Neanura judithae, Smolis & Deharveng, 2017
|
publication ID |
https://doi.org/10.5252/z2017n1a5 |
|
publication LSID |
urn:lsid:zoobank.org:pub:376FF7C0-A9E0-43BD-A7BA-06FA09BB47CF |
|
persistent identifier |
https://treatment.plazi.org/id/74D5D9FD-1334-4C81-86C7-F6048FEB912B |
|
taxon LSID |
lsid:zoobank.org:act:74D5D9FD-1334-4C81-86C7-F6048FEB912B |
|
treatment provided by |
Felipe |
|
scientific name |
Neanura judithae |
| status |
sp. nov. |
Neanura judithae n. sp.
( Fig. 1-3 View FIG View FIG View FIG ; Table 1)
TYPE MATERIAL. — Poland. The Carpathians , Tatra Mts : Waksmundzka valley, 1100-1300 m alt., Norway spruce forest, litter with mosses, decaying wood, 26.IX.1999, Adrian Smolis, holotype ♂ on slide and seven paratypes on slides ( holotype housed in MNHN; paratypes in DIBEC and MSPU) .
OTHER MATERIAL. — Poland. The Carpathians , Tatra Mts : Roztoka valley, 1300 m alt., Norway spruce forest, litter with mosses, 27.IX.1999, Adrian Smolis, juvenile on slide ; Starobociańska valley , 1400 m alt., dwarf-pine shrubs, litter, 7.VIII. 2012, Adrian Smolis, ♀ on slide ; Gąsienicowa valley , 1600 m alt., dwarf-pine shrubs, litter, 31.VII-4.VIII.2014, Adrian Smolis, numerous individuals on slides ( DIBEC) .
DIAGNOSIS. — Habitus typical of the genus Neanura . Dorsal tubercles present and well developed. Body dark bluish grey. Buccal cone short, labrum non-ogival. Head without chaetae C, D, E, Oca and So1. Tubercles Di and De on Th. I present and separate. Thorax and abdomen without free chaetae. Abd. I-III without chaetae De3. Tubercle (Di + Di) of abd. V with 2 + 2 chaetae, chaetae Di3 absent. Cryptopygy absent. Male ventral organ present.
ETYMOLOGY. — The species is named in honour of Dr Judith Najt who has contributed so much to the knowledge of Collembola.
DISTRIBUTION AND ECOLOGY. — Neanura judithae n. sp. has been collected exclusively in Tatra Mts, where it is resident of native Norway spruce forest (upper montane belt, Fig. 3 View FIG ) and dwarf-pine shrubs (subalpine belt). It inhabits litter, mosses and less frequently decaying wood.
DESCRIPTION
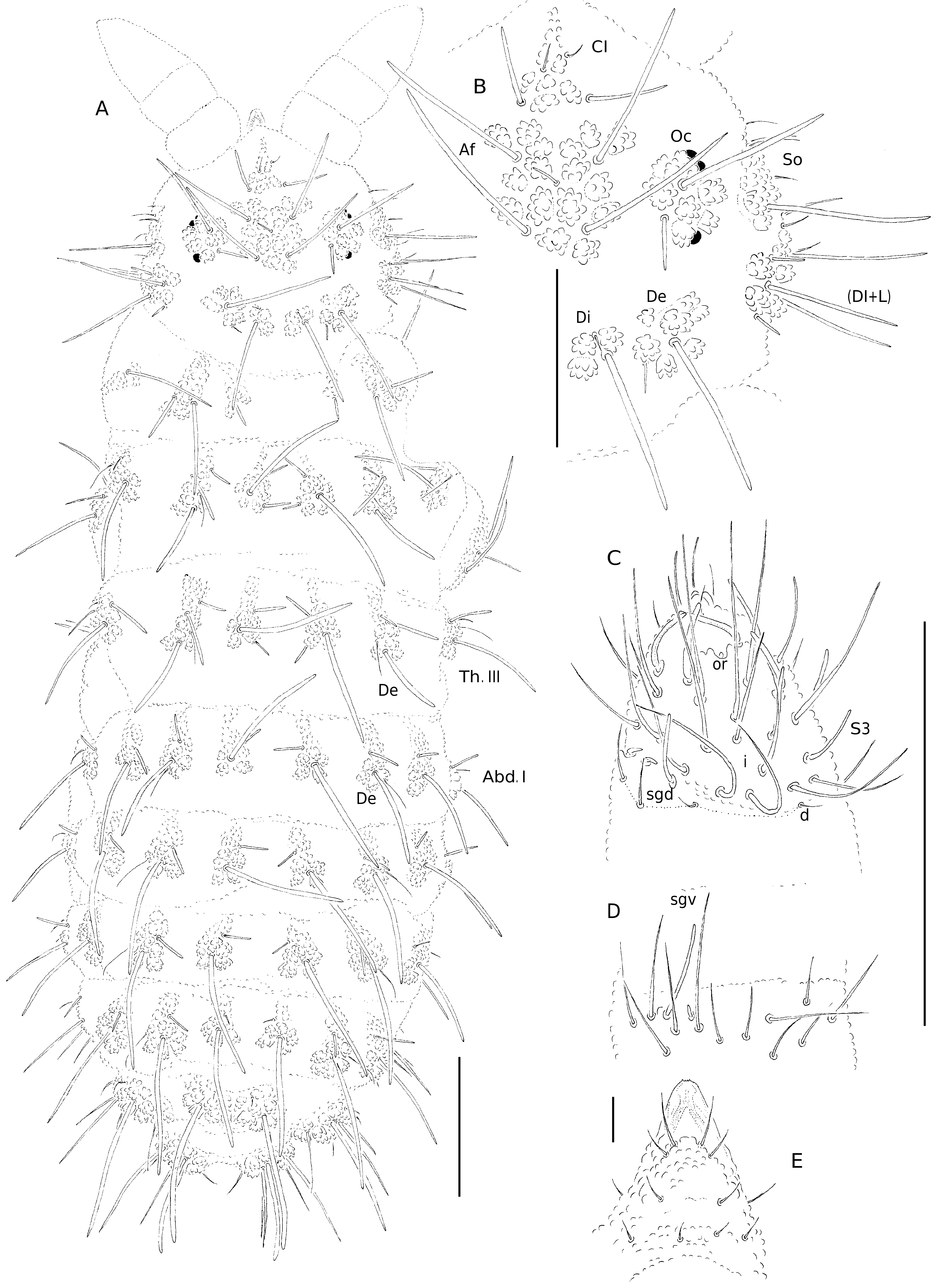
General ( Fig. 1A, B View FIG )
Body length (without antennae): 0.55 to 1.65 mm ( holotype: 0.98 mm). Habitus typical for the genus, parallel and convex.
Colour of the body dark bluish grey, rather intense. 3 +3 medium black eyes, in a typical arrangement for the genus (two anterior and one posterior).
Chaetal morphology ( Figs 1A, B View FIG ; 2 View FIG B-D)
Dorsal ordinary chaetae of five types: long macrochaetae (Ml), short macrochaetae (Mc), very short macrochaetae (Mcc), mesochaetae and microchaetae. Long macrochaetae relatively thick, slightly arc-like or straight, narrowly sheathed, feebly serrated, apically pointed or rounded. Macrochaetae Mc and Mcc morphologically similar to long macrochaetae, but much shorter. Mesochaetae similar to ventral chaetae, thin, smooth and pointed. Microchaetae similar to mesochaetae, but shorter, limited to chaeta Di2 on head and abdominal segments IV-V. S-chaetae of tergites thin, smooth and short, notably shorter than nearby macrochaetae.
Antennae ( Fig. 1C, D View FIG ; Table 1B)
Typical of the genus. S-chaetae of Ant.IV of medium length, subequal and moderately thickened. Apical vesicle distinct, trilobed. Dorsal chaeta d5 absent on ant. III.
Mouthparts ( Fig. 1E View FIG )
Buccal cone relatively short with labral sclerifications nonogival. Labrum chaetotaxy: 4/2, 4. Labium with four basal, three distal and four lateral chaetae, papillae × absent. Maxilla styliform, mandible thin and tridentate.
Dorsal chaetotaxy and tubercles ( Fig. 1A, B View FIG ; 2B View FIG ; Tables 1A, C) Chaetotaxy of head reduced, chaetae C, D, E, Oca and So1 absent.Tubercles Di on Th. I differentiated and not fused with tubercles De. Thorax and abdomen without free chaetae. On Th. II-III, chaetae De2 longer than De3. Abd. I-III without chaetae De3. On Abd. I-III, the line of chaetae De1-chaeta s perpendicular to the dorsomedian line. On Abd. V, tubercles Di fused along midline and with 2+ 2 chaetae, chaetae Di3 absent. No cryptopygy, Abd. VI well visible from above.
Ventral chaetotaxy ( Fig. 2A View FIG ; Table 1C)
On head, groups Vea, Vem and Vep with 3-4, 4, 4 chaetae respectively; group Vi with 6 chaetae. On Abd. IV, furcal rudiment without microchaetae. On Abd. V, chaeta Vl present. Male with thick and forked chaetae (“male ventral organ”) on even anal valves (Abd. VI), in groups Ag (Abd. V) and Ve (Abd. IV).
Legs ( Table 1C)
Claw without internal tooth. On tibiotarsi, chaeta M present and chaetae B4 and B5 relatively short (ratio- inner edge of claw: chaeta B4: chaeta B5; 12: 9: 11) and pointed.
REMARKS
Neanura judithae n. sp. is morphologically most similar to N. parva , resembling that species in having a strong reduction of chaetotaxy on head (chaetae Oca, C, D and E absent) and absence of free chaetae on dorsal side of the body. Nevertheless, they are readily distinguished by different shape of labral sclerifications (non-ogival in N. judithae n. sp. vs ogival in N. parva , Figs 1E View FIG ; 5F View FIG ), number of chaetae De on Th. III ( 4 in N. judithae n. sp. vs 5 in N. parva ), number of chaetae De on Abd. I-III ( 3 in N. judithae n. sp. vs 4 in N. parva ) and number of chaetae Di on Abd. V (2+ 2 in N. judithae n. sp. vs 3 + 3 in N. parva , Figs 2B View FIG ; 5G View FIG ). Furthermore, N. judithae n. sp. is characterized by complete fusion of tubercles Di on Abd. V (in N. parva tubercles are fused only distally, Fig. 5G View FIG ). The new species seems to be a true endemic to the Tatra Mts, whereas N. parva has a much larger distribution range, extending across the whole Western and Eastern Carpathians, Sudetes, Southeastern Alps, and some lowland/upland localities in Poland (Smolis pers. comm.).
| MNHN |
Museum National d'Histoire Naturelle |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.