Parartemia acidiphila, Timms, Brian V & Hudson, Peter, 2009
|
publication ID |
https://doi.org/10.5281/zenodo.190741 |
|
DOI |
https://doi.org/10.5281/zenodo.6225427 |
|
persistent identifier |
https://treatment.plazi.org/id/03C38793-FFBD-8147-7BDA-3AECFE259A5B |
|
treatment provided by |
Plazi |
|
scientific name |
Parartemia acidiphila |
| status |
sp. nov. |
Parartemia acidiphila View in CoL sp. nov.
( Figures 1–3 View FIGURE 1 View FIGURE 2 View FIGURE 3 )
Synonymy. Parartemia sp f, Timms, 2004, p 24, Fig. 30; Timms, 2009c, pp 215–225.
Type material. Holotype. Male, SOUTH AUSTRALIA, Gawler Ranges, 32 km N of Yardea Homestead, “Little Lake” ( 32o 05’ 40”E, 135o 32’ 33’’E), 16 November 1990, P. Hudson, SAM C6787; Allotype. Female (ovigerous), same collecting data as holotype, SAM C6789; Paratypes. Two males, two females, same collecting data as holotype, SAM C6788; two males, two females, same collecting data as holotype, WAM 40315.
Other material. SOUTH AUSTRALIA, Eyre Peninsula, Lake Gilles, ( 33o02’07”S, 136o 36’00”E), 16 September 1990, P. Hudson, SAM C6792; SOUTH AUSTRALIA, Eyre Peninsula, approx 42 km W of Whyalla, small salt lake near Sinclair Gap, ( 33o 08’19”E, 137o 04’14”E), P. Hudson, SAM 6790; SOUTH AUSTRALIA, Eyre Peninsula, approx 42 km W of Whyalla, another small salt lake near Sinclair Gap, ( 33o07’30”S, 137o 03’38”E), 29 July 1990, P. Hudson, SAM 6791; 20 males, WESTERN AUSTRALIA, Esperance hinterland, lake on Guest Rd, ( 33o08’S, 121o49’E), 5 September 2004, B.V.Timms, WAM 40316; 10 males and 10 females, WESTERN AUSTRALIA, Esperance hinterland, lake on Ridley Rd East, ( 33o15’S, 121o59’E), 26 January 2007, B.V. Timms, WAM 40317; 5 males and 5 females, WESTERN AUSTRALIA, Esperance hinterland, Truslove Nature Reserve, ( 33o20’S, 121o46’E), 8 October 2008, B.V. Timms, WAM 40318.
Description. Male. Length 13 mm (head + thorax 7 mm and abdomen 6 mm).
First antenna filiform, about twice as long as eye plus peduncle ( Fig. 1 View FIGURE 1 A). Basal antennomeres of second antenna fused proximally at an angle of about 50 degrees from the body axis. Ventral margin with paired linear, ventral processes (VP, Fig. 1 View FIGURE 1 A) clothed with small spines, spaced 1–2 times their length. Overall dimensions of these processes about 4 times longer than deep with lateral edge a little shorter than medial edge. Distal margin slightly concave, lateral corner produced and medial corner rounded but only slightly enlarged. Posterior surface of fused proximal antennomeres with a small conical mound (CM, Fig 1 View FIGURE 1 B) slightly medial of the lateral corner of the frontal process. Medial process (MP, Fig 1 View FIGURE 1 A) between ventral processes broad and triangular with a bifurcated apex. Anterior surface of proximal antennomeres with a basal elevated area on each side and terminating in a free, conical anterior process (AP, Figs 1 View FIGURE 1 A,F) subequal in length to the medial process and about three times longer than basal width. Distal antennomere of second antenna curved medially, about 1.5 to 2 times the length of the proximal antennomere. Labrum lacking a spine.
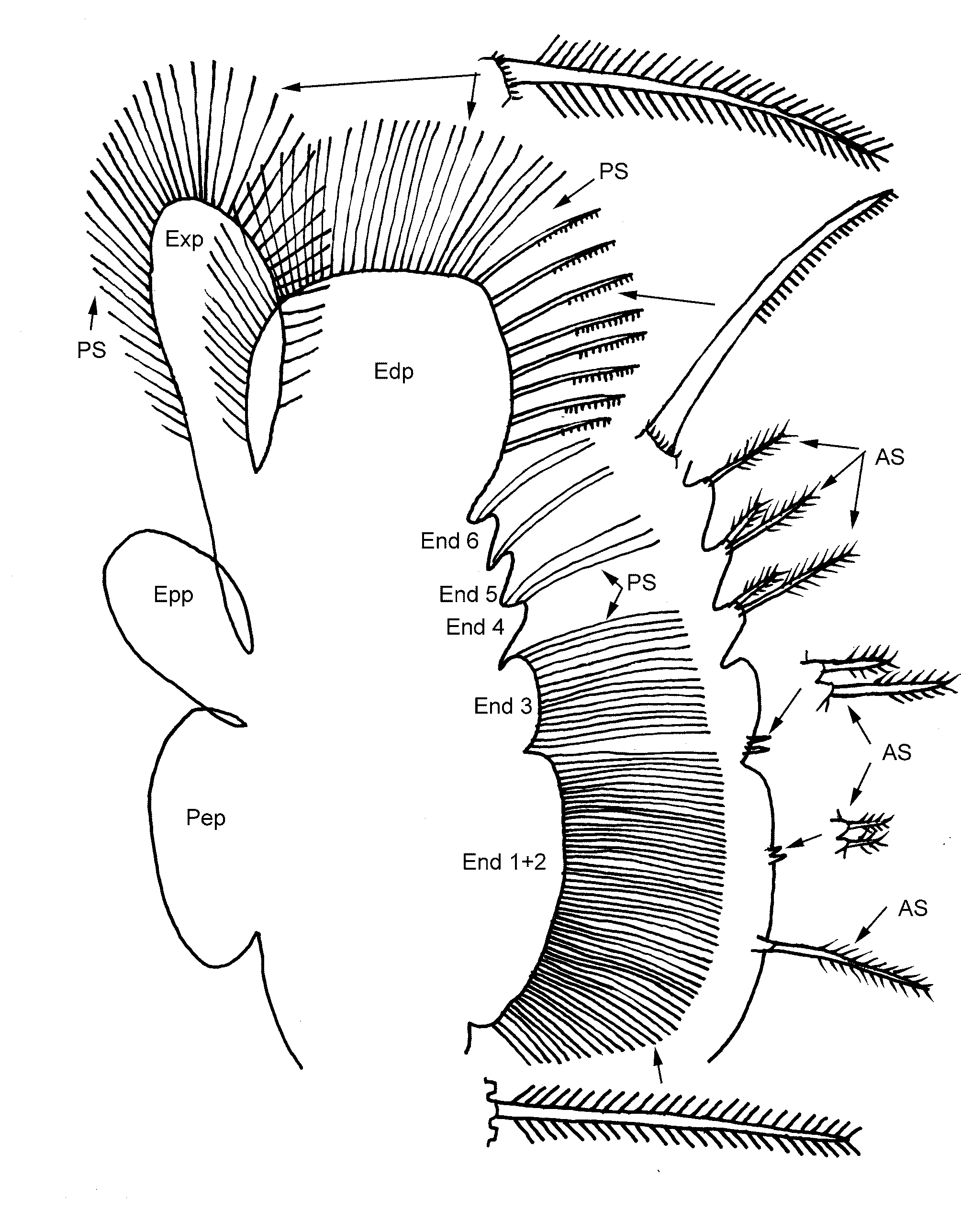
Thoracic segments with minor lateral lobes, all equal in size and proportions. Eleven pairs of thoracopods, variable in size with first two noticeably reduced in size but not in arrangement of its parts. Fifth thoracopod ( Fig. 2 View FIGURE 2 ) with endite 1+2 and 3 evenly curved, the former about three times the size of the latter. Endite 1 anterior seta about as long as adjacent posterior setae (shown enlarged in Fig. 2 View FIGURE 2 ) and with a double pectin of strong setules on apical half. Endite 2 anterior seta very short, about same length as base of anterior seta 1. Its spine even shorter and both weakly setose apically. Endite 3 anterior seta about four times length of anterior setae 2; both it and its subequal spine weakly setose apically. Endites 1 to 3 with posterior setae long and thin, clothed with numerous short setules and numbering about 50 on endite 1+2 and 16 on endite 3. Endites 4 to 6 asymmetrical (i.e. distal edge shorter than proximal edge) and with 2 anterior + 3 posterior setae, 2+ 2 and 1+ 2 respectively. Anterior setae of unequal lengths, but shorter and stouter than posterior setae and with a double pectin of stout setules apically. Posterior setae with sparse setules. Endites clothed basally with small spines generally grouped in 3–5s. Endopodite broadly rounded and bearing about 37 posterior setae, all based with a coronet of numerous (>10) small spines. The first 7 or 8 of these setae (essentially those on the medial edge of the thoracopod) stouter and with a one-sided pectin on the distal half of setae, whereas the remaining setae thinner and closely feathered with setules. Exopodite elongate oval and bearing about 38 posterior setae similar to most on the endopodite. Epipodite oval shaped and praepipodite elongated oval shaped, both unadorned.
Gonopods ( Figs 1 View FIGURE 1 C,G) paired, basal parts fused together and about twice the diameter of tubular free apical part. Basal part with a sharp spine apically and apical tube with a digitiform process subapically (DP, Fig. 1 View FIGURE 1 C). Both processes subequal, unhooked and pointing anteriorly. No specimens with gonopod completely everted, but a partly everted one of a paratype ( Fig. 1 View FIGURE 1 D) with complex expansions with medial row of spines.
Abdominal segments increasing in length and narrowing posteriorly from 1 to 6, particularly 5 and 6 ( Fig. 1 View FIGURE 1 E), so that last segment about twice length of first. Cercopods fringed with long setae on lateral and medial edges; length subequal to 6th abdominal segment.
Description. Female. Length 8.5 mm.
Head ( Figs 3 View FIGURE 3 C,D) with first antenna filiform and a little shorter than the eye plus peduncle. Second antenna flattened, a little longer and wider than the eye plus peduncle and terminating in a markedly narrowed, bevelled acute apex. Naupliar eye prominent midway between the compound eyes. Labrum with a prominent recurved spine.
Posterior thoracic segments ( Figs 3 View FIGURE 3 A,B), particularly those of 5–10th segments expanded laterally into simple laminar squarish lobes (L, Figs 3 View FIGURE 3 A,E). Segment 11 expanded separately into a broad blunt triangle and with surface sclerotised and denticulate. Dorsal surface of segment 10 also sclerotised and denticulate. Eleventh segment with two paired small rounded protuberances laterally, one dorsal to the other (P, Figs 3 View FIGURE 3 A,B). Paired brood pouch ( Figs 3 View FIGURE 3 A,B) lying laterally to the genital segments. Anterior surface of pouch adpressed against 11th thoracic segment, wider and with a squarish anteriolateral corner and then narrowing posteriorly. Pouches joined ventrally to a gonopore on a short tubular protrusion and each pouch with a small ventrolateral lobe. In mature ovigerous females, chamber pigmented dorsally and containing numerous smooth-surfaced eggs (at low magnifications).
Eleven pairs of thoracopods, with 11th pair reduced to two stubs, each bearing 3 plumose setae a little shorter than the stub ( Fig. 3 View FIGURE 3 G). Tenth thoracopod pair also reduced, with significantly fewer posterior setae and some reduction in size of endopodite and praepipodite ( Fig. 3 View FIGURE 3 F). Other thoracopods similar in structure to those of male, but generally with fewer posterior setae. Anterior setae as in 5th thoracopod, but those of 2nd and 3rd endite larger.
Abdominal segments papillate, otherwise proportions as in males.
Etymology. The specific name refers to its habitat of markedly acid waters.
Comments. Limited information on this species has been given in a key in Timms (2004) where it was referred to as species f. This publication specifically states “for nomenclature purposes information given here on the new species of Parartemia do not constitute taxonomic descriptions. Characteristics given in the key are no more than a guide to the identity of the organisms.” This species is also referred to as species f in a regional ecology ( Timms, 2009c).
Variability. There is little difference in the morphology of P. acidiphila between and within sites in South Australia and Western Australia. The medial process can be tightly fitted between the ventral processes or well spaced with the divided apex being well expressed ( Fig. 1 View FIGURE 1 F) or just visible. The lateral lobe on the ventral transverse process is variously developed and its spination varies from almost smooth to dense. Many populations, but not all, in Western Australia have weakly lyrate second antennal distal antennomeres ( Fig. View FIGURE 1
1F). The female 11th thoracic segment has variable surface structure so that the two raised areas on each side vary in size from indistinct to distinct mounds. It seems this character may be dependent on age, as its weak development was seen in younger females.
Generally adult males are 11–14 mm in length and adult females 7 –9 mm. Some specimens (e.g. those from Truslove Nature Reserve) are much larger than the type specimens, measuring 20 mm in males and 11 mm in females.
Differential diagnosis. The most distinctive male feature is the second antennal medial process which is triangular and with a bifurcated apex; a unique morphology in Parartemia . It shares with only a few species, including P. triquestra n. sp. (see later), a small conical mound beneath the lateral edge of the ventral processes. The female thoracic segments lack dorsal and dorsolateral projections (cf. P. s e r v e n t y i, P.
longicaudata Linder, 1941 View in CoL ), nor are there narrow sclerotised rings on later thoracic segments (cf. P. zietziana View in CoL ). The posterior thoracic segments are fairly uniform in serial structures, with the 11th most distinct with two minor protuberances laterally. The brood pouch is double (and hence different from those species with single brood chambers (cf. P. cylindrifera View in CoL , P. minuta View in CoL )) and lacks marked posterior outgrowths (as on P. zietziana View in CoL , P. longicaudata View in CoL , P. informis Linder, 1941 View in CoL ). Most Parartemia View in CoL species have a reduced or absent 11th thoracopod, but of the described species only P. e x t r a c t a Linder, 1941 and P. z i e t z i a n a also have it reduced to a stub bearing few setae; it is easily distinguished from both of these species by its simple brood chamber (cf. large lobes in both other species) and its 10th and 11th thoracic segments being neither contracted ( P. zietziana View in CoL ) or expanded ( P. extracta View in CoL ). Its eggs are not distinct microscopically (but SEM studies have not been made).
Type locality. “Little Lake” is a small (<1 ha) salt lake that fills episodically.
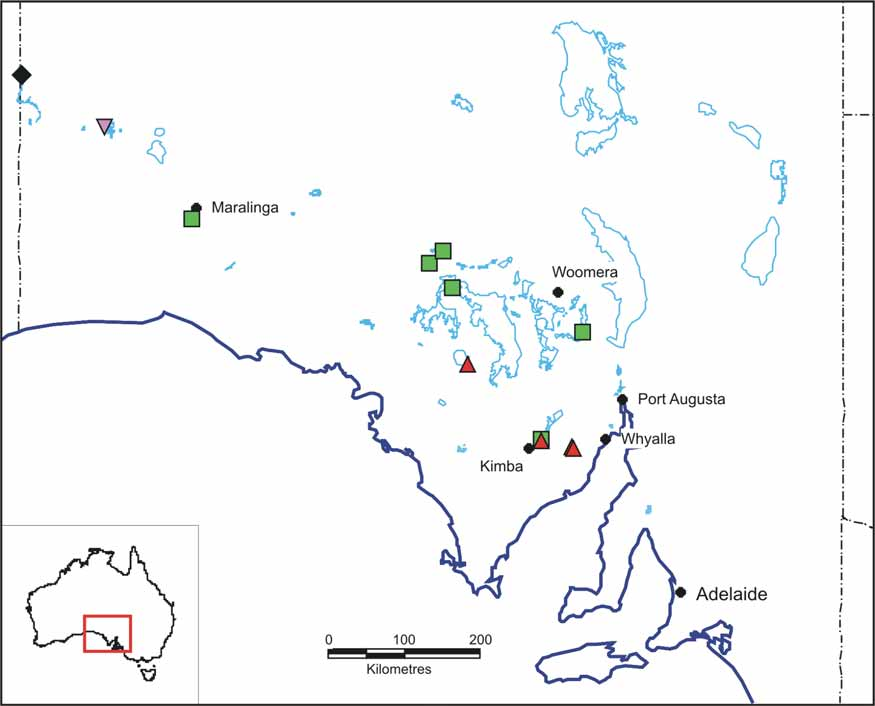
Distribution and ecology. Parartemia acidiphila lives in a few lakes on Eyre Peninsula and in the Gawler Ranges, South Australia ( Fig. 4 View FIGURE 4 ). It is more common in lakes in the Esperance hinterland of Western Australia ( Timms, 2009c). This is the second species shared between southern Western Australia and southern South Australia, the other being P. cylindrifera which is more widely spread in South Australia than this species.
Parartemia acidiphila lives in acid saline lakes. In South Australia it has been recorded over a pH range of 3 – 4 (3 records) (P. Hudson, unpublished data), and in the Esperance hinterland in Western Australia from pH 3.4 to 7.4 (42 records) ( Timms, 2009c). Field salinity range is 68 – 217 g /l in South Australia and 35 – 210 g /l in Western Australia.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Parartemia acidiphila
| Timms, Brian V & Hudson, Peter 2009 |
longicaudata
| Linder 1941 |
P. informis
| Linder 1941 |