Cruziohyla craspedopus ( Funkhouser, 1957 )
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4450.4.1 |
|
publication LSID |
lsid:zoobank.org:pub:54B89172-7983-40EB-89E9-6964A4D4D5AC |
|
DOI |
https://doi.org/10.5281/zenodo.5953567 |
|
persistent identifier |
https://treatment.plazi.org/id/03EC87FD-FFFE-FF92-C2B2-F915442EF8A8 |
|
treatment provided by |
Plazi |
|
scientific name |
Cruziohyla craspedopus ( Funkhouser, 1957 ) |
| status |
|
Cruziohyla craspedopus ( Funkhouser, 1957)
( Figs. 1c View FIGURE 1 , 6c View FIGURE 6 , 7c View FIGURE 7 , 9c View FIGURE 9 ).
Fringed Tree Frog / Rana Arbol Franja
Phyllomedusa craspedopus— Funhouser (1957).
Agalychnis craspedopus— Duellman (1968): Transferred from Phyllomedusa .
Cruziohyla calcarifer— Faivorvich et al (2005): Transferred from Agalychnis .
Holotype. SU 10310. Adult female from Chicherota , Rio Bobonaza, Napo-Pastaza Province, eastern Ecuador.
Diagnosis. Cruziohyla craspedopus is a moderate-sized species of the genus Cruziohyla , SVL in males 53 mm to 66 mm and 68.3 mm to 75.5 mm in females, distinguished by a combination of the following characters: (1) snout is sharply truncate in profile; (2) lacking dark ventral thigh markings either side of the vent; (3) modal webbing formula (fingers): I 2 -—2- II 2 -— 2 III 2-— 2- IV; modal webbing formula (toes): I 1— 1 II 1— 2 III 1— 3 IV 3— 1 V; (4) in life, dark green background colour to very smooth dorsal surface, overlaid with a network of large irregular pale blue-grey blotches often with a thin cobweb-like pattern; (5) head is slightly wider than long; (6) narrow black vertical bands extend on lateral surfaces of the flanks to margin of underside; (7) in life, undersides and concealed surfaces of the flanks and legs are yellow; (8) sharply truncate snout; (9) presence of extensive dermal appendages forming irregular spurs along the posterior margin of the tarsus; (10) dermal fold to fifth toe and on forearm; (11) presence of a dermal fold around margin of lower jaw.
Distribution: East of the Andes in Colombia, Ecuador, Peru and Brazil.
Comparison of C. sylviae sp. n. with the congeners. The morphological features of C. sylviae sp. n., have been previously covered by Duellman (1970), and Savage (2002) under the name C. calcarifer . However, Cruziohyla sylviae sp. n. can be distinguished from both C. calcarifer and C. craspedopus by the following:
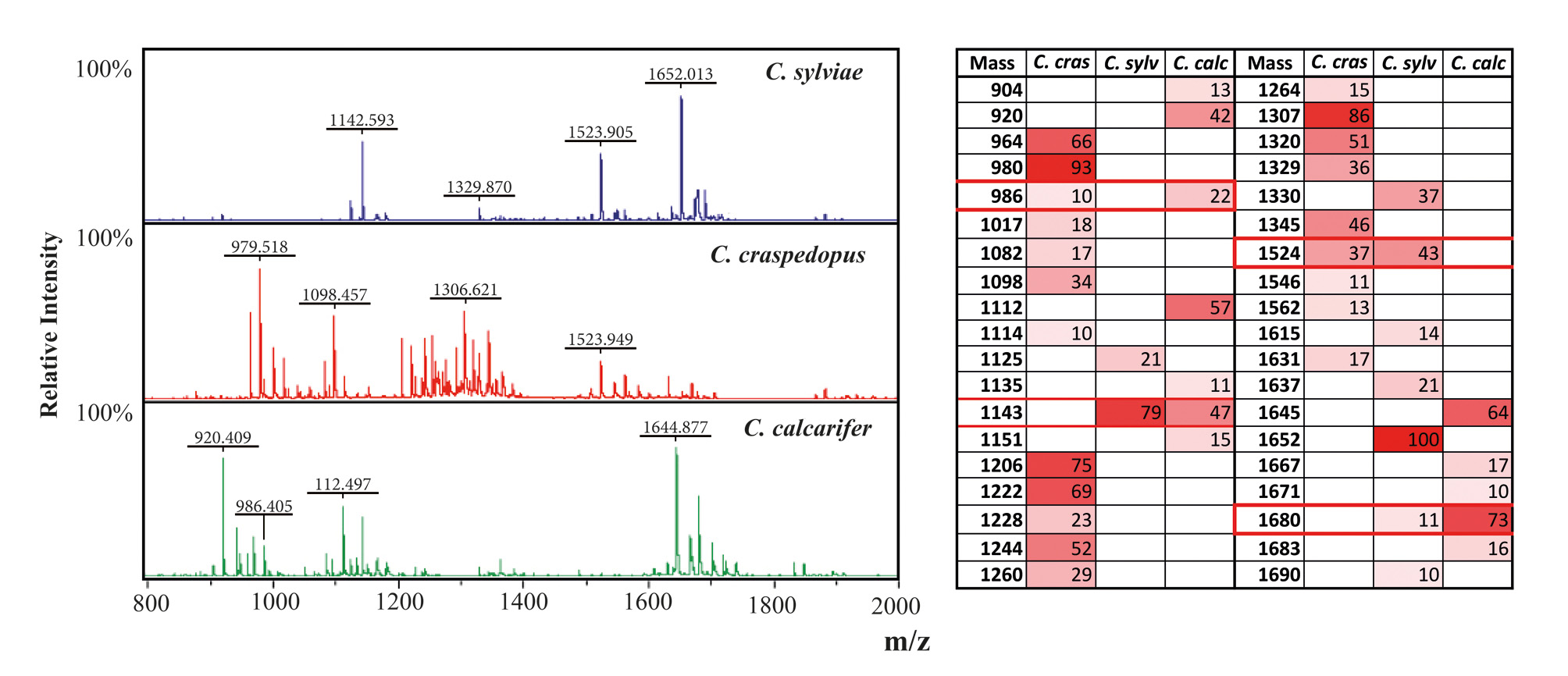
Peptide fingerprinting. In examining the signals it can be seen that the majority of these appear unique to each species, with the greatest commonality seen between C. sylviae sp. n. and C. calcarifer ( Fig. 4a View FIGURE 4 ). Although the number of replicates included are not sufficient to determine a confident diagnostic pattern, the differences do display a clear compositional difference between C. sylviae sp. n. and C. calcarifer ( Fig. 4a View FIGURE 4 ).
Ventral markings. Cruziohyla sylviae sp. n. can be distinguished from C. calcarifer in lacking dark pigmented markings found either side of the vent ( Fig 5b View FIGURE 5 ). This feature is a key character of C. calcarifer and is not present in any other species of Cruziohyla . All specimens of C. calcarifer carry the feature to some degree, with it extending to the ventral parts of the thighs in larger specimens: A juvenile [KHJ884] from Esmeraldas, Ecuador, at 25 mm SVL, has small dark markings either side of the vent, whilst in those collected from Bocas del Toro, Panama, by Myers and Duellman (1982), 71.5–87.0 mm SVL, the markings extend over a wide surface area of the thigh region (Appendix I). All eleven specimens of C. calcarifer collected and detailed by Myers and Duellman (1982) [AMNH A107238–107248] carry the characteristic dark ventral markings to a high degree. The feature is also detailed in the field notes of Dr Myers at the time of specimen collection (April 21st, 1980: AMNH unpublished). The largest female specimen [AMNH A107238] has the most extensive thigh markings seen, thus it can be confirmed that this character is as strongly marked in females as in males.
The dark markings, noted by Cochran and Goin (1970) in reference to the second C. calcarifer specimen detailed by Boulenger (1913) [BM:1913.11.12.126] from Colombia, fade little with age in preservative and are clearly seen as distinct patches either side of the vent on the holotype, and on the specimen from Colombia where they extend to the thighs (Appendix I).
The exclusive presence of this feature in association with key characters seen in all genetically defined C. calcarifer specimens confirms it as a strong and reliable taxonomic character for that species. Therefore, the evidence of differences in ventral markings clearly distinguish C. sylviae sp. n from C. calcarifer . Close up examination with a Zeiss microscope at x 40 magnification clearly highlights gland ducts throughout the darkly pigmented ventral granulations ( Fig. 5c View FIGURE 5 ). However, further work is required to establish the function of this unusual feature.
Head morphology and tympanum width to the diameter of eye (TY/E). C. sylviae has a sloping snout when viewed in lateral profile compared to a sharply truncated snout possessed by C. craspedopus ( Funkhouser, 1957) , and in dorsal profile the snout of C. sylviae sp. n. is more rounded compared with that of C. calcarifer , which is broadly triangular and has an almost vertical loreal region ( Cochran & Goin, 1970).
Cruziohyla sylviae sp n. has a noticeably large tympanum, ranging between 70–100% the diameter of the eye compared with a tympanum size of 40–60% the diameter of the eye in C. calcarifer : the first C. sylviae sp. n. specimen collected, from Barro Colorado Island, Panama, has a tympanum diameter approximately the same size as the eye [ZA (MCZ) 15610]; one specimen from La Mosquitia region [SMF 86287], and two other specimens from Gracias a Dios, Honduras [USNM 559748;USNM 563933] also have tympani approximately the size of the eye ( Moen, 2012). The TY/E of C. sylviae sp. n. specimens from the Guayacán, Limon Province, and Puerto Viejo de Sarapiqui, Heredia Province, Costa Rica are comparable: male TY/E 0.728–0.833 (mean 0.784; n = 17); female TY/E from 0.878–0.974 (mean 0.933; n = 10). These sizes are also comparable with the measurements described by others for C. sylviae sp. n. from Costa Rica, having TY/E of 0.750–0.965 ( Savage, 2002; Duellman, 1970), the latter being a female of 78.5 mm [USC 496] having a tympanum diameter approximately the same diameter as the eye ( Duellman, 1970).
The holotype of C. calcarifer (BM.1902.5.27.26), a small female (57mm) from Esmeraldas, Ecuador, has a tympanum half the diameter of the eye ( Boulenger, 1902), as does a large female (87 mm) from Changuinola, Panama [AMNH 107238]. A wild male C. calcarifer specimen from Alto Tambo, Esmeraldas Province, Ecuador, has TY/E 0.540, and male specimens from Esmeraldas province in captivity at Manchester Museum [MM 1019– 1021] have TY/E 0.533–0.548 (mean 0.541, n = 3). Specimens from Colombia have TY/E to 60% (0.600): two live specimens, from near San Cipriano and Buenaventura, Valle de Cauca, Colombia, have TY/E of 0.417 and 0.567 respectively, the latter being comparable to the 60% ratio given by Cochran and Goin (1970) for the first specimen collected from Colombia [BM 1913.11.12.126]. Allowing for any sexual dimorphism within the overall TY/E ranges identified ( C. sylviae sp. n: 70–100% TY/E; C. calcarifer : 40–60% TY/E), the differing TY/E support the identification of C. sylviae sp. n.
Hands and feet. Cruziohyla sylviae sp. n. and C. calcarifer can both be distinguished from C. craspedopus in lacking the broad, well-developed, fringes found along the posterior margin of the distal part of the foot and fifth toe in that species ( Funkhouser, 1957; Duellman, 1970; Hoogmoed & Cadle, 1991; Meneghelli et al., 2011). Distinctions between C. sylviae sp. n. and C. calcarifer include C. sylviae sp. n. having webbing that extends to the penultimate phalanx on the 4th toe ( Duellman, 1970) whereas C. calcarifer has more moderate webbing that extends to the antepenultimate phalanx on the 4th toe ( Cochran & Goin, 1970) ( Fig. 6 View FIGURE 6 ).
Colouration comparison. All Cruziohyla species are capable of considerable metachrosis, however overall dorsal colouration and patterning is fully consistent when compared intraspecifically and distinctly different when compared interspecifically: C. sylviae sp. n. has a dorsal surface colouration consisting of two shades of green, with an areolated texture, giving a very finely granulated appearance, and also having small pale green lichenose speckles throughout; C. craspedopus has a uniform dark green dorsal surface background colouration with irregular-shaped large pale blue-grey lichenose blotches throughout; C. calcarifer has a consistent uniform green background colouration, often with a fine scattering of white or pale-blue spots ( Fig. 7 View FIGURE 7 ), and possesses a small yellow coloured spot directly posterior to the eye on the very rim. This marking, which is concealed when the frog is at rest, is lacking in both C. craspedopus and C. sylviae sp. n.
Flank barring. All members of the genus Cruziohyla have skin colourations that include a degree of black barring to the orange-yellow flanks, also having similar colourations on the thighs, foot, forearm and outer finger ( Funkhouser, 1957, Duellman, 1970; Savage 2002; Kubicki, 2004). Cruziohyla . craspedopus has narrow black lines on the flanks ( Funkhouser, 1957), C. sylviae sp. n. has barring that form black vertical bands ( Kubicki, 2004), and C. calcarifer has black barring that is short and limited ( Boulenger, 1902). All the examined specimens of C. calcarifer from northwestern Ecuador to southeastern Costa Rica have black barring down the side lateral surface, forming short vertical finger-like bars extending approximately halfway down the flanks. By comparison, specimens of C. sylviae sp. n. and C. craspedopus have black vertical band patterning on their flanks extending further than halfway down the lateral surface, often extensive and interconnecting ( Fig. 11 View FIGURE 11 ).
Dorsal colouration. Cruziohyla sylviae sp. n. is considered a dark green frog ( Savage, 2002). Small green (GK113-114) lichenose speckles are present throughout the whole of the dorsal surface, extending to the forearm, shanks, tarsi, fourth fingers, and fifth toes ( Duellman, 1970) ( Fig. 7a View FIGURE 7 ). The pale green speckling on the dorsum is similar, but smaller, to the larger pale blue-grey lichenose blotches found on C. craspedopus ( Hoogmoed & Cadle, 1991) . Hoogmoed and Cadle (1991) make specific reference to the dorsal coloration of a C. sylviae sp. n. specimen from near Bribri, Limón Province, Costa Rica [ANSP 32501], separately to that of specimens of C. calcarifer (e.g. AMNH 16248) collected in Changuinola, Panama, by Myers and Duellman (1982).
In the description of the C. calcarifer specimen collected by Dr Spurrell in Condoto, Chocó, Colombia, Boulenger (1913) used the collector’s field notes to highlight the dorsal colour of the specimen concerned (BM.1902.5.27.26 [BM. 1947.2.24.22]) as it was found in life: that the skin above is smooth and sage-green ( Boulenger, 1913, pg.1023). There are no discernible differences between that specimen and the type, collected from the Rio Durango, Esmeraldas Province, northwest Ecuador ( Cochran & Goin, 1970). All the specimens of C. calcarifer assessed from Ecuador, Colombia, and western Panama have a uniform green (GK106–107) dorsal skin surface which lacks the lichenose markings found on the other members of the genus ( Fig. 7b View FIGURE 7 ). Specifically, the smooth green dorsal surface of C. calcarifer is most similar to that observed in members of the genus Phynomedusa, the closest known relative of Cruziohyla ( Duellman et al, 2016). Cruziohyla calcarifer does however have a scattering of very fine white or pale blue spots (GK191) to the dorsal surfaces, previously recognised by Myers and Duellman (1982) and reported on by Ibáñez et al. (1999). The white or pale-blue spots found on C. calcarifer contrast significantly when the consistent green dorsal surfaces darken through metachrosis ( Fig. 11 View FIGURE 11 ).
Cruziohyla craspedopus has very smooth uniform dark green background coloration to the dorsal surfaces together with a network of large, highly-contrasting, pale greyish-blue coloured lichenose markings. These vary from thin cobweb-like patterns to large irregular blotches ( Funkhouser, 1957; Hoogmoed & Cadle, 1991; Nunez et al., 2015). The dorsal markings of specimens from Yasuní, Ecuador, and specimens from near Nasuta, in Loreto, Peru [MM 1014–1018] are similarly marked, with large pale grey-blue lichenose blotches (GK287) contrasted against a dark green (GK132) dorsal background ( Fig. 7c View FIGURE 7 ).
Pattern development. The development of the skin colourations from juvenile to adult in all species of Cruziohyla varies considerably: Juvenile C. craspedopus show typical lichose markings on their dorsal surface before metamorphosis, and their juvenile colouration is similar to that of the adults; juveniles of C. calcarifer show bright yellow markings above the mouth and on the lower eyelid, which changes to green as adult; Juvenile C. sylviae sp. n. show no yellow markings above the mouth or on the lower eyelid, being brown initially and eventually changing green with age ( Kubicki, 2004: sub C. calcarifer ). The extensive barring to the flanks seen in C. sylviae sp. n. and C. craspedopus develops as vertical bands on the sides of both species amid the yellow-orange flank background colour. By comparison, the reduced barring seen in C. calcarifer develops gradually with age with each finger-like bar extending slowly down the flanks from the very margin of where the green dorsal surface meets the orange-yellow flank. These differences in pattern development allow for the clear identification of juvenile C. sylviae sp. n ( Fig. 5a View FIGURE 5 ).
Call comparison. Advertisement calls of Cruziohyla consist of single low-pitched notes repeated at regular intervals. C. sylviae sp. n. has a call (note) duration of 170–215 m /secs (x̄= 190 ± 3.7 m /secs, n = 14) ( Fig. 8 View FIGURE 8 ) compared with the call duration of 105–120 m /secs (x̄= 112.5 ± 2.1 m /secs, n = 6) for C. craspedopus , and 135– 212 m /secs (x̄= 171.1 ± 10.1 m /secs, n = 7) for C. calcarifer . Variances in different recording methods and qualities make overall comparisons of frequency band ranges limited. However, the highest intensity peak (maximum energy output) within the dominant frequency band range for C. sylviae sp. n. is located around 750Hz, compared with C. craspedopus at around 700 Hz and C. calcarifer at around 860 Hz. C. calcarifer also has a similarly intense output at around 950 Hz ( Fig. 9a–c View FIGURE 9 ).
Distribution. Cruziohyla craspedopus occurs east of the Andes and is found in Colombia, Ecuador, Peru and Brazil ( Funkhouser, 1957; Hoogmoed & Cadle, 1991; Ruiz-Carranza et al., 1996; Meneghelli et al., 2011; Venâncio et al., 2014; Moraes et al., 2017). The species is primarily associated with Amazonia and has a distributional range that does not overlap with either C. calcarifer or C. sylviae sp. n.
C. calcarifer occurs from Esmeraldas Province in northwest Ecuador ( Boulenger, 1902; Cisneros-Heredia, 2005; Coloma et al., 2008), through western Colombia at several reported localities ( Boulenger, 1913; Cochran & Goin, 1970; Ruiz-Carranza et al., 1996). In Colombia the species has been recorded in Municipal Mecana in the Chocó and Valle de Cauca ( Vargas & Bolaños, 1999; Lynch & Suárez, 2004; Castro-Herrera & Vargas-Salinas, 2008) and Panama ( Myers & Duellman, 1982; Crawford et al., 2010), to the most southerly part of Costa Rica, where it is found in the Fila Carbon area of Comadre, Limón Province ( Faivovich et al., 2010). Thus, there is considerable overlap in the distribution of these two species ( Fig. 10 View FIGURE 10 ).
All specimens of C. calcarifer collected and reported on by Myers and Duellman (1982) from Changuinola Bocas del Toro, northwestern Panama, have been examined as part of this study and are confirmed as C. calcarifer [AMNH A107238–107248] ( Fig. 11 View FIGURE 11 ). Two specimens from the same locality [KHJ150;KHJ151], one specimen from “Omar Torrios”, El Cope, Panama [USNM 572743 (KRL 800)], and two specimens from Comadre, Costa Rica [KHJ30;KHJ31], are also in agreement with all the features of C. calcarifer specimens from all localities highlighted for the species ( Fig. 10 View FIGURE 10 ). These align with specimens of C. calcarifer from Esmeraldas, Ecuador, e.g. KHJ831, a specimen previously sampled by Faivovich et al. (2010), which may be considered genetically representative of the name bearing population ( Faivovich et al., 2010).
Cruziohyla sylviae sp. n. has a distribution range extending from the Gracias a Dios Province of southeast Honduras, southwards through Nicaragua and Costa Rica to Laguna in the Darien Province of southern Panama ( Fig 11 View FIGURE 11 ). Alto Colorado, near Guayacán, Costa Rica, is the type locality for C. sylviae sp. n., and live specimens observed at the nearby CRARC possess all the characteristics of the species, as do specimens from Alajuela and near Bribri, Costa Rica [UCR15601/ANSP 32501]. The collection of the latter C. sylviae sp. n. specimen, approximately 10 km south from that of C. calcarifer specimens collected at Comadre, Fila Carbon, Costa Rica [KHJ150/KHJ151], suggests that in Costa Rica both species may occur sympatrically ( Fig 10 View FIGURE 10 ). In Panama, both species do occur sympatrically: a specimen of C. sylviae sp. n. is observed (iNaturalist, 2018) 3.5 km from the collection site of C. calcarifer [USNM 572743 (KRL 0800)], at “Omar Torrios”, Panama, where specimens of both species were collected from the very same locality and included in the work of Crawford et al. (2010) and this work ( C. calcarifer : USNM 572743 [KRL 0800] and C. sylviae sp. n. (USNM 572744 [KRL 0781]).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Cruziohyla craspedopus ( Funkhouser, 1957 )
| Gray, Andrew R. 2018 |
Cruziohyla sylviae
| Gray 2018 |
C. sylviae
| Gray 2018 |
C. sylviae
| Gray 2018 |
C. sylviae
| Gray 2018 |
C. sylviae
| Gray 2018 |
C. sylviae
| Gray 2018 |
C. sylviae
| Gray 2018 |
C. sylviae
| Gray 2018 |
C. sylviae
| Gray 2018 |
Cruziohyla sylviae
| Gray 2018 |