Micropygomyia (Coquillettimyia) chiapanensis ( Dampf, 1947 )
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4105.5.3 |
|
publication LSID |
lsid:zoobank.org:pub:53AE9F86-CA4B-4F91-910B-A3E89C375EC1 |
|
DOI |
https://doi.org/10.5281/zenodo.5615986 |
|
persistent identifier |
https://treatment.plazi.org/id/03DF9B5F-8222-FFBB-818B-F88671E1FAA0 |
|
treatment provided by |
Plazi |
|
scientific name |
Micropygomyia (Coquillettimyia) chiapanensis ( Dampf, 1947 ) |
| status |
|
Micropygomyia (Coquillettimyia) chiapanensis ( Dampf, 1947) View in CoL
Phlebotomus chiapanensis Dampf, 1947: 108 View in CoL (♂), (Chiapa de Corzo, Chiapas, México).
Lutzomyia chiapanensis (Dampf) View in CoL : Young & Duncan, 1994: 649 (taxonomic history, ♂ and ♀ figures, keys); Ibáñez-Bernal, 2003: 120 (references, ♂, ♀, figures).
Micropygomyia (Coquillettimyia) chiapanensis (Dampf) View in CoL : Galati, 2003: 33, 82 (listed, taxonomic keys); Ibáñez-Bernal et al., 2011: 34 View Cited Treatment (Veracruz records); Ibáñez-Bernal et al., 2015: 157 View Cited Treatment (Chiapas records).
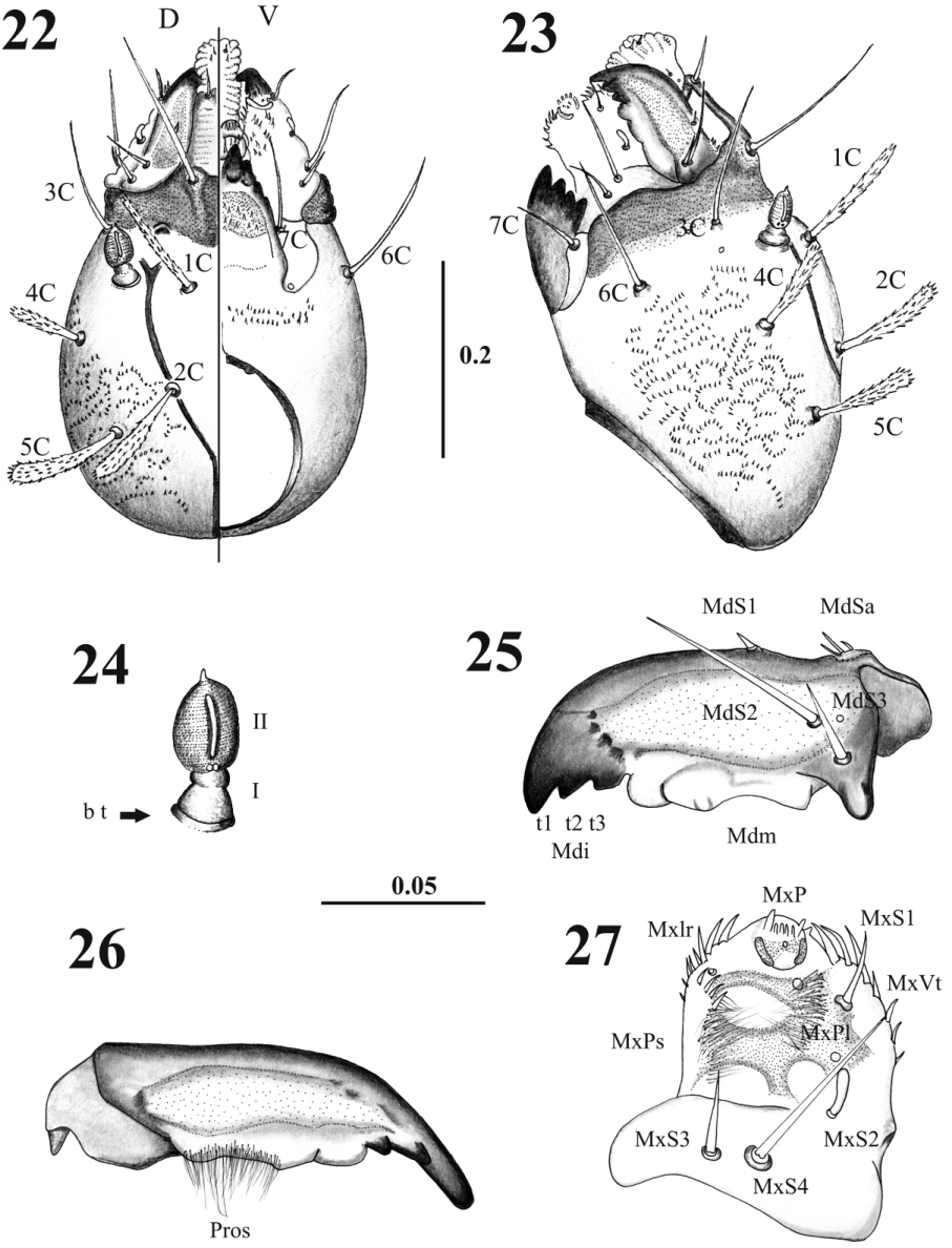
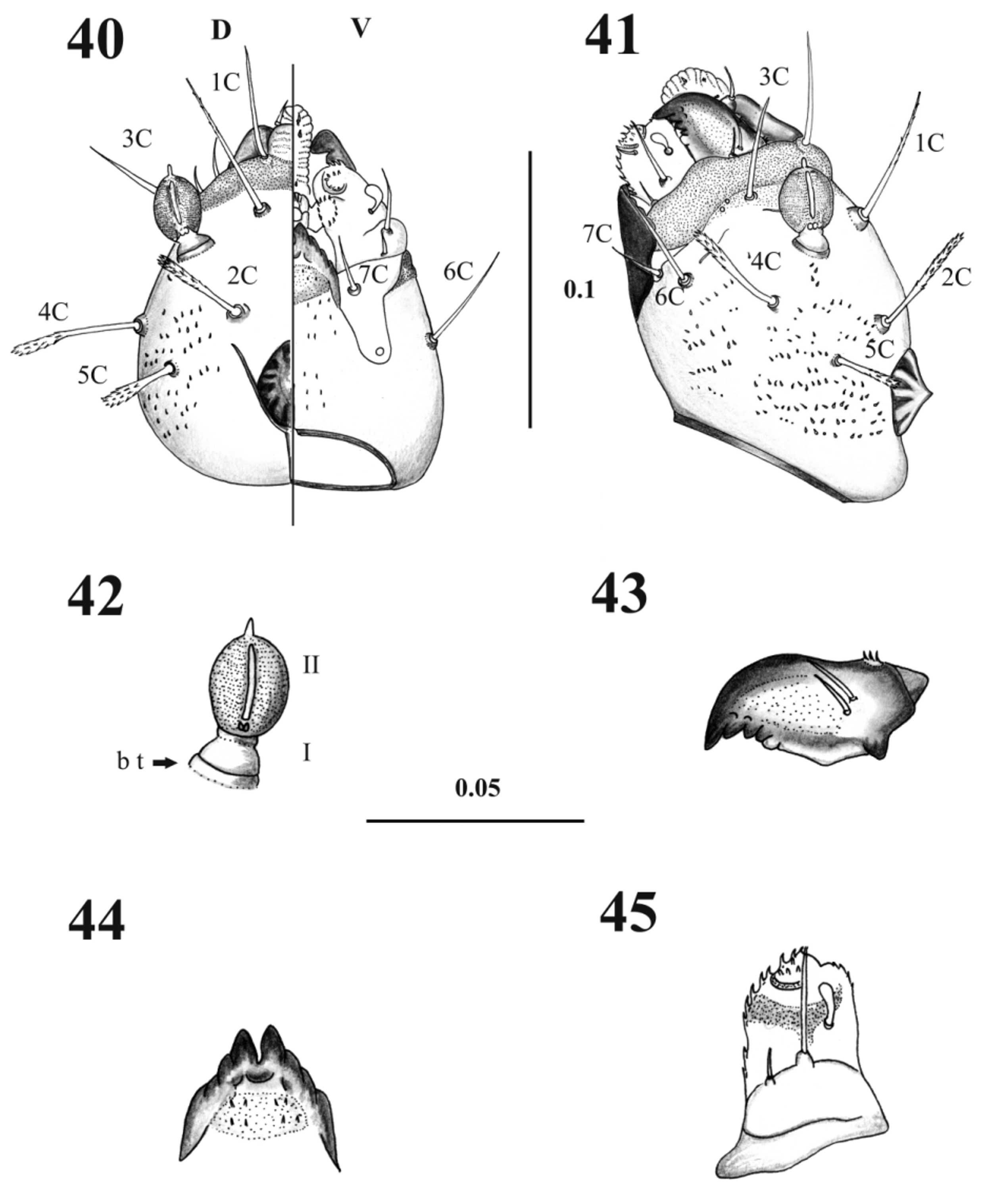
Pupa description. Length without larval exuvia: 2.41 ± 0.08 mm (n= 7) ( Figs. 1 View FIGURES 1 – 3 , 4 View FIGURES 4 – 9 ). Head. Antennal sheath showing sketch of each one of the adult flagellomeres; post-ocular lobe lateral situated, cone-shaped and with divisions. Mouthparts sheath smooth, posterior directed and in ventral position ( Figs. 2 View FIGURES 1 – 3 , 5 View FIGURES 4 – 9 ). Head chaetotaxy as in Table 1 View TABLE 1 ( Figs. 2 View FIGURES 1 – 3 , 6–7 View FIGURES 4 – 9 ).
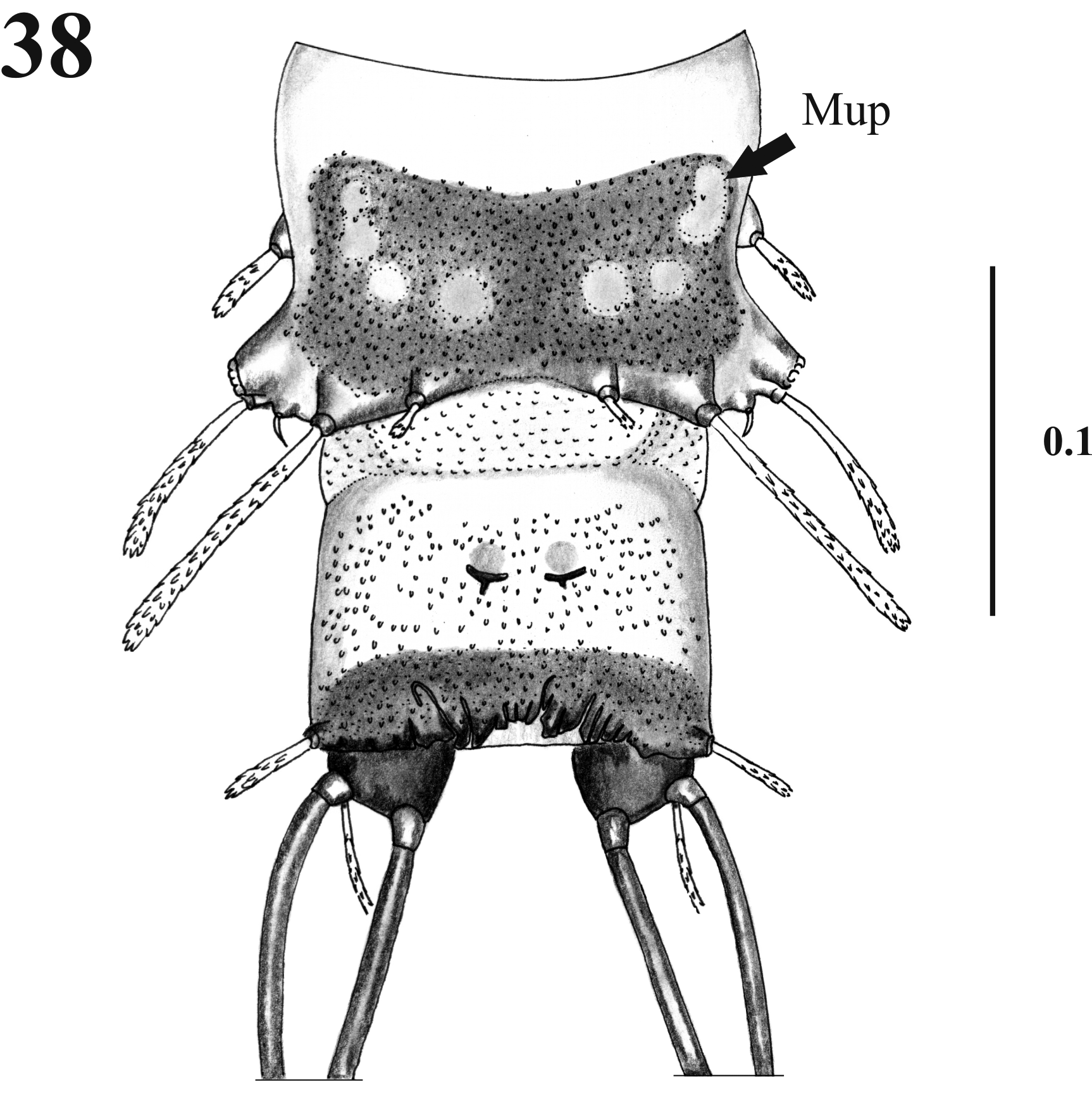
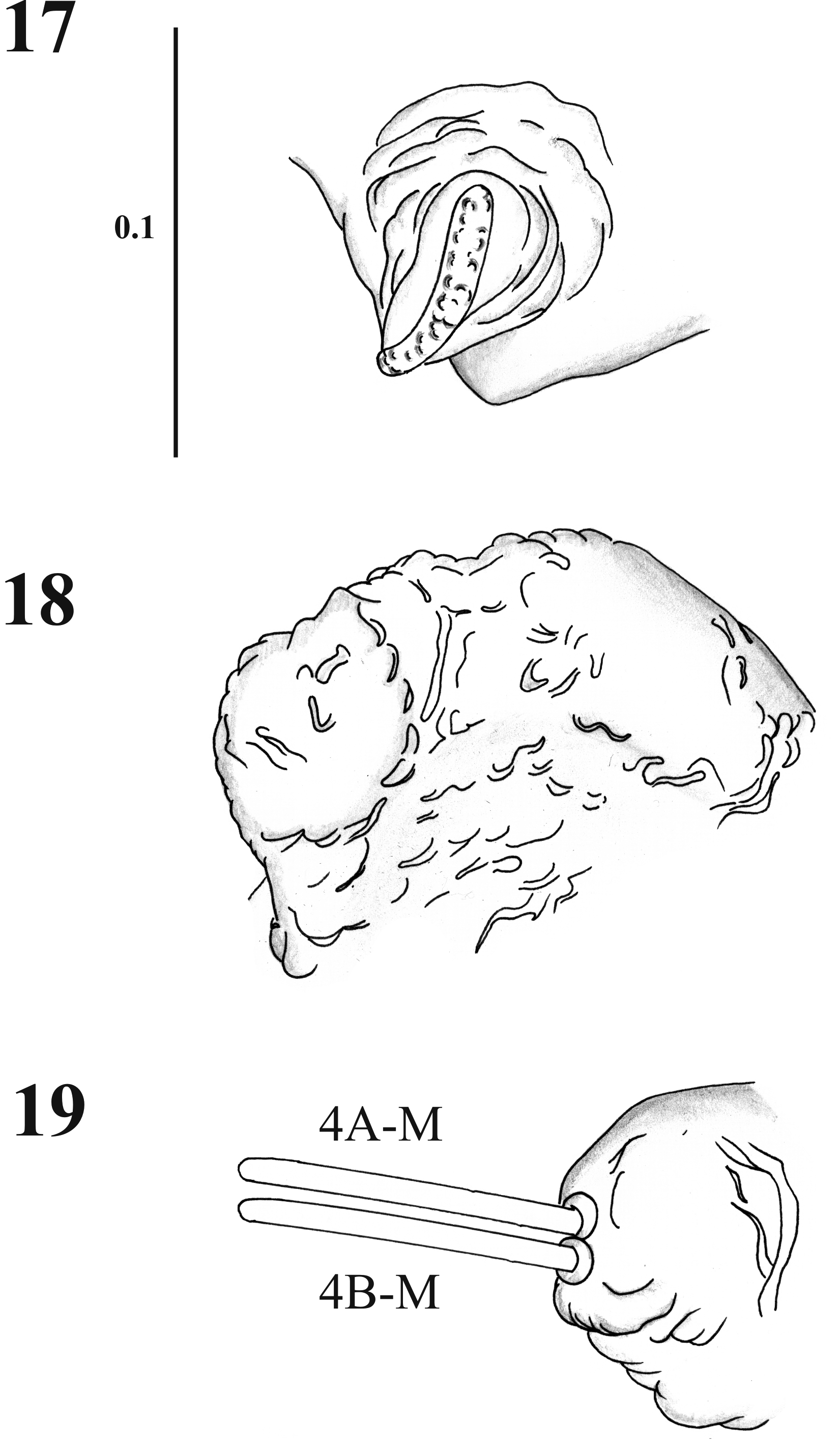
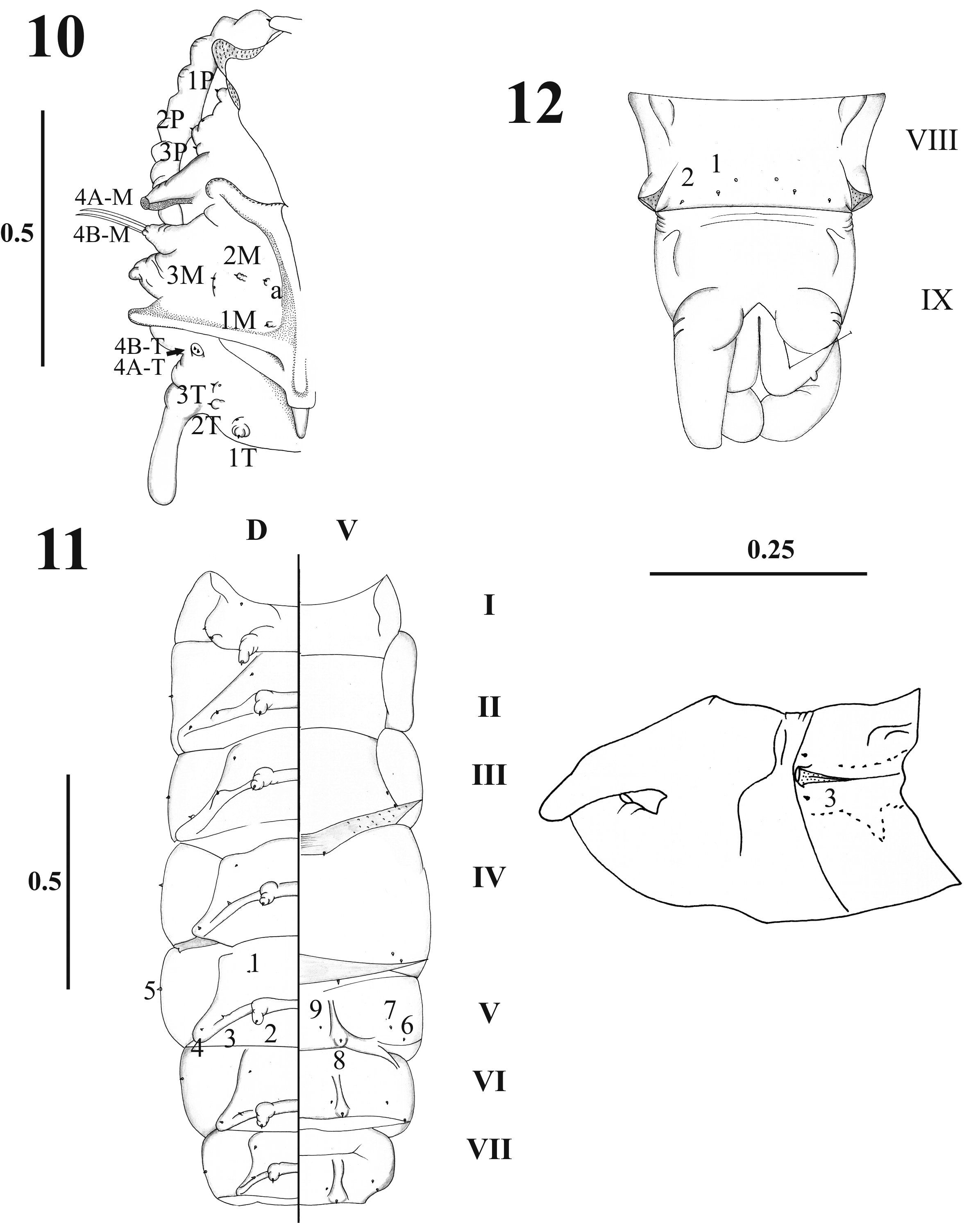
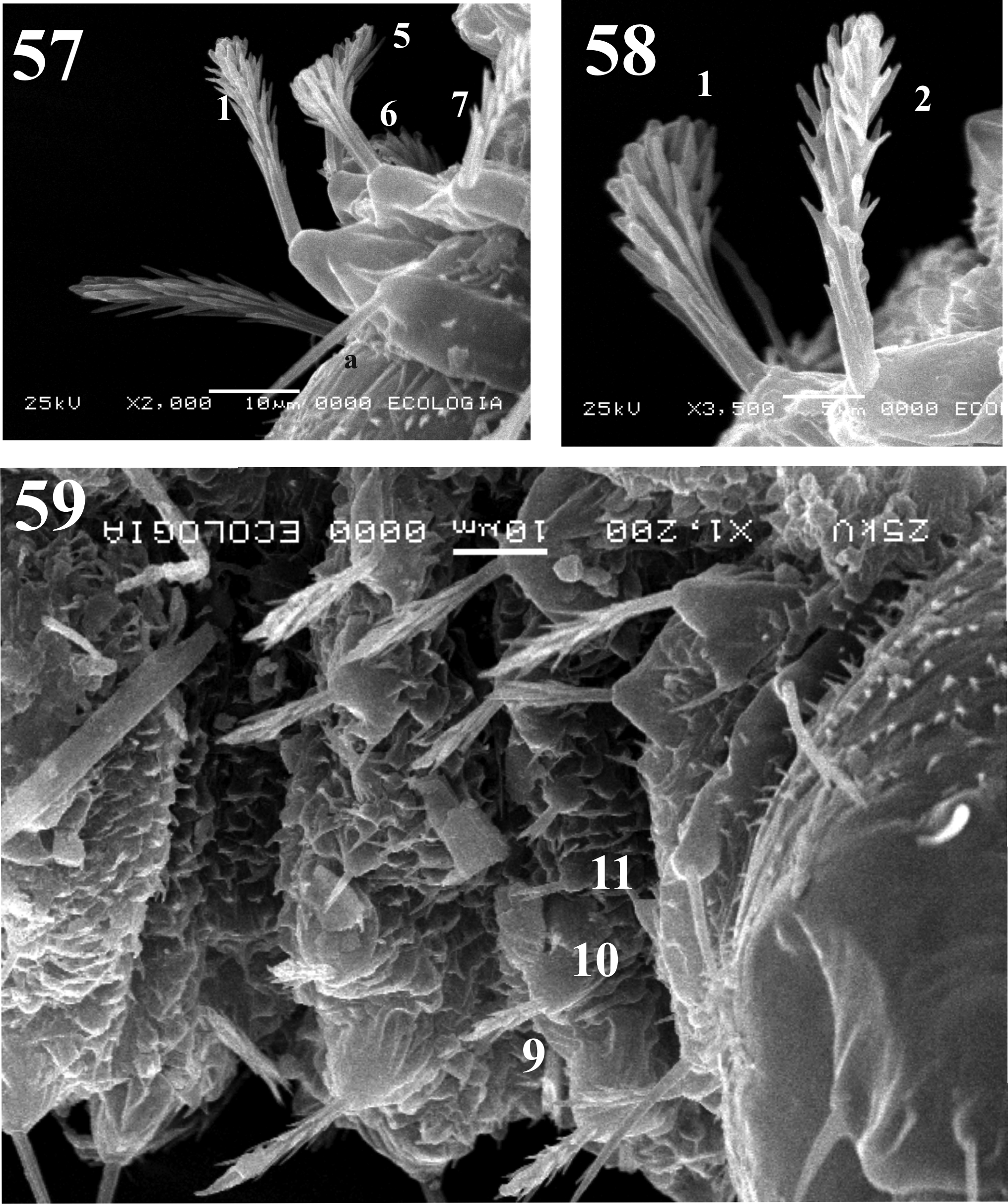
Thorax. With 11 pairs of setae; prothorax with three setae ascendant disposed in relation to the ventilator orifice ( Figs. 9 View FIGURES 4 – 9 , 17 View FIGURES 17 – 19 ); mesothorax with six setae of which two are comparatively large (length 0.08 + 0.001 mm), stout chaetic prealars originate on tubercles (t. pr–a) ( Figs. 10–19 View FIGURES 10 – 12 View FIGURES 13 – 16 View FIGURES 17 – 19 ), and with an accessory seta “a” anterior to 1M; mesonotal tubercle with irregular border ( Fig. 18 View FIGURES 17 – 19 ); metathorax with five sensillae being three basiconic and two styloconic which are implanted on a tubercle near the base of halters ( Fig. 10 View FIGURES 10 – 12 ); thorax chaetotaxy as in Table 1 View TABLE 1 ( Fig. 20 View FIGURES 20 – 21 ).
Abdomen. With nine visible segments, the width of every segment is twice its own length, and they decrease gradually in size towards the caudal region ( Figs. 1, 3 View FIGURES 1 – 3 , 11–12 View FIGURES 10 – 12 ). Abdominal segment I: Tergum with four pairs of sensilla, dorsal posterior internal (2) styloconic, placoid sensillum near sensillum 2 in all abdominal segments; pleuron and sternum covered with the thoracic appendage sheaths. Abdominal segment II–III: Tergum with five pairs of sensilla, the first four sensilla similar in form and location to the abdominal segment I, sternum normally without sensilla, but two specimens with setae 6–7 present in sternum III as showed in Fig. 11 View FIGURES 10 – 12 . Abdominal segments IV–VII: each tergum with five sensilla similarly distributed as previous segments; each sternum with four sensilla ( Figs. 11 View FIGURES 10 – 12 , 13–16 View FIGURES 13 – 16 ); pleura and sterna covered by the thoracic appendage sheaths. Abdominal segment VIII: male and female both with three pairs of very small basiconic sensilla, segment in nature covered by larval exuvia ( Figs. 3 View FIGURES 1 – 3 , 12 View FIGURES 10 – 12 ). Abdominal segment IX: covered by the larval exuvia, but when removed, sexual morphological differences can be observed: in males there are two lobes at each side, one of them is simple covering the lateral lobe and the other one is divided containing the gonocoxite and gonostylus; in females there are two simple short lobes at each side, one covering the oviscape and the other the cercus, without setae ( Figs. 3 View FIGURES 1 – 3 , 12 View FIGURES 10 – 12 ). Abdominal chaetotaxy as in Table 1 View TABLE 1 ( Fig. 21 View FIGURES 20 – 21 ).
Remarks. In this work, we compare some characteristics observed in Mi. chiapanensis with those observed by Leite et al. (1991) in Lutzomyia longipalpis (Lutz & Neiva) ; however, chaetotaxy and morphological comparison was made mainly with those of Dampfomyia beltrani (Vargas & Díaz-Nájera), because the last one is the only species in which a detailed chaetotaxy description has been made, despite they belong to different genera (sensu Galati).
Pupae of Mi. chiapanensis , Da. beltrani and Lu. longipalpis are all of different size, as are the adults, with Da. beltrani being the largest and Mi. chiapanensis the smallest. Chaetotaxy of Mi. chiapanensis is more similar to Da. beltrani than to Lu. longipalpis , as the latter species has a bifurcate pre-alar seta at the thorax, and only six pairs of setae have been described in the abdominal segments. Micropygomyia chiapanensis presents one pair of placoid sensilla in the mesothorax and in the metathorax (3M and 3T), and in the same abdominal segments as Da. beltrani . In Lu. longipalpis there is only one placoid sensillum near the metathoracic seta. These three species can further be differentiated by the shape and size of setae implantation tubercles. In Mi. chiapanensis nearly all thoracic setae and also the abdominal dorsal setae (see the dorsal posterior internal 2 seta in figure 21) are styloconic; in Da. beltrani they are basiconic, but in Lu. longipalpis thoracic setae are basiconic, and the dorsal posterior internal and intermediate abdominal seta are styloconic. Colas-Berlcour (1928) and Barretto (1941) mentioned that differences between the shape and size of seta implantation tubercles can be useful for the recognition of species. Hanson (1968) mentioned that the pupa of Mi. trinidadensis presents prominent implantation tubercles which are similar to those found in Mi. chiapanensis . Another difference is observed in the mesothoracic setae: in Mi. chiapanensis there are five pairs, in Da. beltrani six pairs, and in Lu. longipalpis only three pairs of setae have been described.
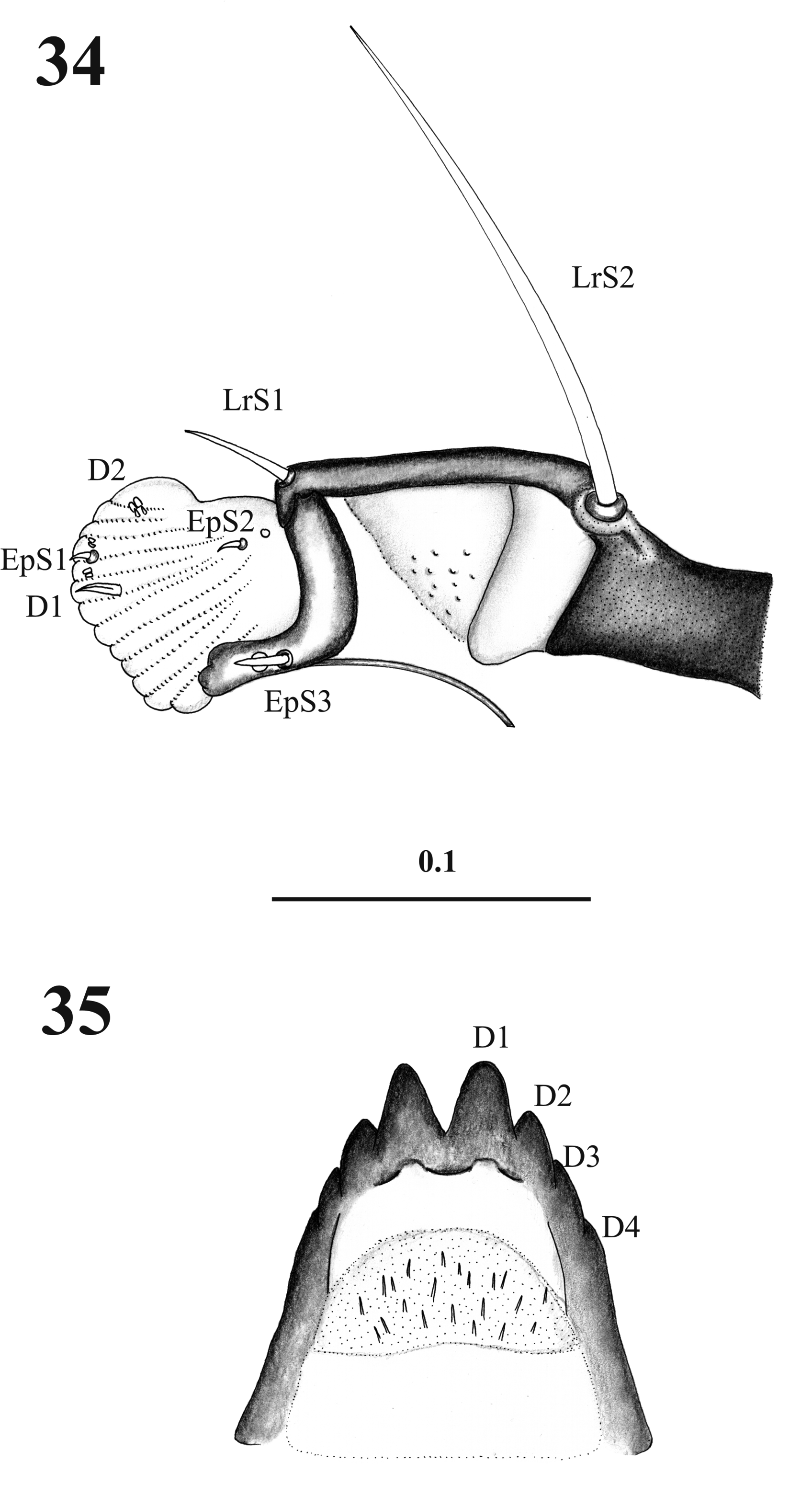
Fourth instar larva description (L4). Body length (excluding caudal setae): 3.14 ± 0.66 mm ( Fig. 28 View FIGURES 28 – 33. L 4 ); head length: 0.37 ± 0.002 mm, maximal width: 0.32 mm (n= 6); antenna length: 0.07 ± 0.001 mm, (n= 5). Head with antennal basal tubercle annular; segment I as long as 0.56 the length of segment II (distal); segment II ovoid, with blunt apex bearing an apical basiconic and a coeloconic sensillum ( Figs. 24–30 View FIGURES 22 – 27. C View FIGURES 28 – 33. L 4 ). Gena and frontoclypeal apotome with medial region and posterior areas covered with spiculae, imbricately distributed ( Figs. 22–23 View FIGURES 22 – 27. C ). Head with seven setae; cephalic apotome with two setae, gena with three setae, postgena with one seta, and subgena with one simple seta which is as long as 0.4 the length of the postgenal seta ( Figs. 22–23 View FIGURES 22 – 27. C , 31 View FIGURES 28 – 33. L 4 ). Mouthparts ( Fig. 32 View FIGURES 28 – 33. L 4 ): labrum with two simple setae; epipharynx with two spiniform laterobasal setae, median teeth spiniform, apical teeth usually spiniform but sometimes capitate, apical setae spiniform, and seta of the U-sclerite spiniform ( Fig. 34 View FIGURES 34 – 35 ); mandible length: 0.016 ± 0.01 mm, maximum width: 0.06 ± 0.04 mm (n= 8), spinose area with three or four spines, incisive lobe (Mdi) strongly sclerotized and dark, incisive area with three teeth, t1 blunt, t2 and t3 blunt or sharp with discontinuous border, molar area with transversal groove and bilobed ( Figure 25–26 View FIGURES 22 – 27. C ); maxilla length: 0.011 ± 0.01 mm, maximal width: 0.08 ± 0.05 mm (n= 4), with three setae, seta 1 (MxS1) simple and apical; seta 2 (MxS2) spatulate; setae 3 (MxS3) and 4 (MxS4) simple, seta 3 about 0.43 as long as seta 4, both basally situated; lacinia poorly differentiated, with maxillary brush (Mxlr) on dorso-lateral border ( Fig. 27 View FIGURES 22 – 27. C ). In dorso-lateral view maxillary ventral teeth (MxVt) are seen, and in ventral view the maxillary plate (MxPl) seems sclerotized and setose ( Fig. 27 View FIGURES 22 – 27. C ). Postmentum with fine spicules at median and basal regions, with four teeth, t1 large with blunt apex and irregular internal border ( Fig. 35 View FIGURES 34 – 35 ).
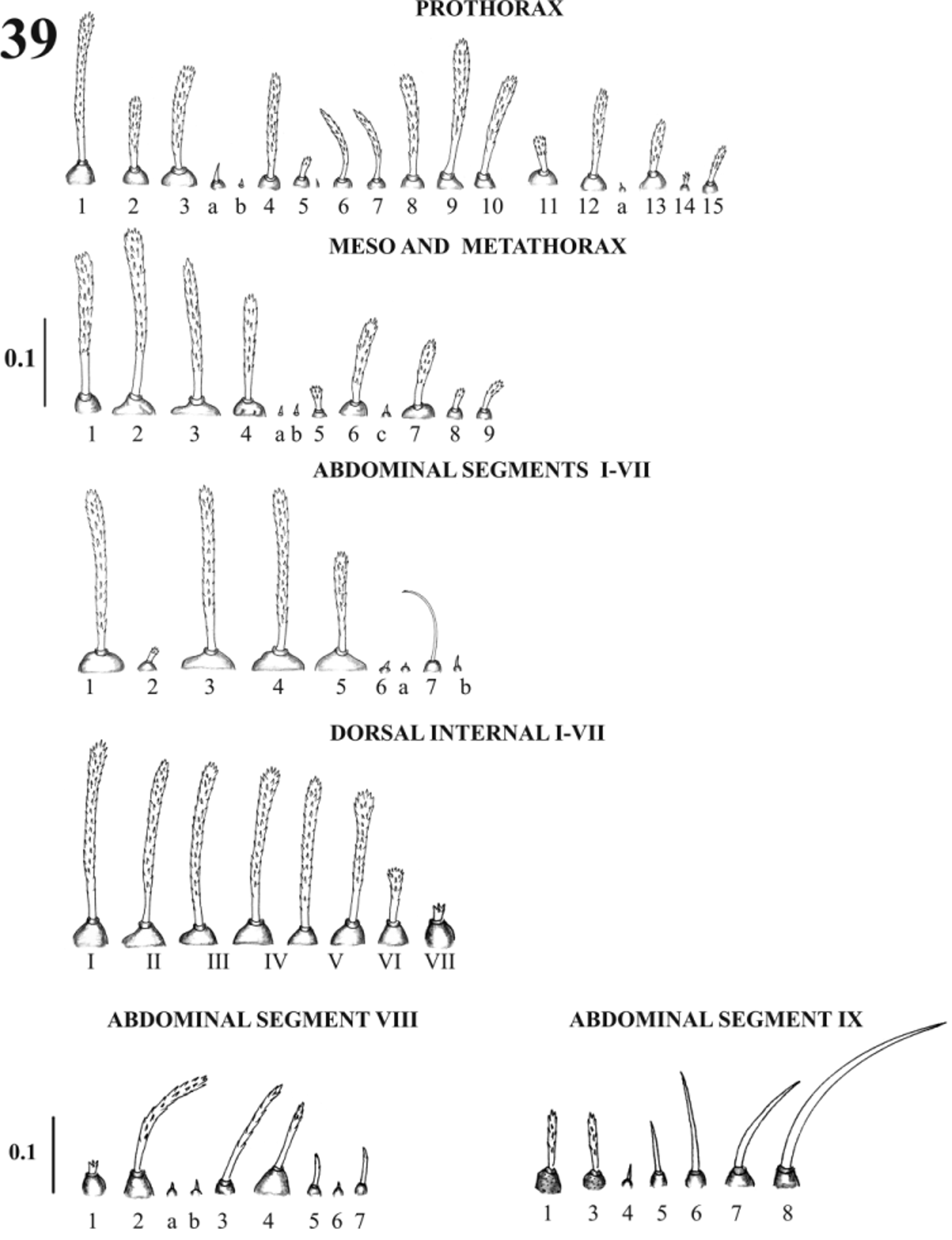
Thorax: anterior prothorax with nine setae, posterior prothorax with nine setae, meso and metathorax with 12 setae ( Figs. 29, 33 View FIGURES 28 – 33. L 4 , 36, 37 View FIGURES 36 – 37 , 39 View FIGURE 39 ). Thoracic chaetotaxy as in Table 2 View TABLE 2 .
Abdomen: nine visible segments; segments I–VII each with nine setae ( Figs. 36, 37 View FIGURES 36 – 37 , 39 View FIGURE 39 ); segment VIII with eight setae ( Figure 36–38 View FIGURES 36 – 37 View FIGURE 38 ), dorsal internal seta small (1), and dorsal intermediate seta (2) 5.3 times the length of the first; segment IX with two setae on each caudal lobe; caudal setae length: 21.26 ± 0.24 mm; anal lobe with five setae, external post-anal (5) with 2.6 times the length of the external lobe seta (1). Segment VIII with a rectangleshaped pigmented area that covers the lobes of setae 1 and 2–VIII, and muscle plaques ( Borkent 2012) distributed as showed ( Fig. 38 View FIGURE 38 ). Abdominal chaetotaxy as in Table 2 View TABLE 2 .
Third instar larva description (L3). Similar to L4 except for the following characteristics. Body length (excluding caudal setae): 2.49 ± 0.11 mm, head length: 0.30 ± 0.05 mm, head width: 0.22 ± 0.01 mm (n=3); antenna length: 0.05 (n=4). Antennal segment I about 0.45 as long as segment II; segment II ovoid, with one apical sensillum which has the apex blunt and also with a coeloconic sensillum. Gena with posterior area covered with disperse spicules in an imbricate pattern, postmentum without spicules. Mouthparts not studied. Abdominal segment VIII without pigmented area; abdominal segment IX caudal lobe with four setae; caudal setae length: 0.95 ± 0.06 mm. Head, and thorax-abdomen chaetotaxy in Table 2 View TABLE 2 .
Second instar larva description (L2). Similar to L4 and L3 except for the following characteristics. Body length (excluding caudal setae): 0.96 ± 0.17 mm; head length: 0.162 ± 0.01 mm, width: 0.15 ± 0.01 mm (n=4); antenna length: 0.04 ± (n= 3); antennal segment I 0.50 the length of II. Gena with posterior area covered with spaced spicules irregularly disposed, postmentum with spicules. Abdominal segment VIII without pigmented area; abdominal segment IX caudal setae length: 0.69 ± 0.03 mm. Head and thorax-abdomen chaetotaxy as in Table 2 View TABLE 2 .
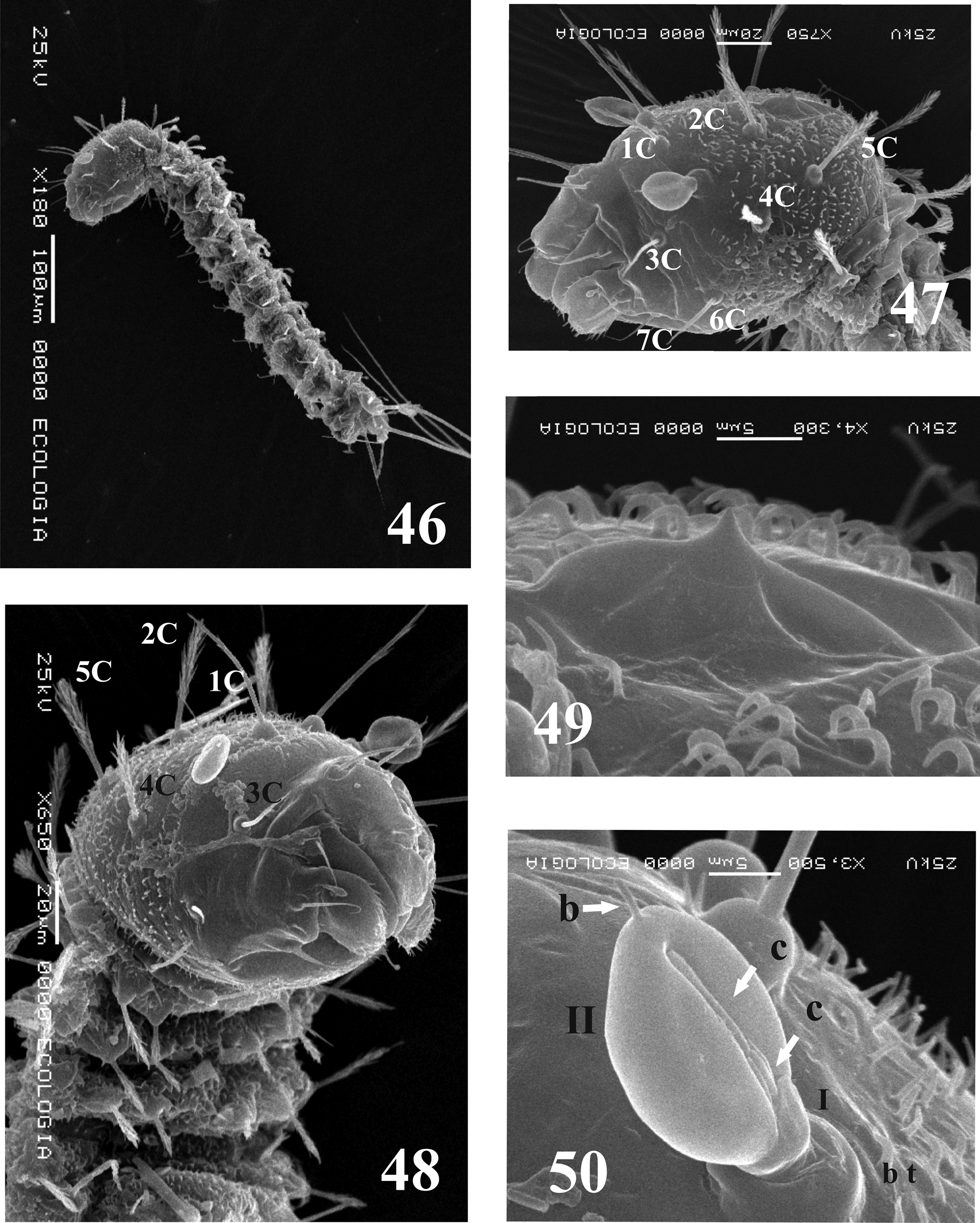
First instar larvae description (L1). Similar to previous described larva instars except for the following characteristics. Body length (excluding caudal setae): 0.61 ± 0.17 mm ( Fig. 46 View FIGURES 46 – 50. L 1 ); head length: 0.12 ± 0.02 mm, width: 0.11 ± 0.01 mm (n=6); antenna length: 0.03 mm (n=5). Oviruptor present. Antennal segment I length about 0.30 as long as segment II; segment II ovoid, apical sensilla as described for other instars ( Figs. 42 View FIGURES 40 – 45 , 50 View FIGURES 46 – 50. L 1 ); genal posterior area with spicules irregularly disposed, some in the postmentum ( Figs. 40, 41 View FIGURES 40 – 45 ); Mouthparts ( Figs. 43–45 View FIGURES 40 – 45 , 52–54 View FIGURES 51 – 54. L 1 ). Head chaetotaxy as in Table 2 View TABLE 2 ( Figs. 40, 41 View FIGURES 40 – 45 , 47, 48 View FIGURES 46 – 50. L 1 , 51 View FIGURES 51 – 54. L 1 ). Anterior prothorax with six setae, posterior prothorax with eight setae ( Table 2 View TABLE 2 Figs. 55–59 View FIGURES 55 – 56 View FIGURES 57 – 59 ). Ventilator orifices not observed ( Fig. 56 View FIGURES 55 – 56 ). Thorax and abdomen whitish, head when emerged grey, later pale brown. Meso and metathorax each with 10 setae ( Table 2 View TABLE 2 and Figs. 55, 56 View FIGURES 55 – 56 ). Abdominal segments I–VII each with eight setae; Abdominal segment VIII with eight setae ( Table 2 View TABLE 2 ) disposed as in Figs. 55 and 56 View FIGURES 55 – 56 . Abdominal segment IX, caudal setae length: 0.67 ± 0.01 mm. Head and thorax-abdomen chaetotaxy as show in Table 2 View TABLE 2 .
Remarks. In relation to the position and morphology of head setae, we only can compare Mi. chiapanensis with Da. beltrani , because Micropygomyia species and other species of other genera previously described have not been completely described. The head chaetotaxy in larvae of Mi. chiapanensis presents the same number of setae as in the majority of the sandfly larvae described. We observed that the position of setae on the head is different in Mi. chiapanensis (probably similar disposed in other Micropygomyia species) and in Dampfomyia beltrani ; in Mi. chiapanensis 5C is posterior to 2C; 4C is posterior to the antenna and the antenna is nearly in the same anteroposterior level of 1C. In Da. beltrani 5C is nearly in the same level of 2C and, anterior to them, 4C, the antenna and 1C are also nearly in the same level. The relative position of setae with respect to other setae was considered by Hanson (1968) for the description and classification of the fourth instar larvae species. According to Hanson´s description, the position of cephalic setae in Mi. (Coquillettimyia) cayennensis and Mi. (Sauromyia) trinidadensis is similar to that found in Mi. chiapanensis . With relation to the morphology of head setae, we observed the cephalic setae barbed in Da. beltrani , while in Mi. chiapanensis they are spinulate.
Sand fly larvae mandibles present characters that could be extremely useful for taxa recognition, and also for phylogenetic analyses as in other Diptera groups ( Wood & Borkent 1989; Oosterbroek & Courtney 1995), but within Phlebotominae it is necessary to assemble more information, as currently few descriptions exist ( Arrivillaga et al. 1999; Arrivillaga & Feliciangeli 2000; Pessoa et al. 2008, and Montes de Oca-Aguilar et al. 2014). Arrivillaga & Feliciangeli (2000) referred that in phlebotomine larvae the mandible morphology, in particular the incisor lobe and molar lobe, is important for taxonomic recognition. In Da. beltrani the incisor teeth lobe is different in shape, with Mdi t1 and Mdi t2 both bilobulate, and the molar lobe (Mdm) simple, while in Mi. chiapanensis the teeth are prominent, simple and blunt, with the Mdm bilobulate.
In addition, larva mandible chaetotaxy of sand flies could be important in systematics and taxonomy approaches, as the mandible of the L4 of Lutzomyia, Dampfomyia and Micropygomyia ( Arrivillaga et al. 1999; Arrivillaga & Feliciangeli 2000; Montes de Oca-Aguilar et al. 2014; in this work) only have three setae as can be observed in dorsal view, whereas Evandromyia spp. additionally have three setae in ventral view ( Pessoa et al. 2008). The latter condition could be autopomorphic for this genus, but it is necessary to add descriptions of more genera sensu Galati (1995, 2003).
According to the morphological classification of the larval antenna of Leite & Williams (1996), Mi. chiapanensis corresponds to category IV for Neotropical larvae, as is the case of Mi. trinidadensis and Mi. cayennensis . The category IV is the more commonly observed antenna shape in sand flies larvae previously described. Species grouped using this character do not correlate to those proposed by the classification systems based on adults ( Young & Duncan 1994; Galati 1995, 2003), but relevance of this character needs to be valued based in the study of more species. L1 of Mi. chiapanensis can be differentiated from L4 by the absence of ventilator orifices of the prothorax, presence of oviruptor and thorax chaetotaxy. The prothorax has dorsal internal, dorsal intermediate, ventral anterior external (5) setae, but a lateroventral posterior (11) seta is absent (present in L2). Also, with the transition of L1 to L2 some setae change their form: in the prothorax in the ventral posterior external and ventral posterior internal setae; in the meso and metathorax, in the ventral internal (8) and ventral external setae (5) (see Table 2 View TABLE 2 ). Additionally, on the meso and metathorax, the lateral anterior seta (4) is associated only with one lateral accessory superior seta (a), whereas in L2 two accessory setae (lateral accessory superior “a” and lateral accessory inferior b), both basiconic and spiniform are associated to it (see Table 2 View TABLE 2 ). The abdominal chaetotaxy is constant but the dorsal anterior intermediate seta varies between spiniform to spinulate; and when the larva change to L2, the lateroventral seta (5) change from simple to spiniform.
The L2 of Mi. chiapanensis differ from L1 because oviruptor is absent and the ventilatory orifices of the prothorax are present. In the anterior prothorax, the dorsal internal, dorsal intermediate and ventral external setae are present, and the dorsal intermediate (1) seta is 0.21 the length to the dorsal internal, while in L3 the dorsal intermediate is as long as 0.5 the length to the dorsal internal. Both instars are different because in the meso and metathorax the L2 has two accessory spiniform setae, superior “a” and inferior “b”, while in L3 the lateralbasal (5) seta is spinulate.
L3 and L4 of Mi. chiapanensis are similar, and can only be differentiated by the size of the body and cephalic capsule, the more defined imbricated disposition of the gena spicules and because segment VIII is pigmented in L4.
Hanson (1968) used the chaetotaxy of the abdominal segment VIII as the principal characteristic to differentiate Mi. (Coquillettimyia) cayennensis and Mi. (Sauromyia) trinidadensis . In Mi. trinidadenis both, the dorsal internal (1) and dorsal intermediate (2), are short. In contrast, in Mi. cayennensis the dorsal intermediate (2) is many times longer than dorsal internal (1), as also observed in Mi. chiapanensis .
In spite of the relative simplicity of Hanson’s (1968) descriptions, it is possible to observe in his illustrations that Mi. cayennensis and Mi. trinidadensis exhibit the same pigmentation pattern than Mi. chiapanensis . L4 of Mi. cayennensis has a different chaetotaxy pattern in the prothorax, the ventral anterior external (5) seta is absent; meso and metathorax lateral accessory superior (a) and inferior (b) are absent, and lateral accessory b is absent in the abdominal segment VIII.
In contrast, L4 of Mi. trinidadensis present more or less the same number and type of setae, except for the following differences: prothorax with ventral anterior external (5) seta spiniform, dorsal posterior external (10) clavate; meso and metathorax with dorsal external (3) seta clavate, and lateroventral accessory a seta absent in the abdominal segments I–VII.
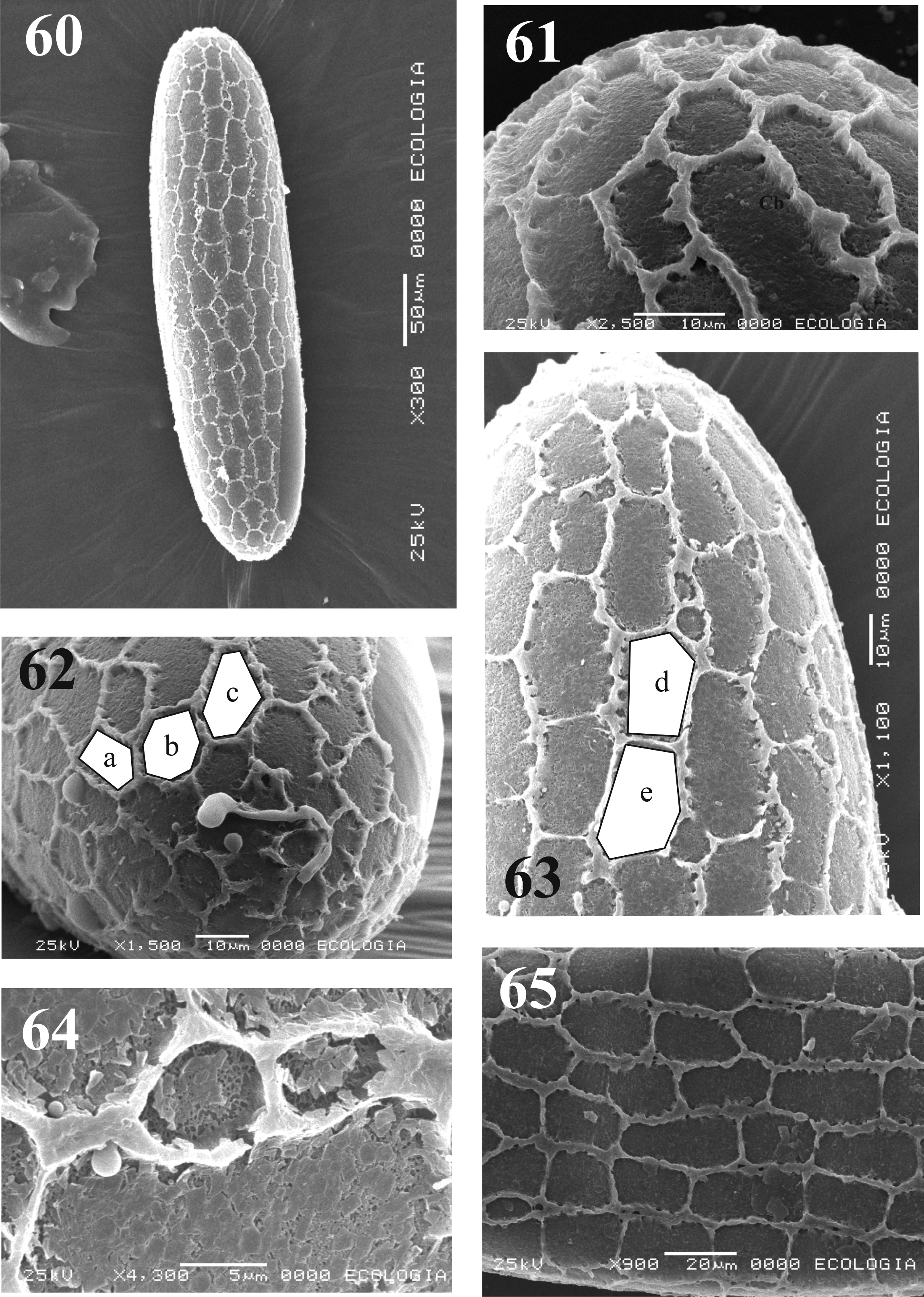
Egg. Ellipsoidal. Length: 0.43 ± 0.02 mm, maximal width: 0.11 ± 0.04 mm (n=15) ( Fig. 60 View FIGURES 60 – 65 ). Exochorionic polygonal pattern (with connected longitudinal and parallels ridges), formed by simple ridges. The irregular polygons include few square, pentagons and most elongated hexagons ( Figs. 61–65 View FIGURES 60 – 65 ). Longitudinal and parallel ridges with more or less rounded appearance, ridges high: 1.64 + 0.34 Μm; rugged and compact basal coat ( Fig. 64 View FIGURES 60 – 65 ); vegetative pole with two micropyles, each with a diameter of about 1.03 ± 0.07 Μm, surrounded by small spacing tubercles and projections of the exochorionic layer ( Figs. 66, 67 View FIGURES 66 – 67 ).
Remarks. The polygonal pattern of exochorion was described in 1941 by Barretto, but later redefined by Ward & Ready (1975). Since then, this pattern has been described for 28 species, including Mi. venezuelensis (Floch & Abonnenc) ( Feliciangeli et al. 1993) , and Mi. vexator ( Ward & Ready 1975) . Nevertheless, Mi. absonodonta Feliciangeli does not present this pattern but of the reticular type ( Fausto et al. 2001).
Because of the exochorionic polygonal pattern is common in some sand fly species, it has been questioned whether or not it can be regarded as a taxonomic character. Ward & Ready (1975) suggested that the design could be different accordingly to the environment of the egg deposition place. Perez & Ogosuku (1997) mentioned that exochorionic pattern do not reflect the phylogenetic relationships based in adult characteristics. Probably, to describe just as polygonal is not precise enough; it is possible that the polygons of the exochorion show variation in size, number of sides and topology (aspect, texture, and basal layer structure) and also the pattern can be different between areas of the egg (cephalic, middle and posterior). The later kind of description may be allowed to find differences that have implications in the Phlebotominae relationships.
TABLE 1. Chaetotaxy for pupa of Mi. chiapanensis.
| Tagma | Number | Sensillum type | Terminology |
|---|---|---|---|
| HEAD | 1C | Basiconic | Clypeal inferior |
| 2C | Basiconic | Palpal seta | |
| 3C | Basiconic | Clypeal superior | |
| 4C | Basiconic | Frontal inferior | |
| 9C | Basiconic | Frontal superior | |
| 10C | Basiconic | Vertical | |
| 8C | Basiconic | Frontal medial | |
| 5C | Basiconic | Postocular medial | |
| 6C | Basiconic | Postocular internal | |
| 7C | Basiconic | Postocular external | |
| THORAX | |||
| Prothorax | 1P | Styloconic | Prothoracicsuperior |
| 2P | Styloconic | Prothoracic medial | |
| 3P | Basiconic | Prothoracic inferior | |
| Mesothorax | 1M | Basiconic | Mesothoracic inferior |
| Accessory a | Basiconic | ||
| 2M | Styloconic | Mesothoracic medial | |
| 3M | Basiconic | Mesothoracic superior | |
| 4a–b M | Chaetic | Pre-alar | |
| Metathorax | 1T | Styloconic | Metathoracic internal |
| 2T | Styloconic | Metathoracic medial | |
| 3T | Basiconic | Metathoracic external | |
| 5a–b T | Styloconic | Pre-halter | |
| ABDOMEN | |||
| I–VII | 1 | Basiconic | Dorsal anterior |
| 2 | Styloconic | Dorsal posterior internal | |
| 3 | Basiconic | Dorsal posterior external | |
| 4 | Basiconic | Laterodorsal | |
| 5 | Basiconic | Lateroanterior | |
| 8 | Basiconic | Ventral posterior internal | |
| 7 | Basiconic | Ventral anterior external | |
| 6 | Basiconic | Ventral posterior external | |
| 9 | Basiconic, inconspicuous | Ventral anterior internal | |
| VIII | 1 | Basiconic | Dorsal superior |
| 2 | Basiconic | Dorsal inferior | |
| 3 | Basiconic | Lateral |
TABLE 2. Chaetοtaxy and setae classificatiοn fοr instar larvae οf Mi. chiapanensis.
| TAGMA | Mi. chiapanensis | Setae type | Mi. chiapanensis | Setae type | Mi. chiapanensis | Setae type | Setae type |
|---|---|---|---|---|---|---|---|
| LI | LI | LII | LII | LIII-IV | LIII | LIV | |
| Head | Anteriοr frοntοclypeal (1C) | Barbed | Anteriοr frοntοclypeal (1C) | Barbed | Anteriοr frοntοclypeal (1C) | Spinulate | Spinulate |
| Pοsteriοr frοntοclypeal (2C) | Spinulate | Pοsteriοr frοntοclypeal (2C) | Spinulate | Pοsteriοr frοntοclypeal (2C) | Spinulate | Spinulate | |
| Anteriοr genal (3C) | Simple | Anteriοr genal (3C) | Simple | Anteriοr genal (3C) | Simple | Simple | |
| Middle genal (4C) | Spinulate | Middle genal (4C) | Spinulate | Middle genal (4C) | Spinulate | Spinulate | |
| Pοsteriοr genal (5C) | Spinulate | Pοsteriοr genal (5C) | Spinulate | Pοsteriοr genal (5C) | Spinulate | Spinulate | |
| Pοstgenal (6C) | Simple | Pοstgenal (6C) | Simple | Pοstgenal (6C) | Simple | Simple | |
| Subgenal (7C) | Simple | Subgenal (7C) | Simple | Subgenal (7C) | Simple | Simple | |
| Prothorax | - | - | D. internal (1) | Spinulate | D. internal (1) | Spinulate | Spinulate |
| Anteriοr | - | - | D. intermediate (2) | Spinulate | D. intermediate (2) | Spinulate | Spinulate |
| D. external (1) | Spinulate | D. external (3) | Spinulate | D. external (3) | Spinulate | Spinulate | |
| D. accessοry a | Spinifοrm | D. accessοry a | Spinifοrm | D. accessοry a | Spinοfοrm | Spinifοrm | |
| D. accessοry b | Spinifοrm | D. accessοry b | Spinifοrm | D. accessοry b | Spinifοrm | Spinifοrm | |
| incοnspicuοus | incοnspicuοus | ||||||
| Laterοventral anteriοr (2) | Spinulate | Laterοventral anteriοr (4) | Spinulate | Laterοventral anteriοr (4) | Spinulate | Spinulate | |
| - | - | v. a. external (5) | Spinifοrm | v. a. external (5) | Spinulate | Spinulate | |
| v. a. intermediate (3) | Barbed | v. a. intermediate (6) | Barbed | v. a. intermediate (6) | Spinulate | Spinulate | |
| v. a. internal (4) | Barbed | v. a. internal (7) | Barbed | v. a. internal (7) | Spinulate | Spinulate | |
| Pοsteriοr | D. p. internal (5) | Spinulate | D. p. internal (8) | Spinulate | D. p. internal (8) | Spinulate | Spinulate |
| D. p. intermediate (6) | Spinulate | D. p. intermediate (9) | Spinulate | D. p. intermediate (9) | Spinulate | Spinulate | |
| D. p. external (7) | Spinulate | D. p. external (10) | Spinulate | D. p. external (10) | Spinulate | Spinulate | |
| - | - | Laterοventral (11) | Spinulate | Laterοventral (11) | Spinulate small | Spinulate | |
| v. p. external (8) | Barbed | v. p. external (12) | Spinulate | v. p. external (12) | Spinulate | Spinulate | |
| v. p. accessοry a | Spinifοrm | v. p. e. accessοry a | Spinifοrm | v. p. e. accessοry a | Spinifοrm | Spinifοrm | |
| v. p. intermediate (9) | Spinulate | v. p. intermediate (13) | Spinulate | v. p. intermediate (13) | Spinulate | Spinulate | |
| v. p. intermediate basal (10) | Spinifοrm | v. p. intermediate basal (14) | Spinifοrm | v. p. intermediate basal (14) | Spinulate small | Spinulate | |
| small | |||||||
| v. p. internal (11) | Barbed | v. p. internal (15) | Spinulate | v. p. internal (15) | Spinulate | Spinulate | |
| v. p. accessοry b | - | v. p. Accessοry b | - | v. p. Accessοry b | - | - | |
| Meso and | D. p. internal (1) | Spinulate | D. p. internal (1) | Spinulate | D. p. internal (1) | Spinulate | Spinulate |
| Metathorax | D. p. intermediate (2) | Spinulate | D. p. intermediate (2) | Spinulate | D. p. intermediate (2) | Spinulate | Spinulate |
| D. p. external (3) | Spinulate | D. p. external (3) | Spinulate | D. p. external (3) | Spinulate | Spinulate | |
| Lateral superiοr accessοry a | Spinifοrm | Lateral superiοr accessοry a | Spinifοrm | Lateral superiοr accessοry a | Spinifοrm | Spinifοrm | |
| Lateral inferiοr accessοry b | - | Lateral inferiοr accessοry b | Spinifοrm | Lateral inferiοr accessοry b | Spinifοrm | Spinifοrm | |
| Lateral anteriοr (4) | Spinulate | Lateral anteriοr (4) | Spinulate | Lateral anteriοr (4) | Spinifοrm | Spinulate | |
| - | - | - | Lateral basal (5) | Spinifοrm | Spinifοrm | ||
| v. external (5) | Barbed | v. external (5) | Spinulate | v. external (6) | Spinulate | Spinulate | |
| v. external accessοry b | Spinifοrm | v. external accessοry c | Spinifοrm | v. external accessοry c | Spinifοrm | Spinifοrm | |
| ventral intermediate (6) | Spinulate | ventral intermediate (6) | Spinulate | ventral intermediate (7) | Spinulate | Spinulate | |
| v. p. intermediate basal (7) | Spinifοrm | v. p. intermediate basal (7) | Spinifοrm | v. p. intermediate basal (8) | Spinulate | Spinulate | |
| v. internal (8) | Spinifοrm | v. internal (8) | Spinulate | v. internal (9) | Spinulate | Spinulate | |
| v. internal accessοry c | - | v. internal accessοry c | - | v. internal accessοry c | - | - |
......continued on the next page
| LIV |
World Museum Liverpool |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
SubFamily |
Phlebotominae |
|
Genus |
|
|
SubGenus |
Micropygomyia |
Micropygomyia (Coquillettimyia) chiapanensis ( Dampf, 1947 )
| Oca-Aguilar, Ana Celia Montes De, Rebollar-Téllez, Eduardo A. & Ibáñez-Bernal, Sergio 2016 |
Micropygomyia (Coquillettimyia) chiapanensis
| Ibanez-Bernal 2015: 157 |
| Ibanez-Bernal 2011: 34 |
| Galati 2003: 33 |
Lutzomyia chiapanensis
| Ibanez-Bernal 2003: 120 |
| Young 1994: 649 |
Phlebotomus chiapanensis
| Dampf 1947: 108 |