Otiorhynchus (Metopiorrhynchus) singularis (Linnaeus, 1767)
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4108.1.1 |
|
publication LSID |
lsid:zoobank.org:pub:B802F2B1-944E-4B84-A856-8091E60D88FC |
|
DOI |
https://doi.org/10.5281/zenodo.6062730 |
|
persistent identifier |
https://treatment.plazi.org/id/03EB857D-FFEB-AD19-0592-FE4942DB15DF |
|
treatment provided by |
Plazi |
|
scientific name |
Otiorhynchus (Metopiorrhynchus) singularis (Linnaeus, 1767) |
| status |
|
Otiorhynchus (Metopiorrhynchus) singularis (Linnaeus, 1767) View in CoL
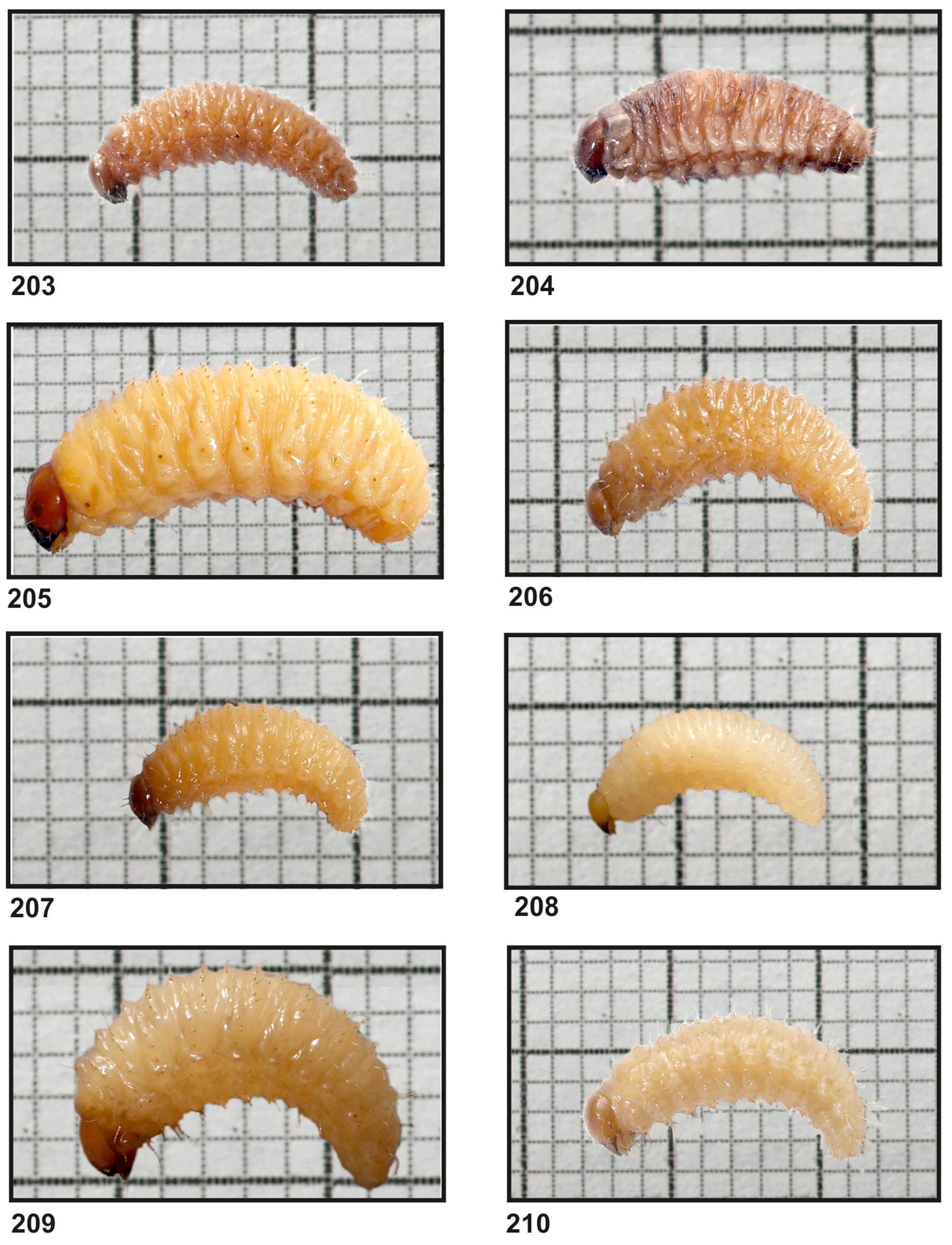
Material examined: 18 larvae ( Fig. 208 View FIGURES 203 – 210 ). Hannover ( Germany), breeding in 2 Bergenia cordifolia cultivar ‘Rotblum’ flowerpots, 18.12.2011, 2 ex.; dto., 23.02.2012, 1 ex.; Ellerhoop, greenhouse, breeding by Thorsten Ufer in Thuja occidentalis flowerpots, 17.01.2012, 3 ex.; JKI, Braunschweig, climate chamber, breeding in Euonymus fortunei flowerpots, 0 1.11.2012, 1 ex.; dto., 13.12.2012, 2 ex.; dto., 10.01.2013, 1 ex.; dto., breeding in Taxus baccata L. flowerpots, 14.03.2013, 1 ex.; dto., Thuja occidentalis cultivar ‘Smaragd’, 18.04.2013, 1 ex.; Hannover-Herrenhausen, Berggarten, bed of Waldsteinia geoides Willd. , 25.06.2015, 5 ex.; 0 4.06.2015, 1 ex.
Remarks about breeding and development. Only a relatively small number of larvae were obtained from 2 breeding attempts in 2011 and 2012. The exact number of all observed larvae cannot be given here, as several larvae were kept to obtain pupae (but in vain) and several larvae later proved to be in fact Otiorhynchus sulcatus (due to a contamination of some of the plants used for breeding).
In 2011 45 eggs, deposited on 18.08.2011, were released in 2 Bergenia cordifolia ‘Rotblum’ flowerpots and kept in the Curculio-Institut in Hannover at room temperature; already in December (second half) 2 mature larvae were obtained. In 2012 breeding was started on 14.05. with 5 flowerpots (1 Euonymus fortunei , 2 Taxus baccata , 2 Thuja occident alis ‘Smaragd’) with 5 adults and around 150 eggs. The first mature larva was received on 0 1.11.2012, the last on 18.04.2013. In March 2013 the flowerpots were placed in a room outside to induce pupation, but air temperature fell and permanent frost killed the larvae. The pupa of this widespread species is still undescribed.
The life history of Otiorhychus singularis is best studied by Willis (1964) and his data are only partly in accordance with data from other authors, who considered only definite parts of the life-cycle or reported about the situation in other regions (e.g. Andison 1942). There are several peculiarities in the life-cycle of this species. Adults start to emerge in April; also in May usually mainly teneral light brownish adults appear on the ground or in the woody vegetation. These specimens did not hatch just before from the pupa (as e.g. in O. sulcatus ). They have been overwintering in their pupal chambers (as in O. indefinitus and in O. raucus ), and their exoskeleton remained weak since the point in time when they hatched from the pupa. In Northern Ireland pupation takes place from end of June to August ( Willis 1964). Larvae and pupae were found at different depths in soil, but usually deeper than O. sulcatus , mainly 9–15 inches (= 23–38 cm) below soil level. In contrast to all other Otiorhynchus species so far investigated, O. singularis has two definite oviposition periods: from (approximately) mid-May to June and with lower significance from mid-September to mid-October (Sprick 2012, in Sprick & Stüben 2012). This partly corresponds with observations of Dieckmann (1980), who dissected numerous weevils and found eggs only once (19th June). Another striking difference to the life history of other Otiorhynchus species is the long life span over 3 years. In the lab 2 specimens also survived during the whole period of the soil-dwelling weevils project of the Curculio Institute (28.04.2008 – 23.02.2012). Under harder outdoor conditions more than a second overwintering is not to be expected. Willis (1964) confirms that adults usually overwinter and contribute to egg-laying in a second summer. It seems that limited egg deposition periods and the long stay in the pupal chamber over 8 to 10 months is an adaptive synchronization to the simultaneous presence of more than one generation, although this species is parthenogenetic and there is no need to mate.
In spring this species showed a conspicuous amount of climbing activity during the day (Sprick 2012, in Sprick & Stüben 2012) and could be recorded per beating tray from shrubs and trees up to over 2.50 m.
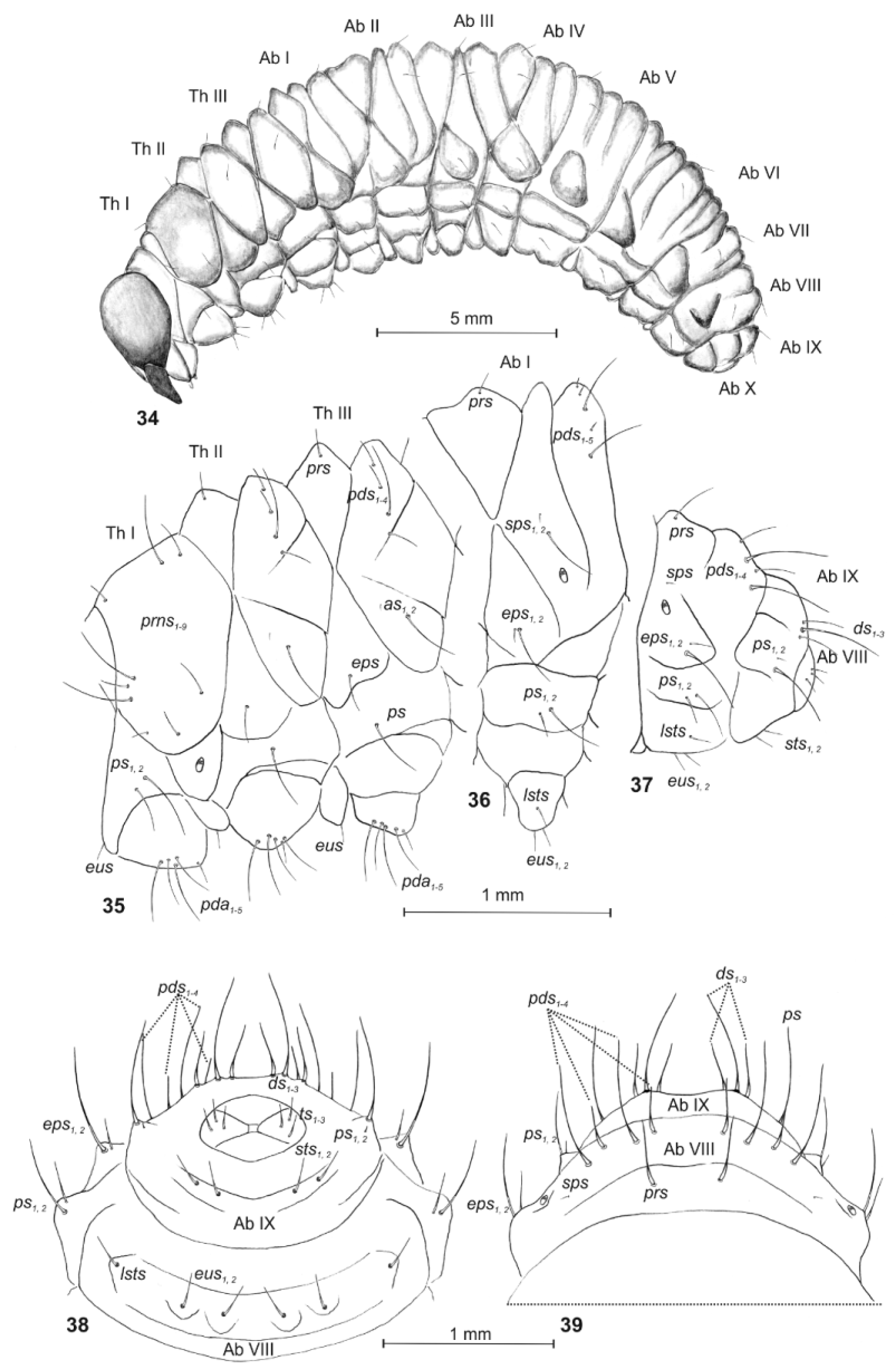
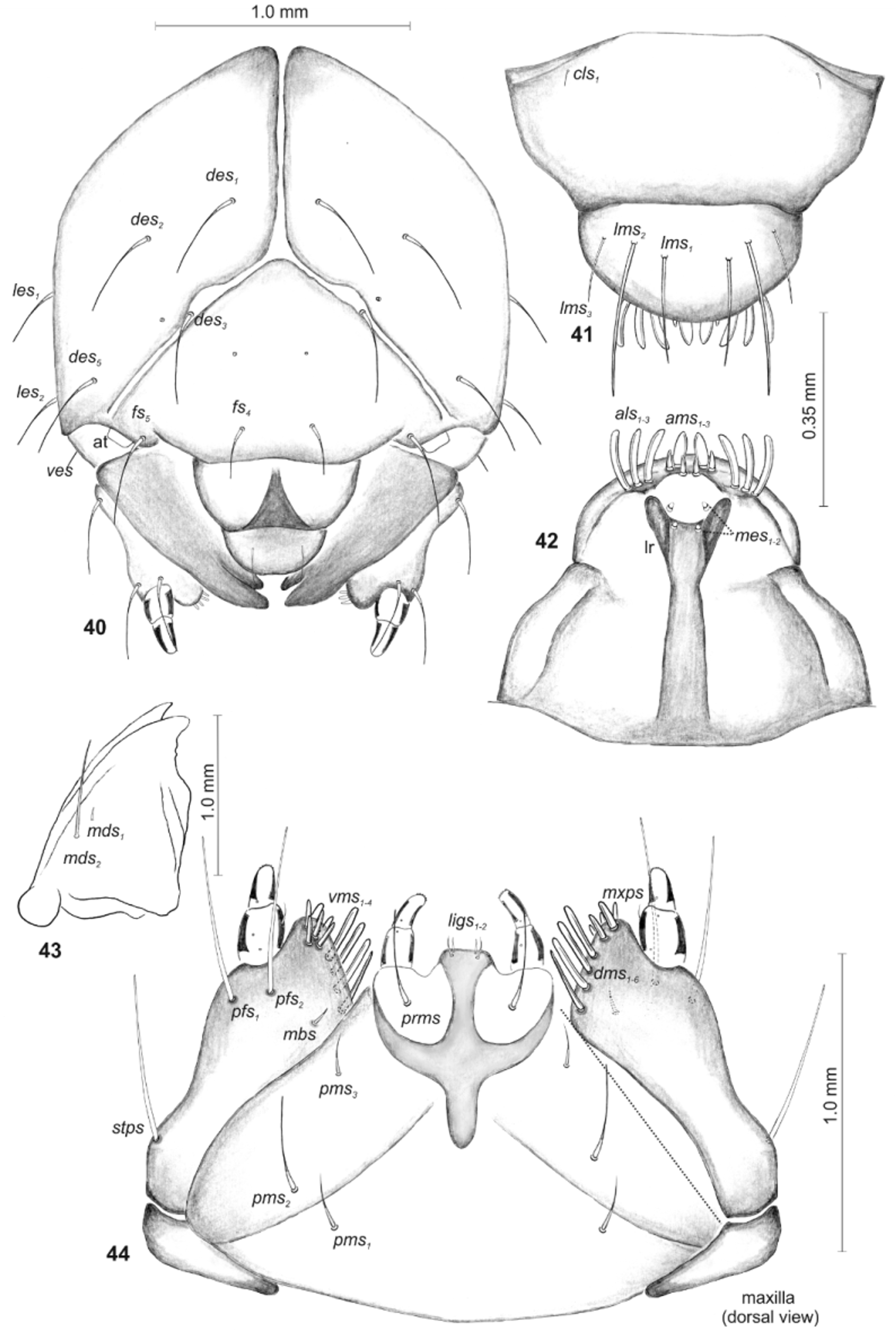
Description ( Figs. 34–44 View FIGURES 34 – 39 View FIGURES 40 – 44 ).
Coloration. Head dark yellow; all thoracic and abdominal segments from dirty yellow to brownish; cuticle with cuticular processes.
Body elongated ( Fig. 34 View FIGURES 34 – 39 ). Chaetotaxy. Setae different in length, relatively short, filiform, greyish. Thorax. Prothorax with 3 long and 6 medium long to short prns; and 2 ps, different in length. Mesothorax with 1 medium long prs; 4 pds (ordered: 2 medium long, 1 long and 1 medium long); 1 long and 1 minute as; 1 eps; and 1 ps. Chaetotaxy of meso- and metathorax similar. Each pedal area of thoracic segments well isolated, with 5 pda, different in length. Each thoracic segment with 1 short eus ( Fig. 35 View FIGURES 34 – 39 ). Abdomen. Abdominal segments I–VIII with 1 short prs; 5 pds (ordered: 2 very short, 1 long, 1 very short and 1 long); 1 long and 1 minute sps [abd. seg. VIII with 1 minute sps and only 4 pds (1 long, 1 medium long, 1 long and 1 medium long)]; 1 long and 1 minute eps; 2 ps, different in length; 1 short lsts; and 2 eus ( Figs. 36, 38, 39 View FIGURES 34 – 39 ). Abdominal segment IX with 3 ds, different in length; 1 very long and 1 minute ps; and 2 sts, equal in length ( Figs. 37–39 View FIGURES 34 – 39 ). Each lateral lobe of abdominal segment X with 3 minute ts.
Head subglobose, slightly narrowed bilaterally ( Fig. 40 View FIGURES 40 – 44 ). Head capsule with 4 relatively long des, des 1 and des 2 placed on central part of epicranium, des 3 located on frontal suture, des 5 located anterolaterally; 2 relatively long fs, fs 4 placed anteromedially, fs 5 located near to antenna (fs 4 slightly shorter than fs 5); 2 relatively long les and 1 relatively long ves. Postepicranial area with a pair of sensilla, frons with a pair of sensilla. Stemmata not well visible. Antennal segment membranous, bearing 1 conical sensorium and 3–4 filiform sensilla. Clypeus 2.1 times as wide as long with 1 cls, placed posterolaterally ( Fig. 41 View FIGURES 40 – 44 ). Labrum about 1.9 times as wide as long with 3 straight lms of different length, placed medially or mediolaterally; lms 3 distinctly shorter than other setae, all lms exceeding the outline of the labrum; the anterior margin of labrum rounded ( Fig. 41 View FIGURES 40 – 44 ). Epipharynx with 3 fingerlike als, different in length, 3 ams, different in length, and 2 very short mes; labral rods (lr) short, reniform, strong convergent ( Fig. 42 View FIGURES 40 – 44 ). Mandible ( Fig. 43 View FIGURES 40 – 44 ) feebly bifid, teeth almost of equal height; with 2 mds, different in length; internal edge with a triangular tooth. Maxilla ( Fig. 44 View FIGURES 40 – 44 ) with 1 very long stps and 2 very long pfs, placed ventrolaterally; 1 short mbs, situated ventrally. Mala with 6 dms, different in length and 4 straight vms, almost equal in length. Maxillary palpi with two palpomeres; basal palpomere larger than distal and with 1 mxps; distal palpomere with a group of 6 conical, cuticular apical processes; each palpomere with a sensillum. Praelabium heart-shaped ( Fig. 44 View FIGURES 40 – 44 ), with 2 very short ligs and 1 long prms. Labial palpi with two palpomeres, relatively elongated; both palpomeres almost equal in length; praemental sclerite well visible. Postlabium with 3 pms, different in length; pms 1 and pms 3 equal in length, almost 2 times shorter than pms 2 ( Fig. 44 View FIGURES 40 – 44 ).
Differential diagnosis. See “Key to larvae of selected Otiorhynchus species” and Tables 1, 2.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
SubFamily |
Entiminae |
|
Genus |