip: (1) present, (0) absent.
[93] Surstylus shape: (0) rectangular, (1) triangular shape
[94] Cerci with a dorsal depression or twist (1) present (0) dorsally flat.
[95] Cercus shape in profile: (1) slightly carinate, (2) moderately carinate, (3) strongly carinate, or (0) almost straight.
[96] Pregonite shape: (0) straight, (1) curved
Female terminalia:
[97] St5 setae: (1) with two pairs of well-developed setae on posterior edge, (2) several well developed setae covering at least distal ¼ of the sternite, and (3) only one pair of well developed setae in the center of the sternite. [98] Sternite 8: (0) absent, (1) present.
[99] Tergite 8: (1) dorsally with a distinctive narrow lobe, (0) dorsally without a lobe
Ratios:
The list of each ratio is present followed in brackets “[]” by the number of the continuous variable involved in the calculation of the ratio and in parentheses “()”by the number of the states and their inclusive ranges.
Head:
Head height to body length [3 to 1]: (1) X≤0.25 and (2) X>0.25
Head width to body length [12 to 1]: (1) X≤0.27, (2) 0.27<X≤0.3, and (3) X>0.3
Eye height to head height [4 to 3]: (1) X≤0.84, (2) 0.84<X≤0.88, and (3) X>0.88
Frontal vitta to vertex [13 to 14]: (1) X≤0.15, (2) 0.15<X≤0.32, and (3) X>0.32
Vertex to head width [14 to 12]: (1) X≤0.16 and (2) X>0.16
Parafacial width to head length [9 to 12]: (1) X≤0.03, (2) 0.03<X≤0.06, and (3) X>0.06
Parafacial width to flagellum width [9 to 8]: (1) X≤0.15, (2) 0.12<X≤0.30, and (3) X>0.30
Flagellum width to head length [8 to 7]: (1) X≤0.18, (2) 0.18<X≤0.24, and (3) X>0.24
Facial ridge length to head height [10 to 3]: (1) X≤0.14, (2) 0.14<X≤0.18, and (3) X>0.18
Palpus length to head height [11 to 3]: (1) X≤0.29, (2) 0.29<X≤0.33, and (3) X>0.33
Pedicel length to flagellum length [5 to 6]: (1) X≤0.26, (2) 0.26<X≤0.32, and (3) X>0.33
Flagellum length to head height [6 to 3]: (1) X≤0.45, (2) 0.45<X≤0.5, and (3) X>0.5
Thorax:
Thorax length to body length [15 to 1]: (1) X≤0.34, (2) 0.34<X≤0.38, and (3) X>0.38
Fore-claw length to last fore-tarsomere length [17 to 16]: (1) X≤1 and (2) X>1
Wing length to body length [18 to 1]: (1) X≤0.85, (2) 0.85<X≤0.95, and (3) X>0.95
Male terminalia:
St5 basal plate length to st5 apical lobe length [20 to 21]: (1) X≤0.8 and (2) X>0.8
St5 basal plate length to st5 length [20 to 22]: (1) X≤0.42 and (2) X>0.42
Cercus length to st5 length [23 to 22]: (1) X≤0.55, (2) 0.55<X≤0.75, and (3) X>0.75
Surstylus length to cercus length [24 to 23]: (1) X≤0.60, (2) 0.60<X≤0.66, and (3) X>0.66
Cercus upper lobe length to cercus length [25 to 23]: (1) X≤0.30, (2) 0.30<X≤0.40, and (3) X>0.40
Cercus medial section length to cercus length [26 to 23]: (1) X≤0.25, (2) 0.25<X≤0.45, and (3) X>0.45
Female terminalia:
St5 width to st5 length [28 to 29]
Sternite 6 length to st5 length [30 to 29]
Sternite 7 length to st5 length [31 to 29]
Tergite 10 length to st5 length [32 to 29]
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
Erythromelana
| Inclan, Diego J. & Stireman, John O. 2013 |
E. convexiforceps
| Inclan & Stireman 2013 |
E. leptoforceps
| Inclan & Stireman 2013 |
E. arciforceps
| Inclan & Stireman 2013 |
E. distincta
| Inclan & Stireman 2013 |
E. curvifrons
| Inclan & Stireman 2013 |
E. eois
| Inclan & Stireman 2013 |
E. leptoforceps
| Inclan & Stireman 2013 |
E. cryptica
| Inclan & Stireman 2013 |
E. catarina
| Inclan & Stireman 2013 |
E. convexiforceps
| Inclan & Stireman 2013 |
E. arciforceps
| Inclan & Stireman 2013 |
E. napensis
| Inclan & Stireman 2013 |
E. jaena
| Townsend. Scale 1919 |
E. jaena Townsend
| Townsend. Scale 1919 |